
Optical coherence tomography findings in a case of cilioretinal artery occlusion reversal, treated with mannitol and carbogen administration
Introduction
Cilioretinal artery occlusion (CLRAO) involving the macula causes sudden catastrophic loss of vision due to neuronal destruction of the inner layers of the retina. CLRAO has no predefined algorithm of treatment. Systemic screening and observation describe ophthalmologist’s modus operandi. It is a distinct clinical entity, as cilioretinal arterioles arise from the posterior ciliary artery, contrary to the central retinal artery that supplies the inner retina. Etiologically it is classified in three distinct entities: (I) nonarteritic CLRAO alone, (II) nonarteritic CLRAO associated with central vein occlusion (CRVO) and (III) arteritic CLRAO associated with giant cell arteritis (GCA). Of the three types, those associated with GCA and those with central vein occlusion require detailed discussion about their pathogeneses, which are controversial (1).
Approximately 15–30% of the general population has a cilioretinal artery which supplies the whole or a part of the fovea, CLRAO has an incidence of approximately 1 to 10 in 100,000 (1). Symptomatic CLRAO is less common than retinal artery occlusions (RAO), comprising about 5.3–7.1% of all RAOs (1).
Our search in peer review articles showed that there had been no reports presenting isolated CLRAO treated effectively with combined intra venous mannitol (2) and carbogen inhalation (3) which simultaneously recorded edema recession with consecutively macular thickness and reflectivity optical coherence tomography (OCT) scans and contrasts with previously reported cases. The case report was conducted in accordance with the ethical standards stated in the Declaration of Helsinki and was approved by an institutional review board; written consent was obtained from the participant.
Case presentation
A 67-year-old man came to our vitreoretinal clinic with a sudden painless loss of vision, in the left eye with onset 6 hours prior to presentation. Ocular history was not contributory. Patient’s blood laboratory findings, systemic physical examination, electrocardiogram, and chest x-ray were all unremarkable. Carotid artery triplex ultrasound revealed bilateral internal carotid artery stenosis which was more severe in the left side, 30–35%.
On ophthalmic examination, best-corrected visual acuity was 8/10 in the right eye (OD) and counting fingers (CF) in the left eye (OS). Intraocular pressure was 12 mmHg in OD and 22 mmHg in OS. Anterior segment examination with slit-lamp was normal in both eyes. Funduscopy of the right eye was normal; left eye showed well demarcated papillomacular bundle edema centered on cilioretinal artery adjacent to the fovea and cherry red spot (Figure 1A). Fluorescein angiography (FFA) of the left eye showed masking of choroidal fluorescence secondary to retinal edema and delayed filling of cilioretinal artery with late hyperfluorescence (Figure 1B). FFA which was performed on the second week after treatment demonstrated that the cilioretinal artery was recanalized (Figure 1C).
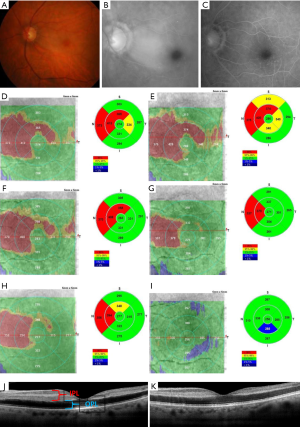
OCT thickness was automatically calculated by mapping software on admission, confirming nasal paramacular and perimacular edema (412 & 373 µm, Figure 1D). Humphrey visual field testing showed a significant central visual field defect. We diagnosed the disease as CLRAO.
Therapy was started 7 hours after the onset of symptoms. Carbogen, a mixture of 4–7% carbon dioxide and 93–96% oxygen was conducted for 10 minutes hourly during waking hours, and 10 min every 4 hours at night and continued for 72 hours. Intravenous mannitol was administered in the dosage of 2 gr per kilogram of body weight twice per day and continued for 72 hours. Dorzolamide and brimonidine tartrate was topically applied three times daily (t.i.d.).
OCT macular scans were acquired from both eyes every 12 hours demonstrating gradual thickness reduction (Figure 1E,F,G,H). The last performed scan showed that paramacular, and perimacular thickness was restored to normal, 336 and 310 µm (Figure 1I). Increased reflectivity of the innermost layers of the retina, was identified and a corresponding reduction of reflectivity in the outer retina and the retinal pigment epithelium and choriocapillaris layers (Figure 1J,K).
Discussion
CLRAO has been reported in cases related to antiphospholipid syndrome, cisplatin, embolism, migraine, sildenafil, SLE, pregnancy, systemic hypertension, and hyperhomocysteinemia (4-7). In our case, there was a 30–35% stenosis of the left internal carotid artery, an associated risk factor for CLRAO. As oxygen consumption of the retina is 13 mL/100 g/minute (8) the highest rate of any organ in the body, it is susceptible to ischemia. CLRAO results in inner layer edema and pyknosis of the ganglion cell nuclei, ischemic necrosis, and retina opacification. In order to prevent irreversible damage to the retina, therapy must be provided as soon as possible after the onset of vision loss irreversible retinal damage occurs 240 minutes after the occlusive event (9).
