
Short-term observation of management of sclera patch grafts used in the scleral defects
Introduction
Sclera has the inherent risk of necrosis and thinning secondary to surgical, inflammatory, and degenerative conditions (1), which may induce scleral defect. Scleral defect is a serious clinical manifestation that can affect the integrity of the eye, including necrotizing scleritis, scleral melting/perforation, scleral staphyloma, and so on. The exposure or prolapse of the underlying uveal tissues, or escape of ulcerative tissue under the condition of progressive scleral necrosis, may lead to the possibility of threatening the integrity of the eye. Treatment includes the use of systemic glucocorticoid, anti-inflammatory drugs, and immunosuppressive therapy. However, when it comes to the scleral defect, surgical repair maybe needed. A variety of human and commercial materials were introduced in scleral patch grafts for managing scleral defects or tectonic instability of the eye. It had been successfully used as scleral grafts in the past decades, including fascia lata, cartilage, cadaveric aortic tissue, tibial periosteum, synthetic Gore-Tex, skin, amniotic membrane, autologous sclera, and homologous sclera (2-6). Processed allograft materials like sclera and pericardium are available commercially. Still, none is universally accepted as the ideal graft material until now. Autologous preserved scleral patch graft remains thicker than commercial materials, which is available from whole-eye donors and can be reserved for months. Therefore, it offers a potential advantage in terms of overall risk of exposure when compared to the other proposed grafts, including the strong, flexible, and the natural curvature of the sclera allowing a better fit to the host defects. Furthermore, donor sclera is easy to handle and is well-tolerated by the host with little inflammatory reaction and rare rejections (3). However, how to avoid complications such as necrosis and melting of the graft, dehiscence, and postoperative endophthalmitis, is still the most critical issue of scleral patch graft surgery. An important factor in that is to undergo epithelization and vascularization of the avascular scleral patch, which is stimulated with the conjunctiva flap covering, or an amniotic membrane graft (3,7,8).
Although rarely required in our hospital, scleral patch grafts have been found helpful for select cases. In this study, we performed scleral patch grafts, combined autologous conjunctival pedicle flap, with/or partial medial rectus translocation/autologous pedicle tenon graft simultaneously according to the patient’s needs. We review the patients and discuss our experience with the use of scleral patch grafts, to explore the safety and effectiveness of sclera patch grafts in the management of scleral defect.
Methods
All patients were referred to The Joint International Eye Center of Shantou University and The Chinese University of Hong Kong from December 2010 to June 2015. Institutional review board approval was obtained, and the study was conducted in accordance of the guide-lines of the Declaration of Helsinki [ethic approval ID: EC 20160616(4)-A11] and the Health Insurance Portability and Accountability Act of 1996. Cases inclusion criteria included necrotizing scleritis, scleral melting/perforation and sclera staphyloma, and exclusion criteria included ocular tumors (e.g., uveal melanoma) and Wegener’s granulomatosis. Surgical methods were sclera patch graft, combined autologous conjunctival pedicle flap, with/or partial medial rectus translocation/autologous pedicle tenon graft at the same time if necessary. Allogeneic sclera, which was glycerol cryopreservation, was provided by the eye bank of Joint International Eye Center of Shantou University and The Chinese University of Hong Kong. Before implantation, it was soaked in three consecutive sets of ringer lactate solution for 10 minutes each, then 5% povidone iodine for 10 minutes, and finally in tobramycin solution 20 mg/mL for 10 minutes. Then, the surgeon custom cut the donor sclera to accommodate to the size needed.
In the chart review, recorded data included patient medical history, clinical findings, and ancillary tests. These included ultrasonography, ultrasound biomicroscopy, optical coherence tomography, photography (slit-lamp and fundus). In addition, patients’ systemic evaluations, operative records, histopathologic results, follow-up period, complications, and final outcomes were reviewed. All cases were performed by a single surgeon (Y Yao). Main outcomes were defined as structural integrity, best corrected visual acuity (BCVA), scleritis remission, stability of the grafts (sclera rejection and melt), and ocular symptoms.
Surgical procedures (e.g., Figure 1, Patient 5):
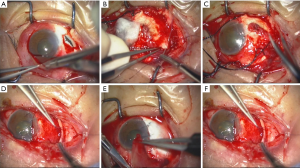
- Anterior chamber puncture to reduce intraocular pressure and avoid pigment membrane rupture or rupture expand when separate the tissue, if necessary;
- separate conjunctiva and pigment membrane tissue/sclera gently, both sharp and blunt, to avoid damaging conjunctiva and pigment membrane tissue. No damage to muscle and conjunctival blood supply, to prevent ischemia;
- completely eliminate necrotic inflammatory tissues;
- perform partial medial rectus translocation/autologous pedicle tenon graft to patients with pigmented membrane perforation or extensive range Ischemic, according to the patient’s needs;
- perform edge corneal lamellar resection, if necessary;
- trim sclera flap, edge is at least 3 mm larger than defects. Scleral graft was secured into place with interrupted 5-0 nylon thread in sclera and 10-0 Alcon in limbus;
- perform autologous conjunctival pedicle flap. The conjunctiva was then re-approximated at the limbus to entirely cover the graft, sclera and muscles.
