
Fibrovascular proliferation of retinopathy of prematurity in zone II after initial ranibizumab treatment
Introduction
Retinopathy of prematurity (ROP) remains an important cause of preventable blindness in children (1,2). Abnormal growth of retinal blood vessels is the mainly pathogenic pathway of ROP which is mainly modulated by vascular growth factors (3,4). Therefore, blocking the action of vascular endothelial growth factor (VEGF) might be expected to reduce the vascular activity. Recently, sufficient publications have described anti-VEGF drugs’ application in premature for their efficiency in reducing pathologic angiogenesis (5). It is not surprised that anti-VEGF drugs have experienced a promise application prospect (6,7).
However, it is not uncommon to see the diseases developed against the expectation in some cases, which are witnessed by vitreoretinal traction, retinal detachment in ROP after IV treatment (8-10). Still there are patients who showed no regress of fibrovascular proliferation and even fibrosis aggravating ultimately. The potential influence factors why those patients did not respond to the drugs and fibrosis proliferation progress are worth to explore.
Our study concerned about the progress fibrovascular proliferation after initial intravitreal injections of bevacizumab and the susceptible risk factor in zone II ROP.
Methods
The medical records of infants who met criteria for ROP screening and developed zone II stage 3 were reviewed retrospectively at our department from January 2013 to July 2014, including all preterm infants who accepted intravitreal injection of ranibizumab as an initial treatment after informed consent were obtained. Eyes with zone II stage 3 ROP and plus disease were included; those who accepted laser treatment, cryotherapy, anti-vascular endothelial growth factor (anti-VEGF) drugs before and did not accomplish the follow-up were excluded. The details of retinal changes for ROP in all examinations were performed by senior ophthalmologists. A weekly or biweekly basis examination was performed initially, depending on the retinal findings and continued until vascularization had reached zone III. If the fibrovascular proliferation do not regressed or even progressed, further treatment will be required. Data collected from the infants’ record: gender, gestational age, birth weight and oxygen exposure. ROP findings included the preoperative ROP zone and intraocular hemorrhage, iris neovascularization. ROP findings were recorded by detailed retinal drawings and RetCam.
Statistical analysis
The eyes were divided into two groups based on regress of fibrovascular tissue or not after initial ranibizumab treatments within 2 weeks. Statistical analyses were performed to compare the two groups using statistical software (StatLab, SPSS for Windows, version 16.0; SPSS, Inc., Chicago, Illinois, USA). Univariate analyses to determine the association risk factors were performed using the Mann-Whitney U test, the t test, and the Fisher exact test.
Results
A total of 108 eyes of 57 patients (18 females, 39 males) underwent initial intravitreal injection of ranibizumab. The mean birth weight was 1,440.0±357.7 g with a mean gestational age of 30.5±2.0 weeks and a mean follow-up of 7.0±3.8 months. Eight of 57 (14.0%) patients’ diseases did not regress after initial intravitreal injection within 2 weeks observation period. Eleven of the 14 eyes (78.6%) underwent laser treatment, mean at 2.3±2.3 weeks after initial ranibizumab treatment. Three eyes (21.4%) progressed to surgery.
The number of patients with intraocular hemorrhage in the unfavorable group was more than the other group, and the difference was significant (P<0.030). The hemorrhage mainly occurred around the rages within one quadrant avoiding macular zone (Figure 1). The macular zone was covered by hemorrhage in four eyes.
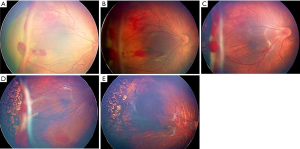
For the diseases mainly by fibrovascular membranes proliferation or aggravated after intravitreal ranibizumab (IVR), photocoagulation was the priority recommendation. The number of patients applied photocoagulation in the unfavorable group was more than the no regress group (P<0.001). There were no differences between the two groups in the other baseline characteristics. The data and statistics are summarized in Table 1.