The ocular perfusion pressure is equal to the difference between the central retinal artery and the central retinal vein. Equilibrium between the pressure inside retinal veins and the intraocular pressure (IOP) is required; reduction of the later may increase the retinal artery perfusion or help dislodge the embolus. Many strategies have been employed by surgeons for this purpose, such as bulbus massage, anterior chamber paracentesis, and pharmacological administration of agents reducing the IOP, as mannitol.
Carbogen administration is based on the hypothesis that carbon dioxide will prevent oxygen-induced vasoconstriction and hence improve or maintain the blood flow while maintaining the oxygenation of the retina.
Conclusions
Retinal vascular occlusions are serious events that require investigation for a life-threatening underlying systemic disease. The spectrum of clinical variability was related to the extent of the ischemic area, the location of the occlusion, and the degree of macular involvement. A thorough assessment is required in order to identify the etiology. CLRAO should be considered as an ocular emergency analog of cerebral stroke or infarction. Atherosclerotic risk factors must be evaluated to prevent further medical comorbidities. Current systemic management in CLRAO is considered to have limited efficacy in improving vision and announcements suggest that for treatment to be effective, it must be initiated within 240 minutes symptoms onset. Oxygen deprivation of the inner retinal layers leads to cell and nerve fibers edema and lowering of light transmission. Spectral domain OCT is based on light interference of a reference arm and the sample arm. In the sample arm, different structures will have different indices of refraction and light will be backscattered when it encounters materials of different refractive index. The edematous high index layers cause optical shadowing of the outer layers due to high reflectivity.
SD-OCT images in the acute stage shows: (I) increased macular thickness which gradually improved during treatment; (II) inner retinal layers; the nerve fiber, ganglion cell, inner plexiform, inner nuclear and outer plexiform layers, present increased reflectivity corresponding to higher thickness; (III) the outer retinal layers; outer nuclear and retinal pigment epithelium, shows reduced reflectivity. At a late phase OCT findings of reflectivity was reversed.
To our knowledge this is the only case of CLRAO that reports effective treatment with combined administration of mannitol and carbogen inhalation. Clinical observation is accompanied with FFA documentation of recanalization and complete follow-up data of retinal thickness and reflectivity. A retrospective review (10) describes similar SD-OCT findings of two cases with central RAO.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2019.02.01). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hayreh SS. Acute retinal arterial occlusive disorders. Prog Retin Eye Res 2011;30:359-94. [Crossref] [PubMed]
- Varma DD, Cugati S, Lee AW, et al. A review of central retinal artery occlusion: clinical presentation and management. Eye (Lond) 2013;27:688-97. [Crossref] [PubMed]
- Arend O, Harris A, Martin BJ, et al. Retinal blood velocities during carbogen breathing using scanning laser ophthalmoscopy. Acta Ophthalmol (Copenh) 1994;72:332-6. [Crossref] [PubMed]
- Park SU, Lee SJ, Kim M. Isolated cilioretinal artery occlusion in a patient with hypertensive crisis. BMJ Case Rep 2014;2014.
- Alkan A, Talaz S. Cilioretinal artery occlusion associated with cisplatin. J Oncol Pharm Pract 2018;1078155218759805 [Epub ahead of print]. [PubMed]
- Berkani Z, Kitouni Y, Belhadj A, et al. Cilioretinal artery occlusion and central retinal vein occlusion complicating hyperhomocysteinemia: a case report. J Fr Ophtalmol 2013;36:e119-27. [Crossref] [PubMed]
- Basu A, Eyong E. Cilioretinal arterial occlusion phenomenon: a rare cause of loss of vision in pregnancy. Eur J Obstet Gynecol Reprod Biol 2008;137:251-2. [Crossref] [PubMed]
- Werkmeister RM, Schmidl D, Aschinger G, et al. Retinal oxygen extraction in humans. Sci Rep 2015;5:15763. [Crossref] [PubMed]
- Hayreh SS, Zimmerman MB, Kimura A, et al. Central retinal artery occlusion. Retinal survival time. Exp Eye Res 2004;78:723-36. [Crossref] [PubMed]
- Falkenberry SM, Ip MS, Blodi BA, et al. Optical coherence tomography findings in central retinal artery occlusion. Ophthalmic Surg Lasers Imaging 2006;37:502-5. [PubMed]
Cite this article as: Christodoulou P, Katsimpris I. Optical coherence tomography findings in a case of cilioretinal artery occlusion reversal, treated with mannitol and carbogen administration. Ann Eye Sci 2019;4:13.