Results
Eight patients underwent scleral patch grafts between December 2010 and June 2015 (Table 1). Patient ages ranged between 3 to 87 years (mean, 51.2 years). There were 4 male patients and 3 right eyes. Follow-up ranged from 6 to 48 months (median, 18.5 months). Four patients had sclera staphyloma second to surgery (Patient 1: corneal dermoid resection; Patient 4: pterygium surgery; Patients 6 and 7: cataract surgery). Two patients had necrotizing scleritis (Patient 2: rheumatic arthritis 10 years; Patient 3). Two patients had scleral melting and pigment membrane small perforation after thermal burns (Patient 5: preformed sclera patch graft + partial medial rectus translocation + autologous conjunctival pedicle flap; Patient 8: preformed sclera patch graft + autologous pedicle tenon graft + autologous conjunctiva cover).
Table 1
| Case No. | Sex/age (y) | Eye | Follow-up (M) | Previous history/duration | Vision preop | Vision postop | IOP preop (mmHg) | IOP postop (mmHg) | Diagnosis | Surgery | Complications | Others |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | F/3 | OS | 30 | Resection of corneal dermoid/2 years | 0.20 | 0.50 | 15.0 | 17.0 | Scleral staphyloma | SPG | NO | |
| 2 | F/87 | OD | 48 | Rheumatic arthritis/10 years | 0.15 | 0.15 | 8.0 | 15.0 | Necrotizing scleritis | SPG + ACPF | NO | |
| 3 | M/58 | OS | 26 | No history of systemic or other ocular disease | 0.30 | 0.25 | 15.0 | 28.0 | Necrotizing scleritis | SPG + ACPF | High IOP | |
| 5 | M/38 | OS | 13 | Thermal burns/1 month and AMT/2 weeks | 0.40 | 0.50 | 5.0 | 16.0 | (I) Sclera, pigment layer dissolution perforation; (II) choroidal detachment; (III) Thermal burns | SPG + partial medial rectus translocation + ACPF | NO | |
| 6 | M/63 | OS | 12 | Cataract surgery/20 years | HM | HM | 15.0 | 16.0 | (I) Scleral staphyloma; (II) iridodialysis; (III) aphakia; (IV) obsolete explosive injury | SPG | NO | |
| 7 | F/74 | OD | 7 | Cataract surgery/12 years; eyeball atrophy OS (standing RD)/20 years | HM | HM | 12.0 | 14.0 | (I) scleral staphyloma; (II) aphakia | SPG + ACPF | Partial scleral patch melt | |
| 8 | M27 | OS | 6 | Thermal burns/3 days | HM | 0.10 | 7.0 | 18.0 | (I) Sclera, pigment layer dissolution perforation; (II) Thermal burns | SPG + autologous pedicle Tenon graft + ACPF | NO |
OS, left eye; OD, right eye; HM, hand move; IOP, intraocular pressure; SPG, sclera patch graft; ACPF, autologous conjunctival pedicle flap.
Structural integrity
Satisfactory anatomic outcomes were achieved in the follow up of all the eight cases.
Visual acuity
No changes in vision could be attributed directly to scleral patch graft surgery in this series. Six patients had maintained almost the same vision as that of preoperation. The BCVA of Patient 1 improved from 4/20 to 10/20, because of reduction of astigmatism (from −3.5D×135° to −0.75D×120°). The BCVA of Patient 8 improved from hand movies to 2/20, because of reduction of corneal edema and vitreous hemorrhage.
Condition of grafts and complication
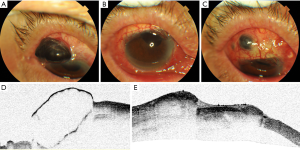
Graft acceptance was achieved in all cases. However, in Patient 7, secondary atrophy of the graft was noted 1 month post operatively, with 2×2 mm partial uveal exposure (Figure 2: Patient 7). Topical corticosteroids discontinued 1 month later for the course that the corticosteroids may cause sclera melt aggravate, and Topical immunosuppressants (Tacrolimus), artificial tears continued. We suggest the contralateral eye autologous sclera patch graft if necessary but it was not accepted by the patient’s family. No further treatment was accepted except local eye drops. Fortunately, Sclera atrophy of the graft presented no further aggravating, and the patients had no foreign body sensation, pain, or other symptoms. Regrafting was not performed and the patient is still in follow-up now. In Patient 6, high intraocular pressure (28 mmHg) was noted three weeks post operatively, which can be controlled by a kind of medicine (0.5% Timolol or Brinzolamide eye drops). None of the patients experienced graft infection, necrosis, or recurrence of inflammation.