Table 1
| Clinical data | Unfavorable group | Regress group | P value |
|---|---|---|---|
| No. eyes/patients | 14/8 | 94/49 | – |
| Birth weight (g) | 1,293.8±209.5 | 1,469.3±372.5 | 0.065# |
| Gestation age (weeks) | 34.5±2.0 | 30.3±1.39 | 0.155# |
| Oxygen administration | 4 | 22 | 0.542† |
| Preretinal hemorrhage | 9 | 32 | 0.030* |
| Iris neovascularization | 0 | 5 | 1.000† |
| Laser treatment | 11 | 30 | 0.001* |
*, Mann-Whitney U test; #, independent samples
Discussion
In our study, 14 out of 108 eyes (13.0%) treated with ranibizumab had unfavorable fibrovascular proliferation after response to the initial treatment within 2 weeks. The rages of those cases presented a remarkable fibrovascular proliferation or even aggravate fibrosis proliferation with pale membrane appearance at the posterior pole. For the unfavorable eye mainly by fibrovascular membranes proliferation, photocoagulation was the priority recommendation. Three infants progressing rapidly turned to vitrectomy.
Anti-VEGF drug is a good way to treat ROP which is characterized by abnormal retinal vascular (11). However, in some cases, as a response to the decreased levels of VEGF by the drugs, the rapid neovascular involution causes the progression of accelerated fibrosis and posterior hyaloid (12,13). The contraction of the fibrovascular membrane in the course of ROP treatments phenomenon is frequently observed clinically. This kind of performance often indicates a poor prognosis. One previous study reported a case of advanced ROP which showed an acute contraction of the proliferative membrane after an intravitreal injection of bevacizumab (14). The vascular component of the fibrovascular membrane regressed, and the appearance became fibrotic, which caused a deterioration of tractional retinal detachment. Previous study had showed differential role of VEGF isoforms in retinal physiological angiogenesis in a rabbit model. Ranibizumab is an aptamer which specifically blocks VEGF [165], affecting the blood vessel growth, but VEGF [189] is rather involved in fibrosis (15). This explained why ranibizumab treatment may result in the progression of fibrosis in ROP.
There are statistical differences between the two groups in hemorrhage in our study. Significant preretinal hemorrhages are likely to contribute to abnormal vitreous organization, resulting in an unfavorable outcome (6). In Nazari et al. series, vitreous hemorrhages often lead to irreparable retinal detachment despite surgical intervention (16). A prospective interventional clinical case series by Krishna A. Rao suggested that vitreous hemorrhage was the only independent risk factor for severe ROP (17). Ehrenberg et al. found that after the injection of blood into the vitreous, almost all eyes developed glial membranes on the peripheral retina and fibrous membranes caused local retinal contraction. Untreated premacular hemorrhages usually cause surface wrinkling or traction macular detachment due to the progressive fibrovascular proliferation and contraction along the posterior surface of the vitreous (18). It is reasonable to consider that hemorrhages, a severe destruction of vessels form, may cause a high recurrence of ROP and detachment by proliferation and contraction.
Oxygen is considered a critical factor in the pathogenesis of ROP (19,20). Inadequate oxygen expose resulted in the extent of ROP (21). But in our study there was no evidence that the factors had impact on the abnormal response of ROP. Another possible cause is that regular doses of bevacizumab which has a short terminal half-life may not be effective in inducing regression of sever ROP, so the fibrovascular organization continued to aggravate (8,22). Further studies are required to verify it.
Although the initial responses to part of the patients were poor, the patients’ overall prognoses are favorable after laser treatment in our study, but the structural damage by laser may require long-term extended observation. In conclusion, the incidence of fibrovascular proliferation not regressed was not high, and the presence of hemorrhages was a possible influence factor of progression to the unfavorable ROP after ranibizumab treatment. A close screening examination and follow-up and timely intervention are needed for the management of ROP.