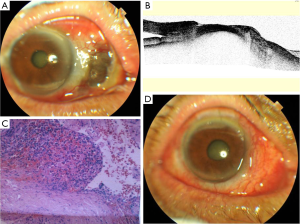
Histopathologic analysis and microbiological culture
Histopathologic evaluation of the resected tissue was got only in two patients (Patients 2 and 3) and it showed chronic suppurative inflammation. Microbiological culture showed negative except one positive (Figure 3, Patient 2: pseudomonas aeruginosa bacteria. Drug sensitivity test showed resistant to cefazolin and sensitive to levofloxacin, tobramycin and other drugs; systemic application of sensitive antibiotics for 1 week and local application of sensitive antibiotics for 4 weeks).
Ocular symptoms
No eye pain, foreign body sensation, photophobia, and other discomforts showed in all the patients, under the condition that topical immunosuppressants and artificial tear.
Discussion
Scleral defects are usually caused by necrotic scleritis, trauma or surgery. Scleritis is a serious inflammatory disease with an etiology of idiopathic, autoimmune, or infectious, that can cause severe ocular pain and occasionally impaired vision. It is usually associated with systemic diseases such as systemic lupus erythematosus, rheumatoid arthritis, and granulomatosis with polyangiitis. Responsive sclera inflammation occasionally arouse in the process of infectious diseases such as syphilis, tuberculosis, and zoster. Scleritis associated with bacterial or fungal invasion is uncommon, and account for less than 2% of all eye infections (9). Gram-negative bacteria, Gram-positive cocci and filamentous fungi, which are also responsible for microbial keratitis, are often reported causes. Approximately 85% of bacterial scleritis was invited by Pseudomonas aeruginosa. In our review, two cases (25%) were association with scleritis and one of them (with a history of rheumatoid arthritis, Rheumatoid factor 52.8 IU/mL) was found to be associated with Pseudomonas aeruginosa bacteria (50%), which were proved by histopathologic evaluation and culture. Fortunately, the graft were stable and did not developed graft necrosis after a surgery of sclera patch graft and autologous conjunctival pedicle flap in the 36 months follow-up. Nevertheless, it is necessary to screen and rule out systemic vasculitic disorders preoperatively. Non-emergency surgery should be deferred until medical therapy, such as systemic glucocorticoids or immunosuppressive agents, has controlled the primary disease, if detected. In addition, two patients (25%) were caused by thermal burns. Furthermore, four cases (50%) were association with surgery (corneal dermoid resection, pterygium surgery, and cataract surgery). Attention should be paid to iatrogenic injury in ocular medical activities.
A patch graft helps to restore structural integrity of the globe. A various of material and techniques are commonly employed today to manage ocular diseases that devastate to structural stability of the eye. Still no material has been proved to be generally accepted. Allogeneic sclera was commonly used as a graft in cases of impending scleral rupture, ectasias, or dehiscence. Varied success has been reported with the use of scleral patch grafts. Scleral patch graft remains a universal option (10) due to its relatively availability, strength, flexibility and easy to preserve. The only criticism is that it may involve in the ongoing process of necrosis and melting because of avascular.
The scleral stroma is normally avascular and receives its nutritional supply from choroidal blood vessels and from the vascular plexus in the tenon capsule and on the episcleral surface (11). Acute severe chemical, thermal burns and ocular surgeries are the common causes of scleral ischemia, thinning, and melt. Scleral melt can occur in the event of medical activities such as bare sclera pterygium excision, especially with adjuvant therapies such asβ-irradiation and jeopardized mitomycin C (8,12). In addition, scleral melt has been described after retinal detachment repair, glaucoma surgery, and cataract surgery. How to deal with scleral dissolution is still challenging. Victoria E. Casas reported four successfully cases of use tenon pedicle graft to cover the ischemic zone for scleral ischemia and melt (7). Yazici B reported two successfully cases of use of Conjunctiva-Muller muscle pedicle flap in surgical treatment of necrotizing scleritis (13). So, a cover of conjunctival flap on the scleral graft is usually necessary to prevent its necrosis and melting, for scleral graft does not contain epithelium and its survival is arduous on avascular surfaces. While vascularization might speed resolution of infection, we generally recommended autologous conjunctival pedicle flap and other grafting procedures until scleral inflammation is medically controlled. In our review, case 5 had a huge scleral melting/perforation secondary to thermal burns on the nasal. Conjunctiva-Muller muscle pedicle flap or tenon pedicle flap seems not possible. We used partial medial rectus translocation to cove the small perforation of uvea, which may increase blood supply of sclera patch graft simultaneously. In the period of 13 months follow-up, this new surgical method seems a feasible alternative for treating scleral melting/perforation. Another patient (case 8) had relatively small scleral melting/perforation on the superior secondary to thermal burns. We performed autologous pedicle tenon graft at the same time. All sclera grafts were coved with conjunctiva (6 in 8 patients performed autologous conjunctival pedicle flap), and no conjunctival melt was presented. If the adjacent conjunctiva cannot be mobilized, a free conjunctival flap or amniotic membrane transplantation may be considered.