Acknowledgments
Funding: This work was supported by the National Natural Science Foundation of China (grant No. 81470649).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2017.03.04). The authors have no conflicts of interest to declare. Funding institutions had no role in the study design, data collection and analysis, the decision to publish or preparation of the manuscript.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Steinkuller PG, Du L, Gilbert C, et al. Childhood blindness. J AAPOS 1999;3:26-32. [Crossref] [PubMed]
- Wheatley CM, Dickinson JL, Mackey DA, et al. Retinopathy of prematurity: recent advances in our understanding. Arch Dis Child Fetal Neonatal Ed 2002;87:F78-82. [Crossref] [PubMed]
- Romagnoli C. Risk factors and growth factors in ROP. Early Hum Dev 2009;85:S79-82. [Crossref] [PubMed]
- Mutlu FM, Sarici SU. Treatment of retinopathy of prematurity: a review of conventional and promising new therapeutic options. Int J Ophthalmol 2013;6:228-36. [PubMed]
- Bakri SJ, Snyder MR, Reid JM, et al. Pharmacokinetics of intravitreal bevacizumab (Avastin). Ophthalmology 2007;114:855-9. [Crossref] [PubMed]
- Coats DK, Miller AM, Hussein MA, et al. Involution of retinopathy of prematurity after laser treatment: factors associated with development of retinal detachment. Am J Ophthalmol 2005;140:214-22. [Crossref] [PubMed]
- Wu WC, Kuo HK, Yeh PT, et al. An updated study of the use of bevacizumab in the treatment of patients with prethreshold retinopathy of prematurity in taiwan. Am J Ophthalmol 2013;155:150-158.e1. [Crossref] [PubMed]
- Yokoi T, Yokoi T, Kobayashi Y, et al. Risk factors for recurrent fibrovascular proliferation in aggressive posterior retinopathy of prematurity after early vitreous surgery. Am J Ophthalmol 2010;150:10-15.e1. [Crossref] [PubMed]
- Hapsari D, Sitorus RS. Intravitreal Bevacizumab in Retinopathy of Prematurity: Inject or Not? Asia Pac J Ophthalmol (Phila) 2014;3:368-78. [Crossref] [PubMed]
- Chen SN, Lian I, Hwang YC, et al. Intravitreal anti-vascular endothelial growth factor treatment for retinopathy of prematurity: comparison between Ranibizumab and Bevacizumab. Retina 2015;35:667-74. [Crossref] [PubMed]
- Mintz-Hittner HA, Kennedy KA, Chuang AZ, et al. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N Engl J Med 2011;364:603-15. [Crossref] [PubMed]
- Moradian S, Ahmadieh H, Malihi M, et al. Intravitreal bevacizumab in active progressive proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 2008;246:1699-705. [Crossref] [PubMed]
- Van Geest RJ, Lesnik-Oberstein SY, Tan HS, et al. A shift in the balance of vascular endothelial growth factor and connective tissue growth factor by bevacizumab causes the angiofibrotic switch in proliferative diabetic retinopathy. Br J Ophthalmol 2012;96:587-90. [Crossref] [PubMed]
- Honda S, Hirabayashi H, Tsukahara Y, et al. Acute contraction of the proliferative membrane after an intravitreal injection of bevacizumab for advanced retinopathy of prematurity. Graefes Arch Clin Exp Ophthalmol 2008;246:1061-3. [Crossref] [PubMed]
- Van Bergen T, Vandewalle E, Van de Veire S, et al. The role of different VEGF isoforms in scar formation after glaucoma filtration surgery. Exp Eye Res 2011;93:689-99. [Crossref] [PubMed]
- Nazari H, Modarres M, Parvaresh MM, et al. Intravitreal bevacizumab in combination with laser therapy for the treatment of severe retinopathy of prematurity (ROP) associated with vitreous or retinal hemorrhage. Graefes Arch Clin Exp Ophthalmol 2010;248:1713-8. [Crossref] [PubMed]
- Rao KA, Purkayastha J, Hazarika M, et al. Analysis of prenatal and postnatal risk factors of retinopathy of prematurity in a tertiary care hospital in South India. Indian J Ophthalmol 2013;61:640-4. [Crossref] [PubMed]
- Ehrenberg M, Thresher RJ, Machemer R. Vitreous hemorrhage nontoxic to retina as a stimulator of glial and fibrous proliferation. Am J Ophthalmol 1984;97:611-26. [Crossref] [PubMed]
- Giannantonio C, Papacci P, Cota F, et al. Analysis of risk factors for progression to treatment-requiring ROP in a single neonatal intensive care unit: is the exposure time relevant? J Matern Fetal Neonatal Med 2012;25:471-7. [Crossref] [PubMed]
- Hellström A, Smith LE, Dammann O. Retinopathy of prematurity. Lancet 2013;382:1445-57. [Crossref] [PubMed]
- Flynn JT, Bancalari E, Snyder ES, et al. A cohort study of transcutaneous oxygen tension and the incidence and severity of retinopathy of prematurity. N Engl J Med 1992;326:1050-4. [Crossref] [PubMed]
- Avery RL. Bevacizumab (Avastin) for retinopathy of prematurity: wrong dose, wrong drug, or both? J AAPOS 2012;16:2-4. [Crossref] [PubMed]
Cite this article as: Hu Q, Bai Y, Chen X, Xia H, Huang L, Chen Y, Li X. Fibrovascular proliferation of retinopathy of prematurity in zone II after initial ranibizumab treatment. Ann Eye Sci 2017;2:48.