In our review of scleral patch grafting for cases of scleral defects of varying etiologies, favourable structural outcome was achieved in 7/8 eyes. One patient had complications in the form of graft melting one month postoperative. Fortunately, sclera atrophy of the graft presented no further aggravating, and the patients had no foreign body sensation, pain, or other symptoms. We suggest the contralateral eye autologous sclera patch graft if necessary but it was not accepted by the patient’s family. Regrafting was not performed and the patient is still in follow-up now. Another patient had high intraocular pressure (28 mmHg), which was noted 3 weeks postoperative, and it can be controlled by a kind of medicine (0.5% Timolol or Brinzolamide eye drops).
Our study has some limitations, including its retrospective design, the small number of patients, and the lack of a control group. Despite this, we can conclude that sclera patch grafts is a safe and effective methods for management scleral defects. However, sclera rejection and melt should be considered postoperatively.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2017.04.03). The authors have no conflicts of interest to declare.
Ethnical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Institutional review board approval was obtained, and the study was conducted in accordance of the guide-lines of the Declaration of Helsinki (as revised in 2013) [Ethic Approval ID: EC 20160616(4)-A11] and the Health Insurance Portability and Accountability Act of 1996. Written informed consent was obtained from the patient for publication of cases report and accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Barman M, Finger PT, Milman T. Scleral patch grafts in the management of uveal and ocular surface tumors. Ophthalmology 2012;119:2631-6. [Crossref] [PubMed]
- Koenig SB, Sanitato JJ, Kaufman HE. Long-term follow-up study of scleroplasty using autogenous periosteum. Cornea 1990;9:139-43. [Crossref] [PubMed]
- Sangwan VS, Jain V, Gupta P. Structural and functional outcome of scleral patch graft. Eye (Lond) 2007;21:930-5. [Crossref] [PubMed]
- Ozcan AA, Bilgic E, Yagmur M, et al. Surgical management of scleral defects. Cornea 2005;24:308-11. [Crossref] [PubMed]
- Yalçindag FN, Celik S, Ozdemir O. Repair of anterior staphyloma with dehydrated dura mater patch graft. Ophthalmic Surg Lasers Imaging 2008;39:346-7. [Crossref] [PubMed]
- Stunf S, Lumi X, Drnovšek-Olup B. Preserved scleral patch graft for unexpected extreme scleral thinning found at the scleral buckling procedure: a case report. Indian J Ophthalmol 2011;59:235-8. [Crossref] [PubMed]
- Casas VE, Kheirkhah A, Blanco G, et al. Surgical approach for scleral ischemia and melt. Cornea 2008;27:196-201. [Crossref] [PubMed]
- Lee JS, Shin MK, Park JH, et al. Autologous advanced tenon grafting combined with conjunctival flap in scleromalacia after pterygium excision. J Ophthalmol 2015;2015:547276
- Cunningham MA, Alexander JK, Matoba AY, et al. Management and outcome of microbial anterior scleritis. Cornea 2011;30:1020-3. [Crossref] [PubMed]
- Dubey S, Rajurkar K, Kapur N, et al. Heterologous, Human Donor Sclera as the Patch Graft in Glaucoma Drainage Device Surgery. J Glaucoma 2017;26:e128-9. [Crossref] [PubMed]
- Watson PG, Young RD. Scleral structure, organisation and disease. A review. Exp Eye Res 2004;78:609-23. [Crossref] [PubMed]
- Yamazoe K, Shimazaki-Den S, Otaka I, et al. Surgically induced necrotizing scleritis after primary pterygium surgery with conjunctival autograft. Clin Ophthalmol 2011;5:1609-11. [Crossref] [PubMed]
- Yazici B. Use of conjunctiva-Müller muscle pedicle flap in surgical treatment of necrotizing scleritis. Ophthal Plast Reconstr Surg 2008;24:19-23. [Crossref] [PubMed]
Cite this article as: Yao Y, Jhanji V. Short-term observation of management of sclera patch grafts used in the scleral defects. Ann Eye Sci 2017;2:30.


