
Letter to the Editor—subtype-specific orbital lymphomas
Through the last decades, our knowledge about ocular adnexal lymphoma (OAL) including orbital lymphoma has increased markedly due to the focus on lymphoma subclassification and the focus on improved staging of OAL. Our retrospective study of 797 patients with orbital lymphoma (1), discussed in the Editorial by Stagner and Zukerberg (2), provides additional insight into this relatively rare disease. Though orbital lymphoma is uncommon, lymphoma is the most common malignant neoplasm to arise within the orbit; hence it is of great importance to investigate the clinical and histopathological features of lymphomas of this specific site. As pointed out by Stagner and Zukerberg (2), a cohort with the size of nearly 800 orbital lymphoma patients has never been analyzed. The material is unique with seven contributing eye cancer centers from different parts of the world.
The Ann Arbor staging system has long been used to stage both Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL) (3). When it comes to the staging of OAL, the Ann Arbor staging system has some shortcomings as it lacks information about the anatomical location of tumor, resulting in a disproportionately high percentage of patients staged as a stage IE lymphoma (E standing for extranodal). Consequently, The American Joint Committee on Cancer (AJCC) has developed a staging system for OAL based on the TNM staging system, taking the precise tumor extent into account (4). In the AJCC TNM staging system, stage T1 defines a tumor limited to the conjunctiva, stage T2 defines a tumor involving the orbit ± the conjunctiva, stage T3 defines a tumor involving the eyelid ± conjunctiva and/or orbit, and stage T4 defines a tumor extending beyond the orbit to adjacent structures. Ophthalmologists did not earlier distinguish between OAL subtypes. Through the last decades, our understanding of OAL has increased markedly, and today we know that the majority of OAL are of B cell origin, with mainly 4 subtypes being encountered: the 2 low-grade lymphoma subtypes namely extranodal marginal zone B cell lymphoma (EMZL) and follicular lymphoma (FL), and the 2 high-grade lymphoma subtypes namely diffuse large B cell lymphoma (DLBCL) and mantle cell lymphoma (MCL). This distribution of lymphoma subtypes is confirmed in our study in which EMZL is found to be the most common lymphoma subtype (57%), followed by DLBCL (15%), FL (11%), and MCL (8%) (Figure 1). Hence, the majority of orbital lymphomas are indolent slow-growing tumors. As expected, the low-grade lymphomas EMZL and FL were in our study found to have a favorable 10-year disease-specific survival (DSS) of 92% and 71%, respectively, whereas the high-grade lymphomas DLBCL and MCL were found to have a poorer prognosis with a 10-year DSS of 41% and 32%, respectively.
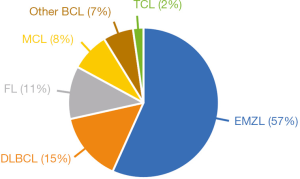
Interestingly, the frequency of orbital lymphoma differs from lymphomas arising in other tissues of the body. In a large study on 2,480 space-occupying orbital lesions, lymphoma was found to comprise 37% of all orbital neoplasms, and EMZL is by far the most common subtype (comprising 57% in our cohort) (5,6). However, EMZL comprises only up to 8% of all B-cell lymphomas, arising in a wide range of extranodal tissues (7). The stomach is the most frequently involved site, reported to account for 70% of all EMZL cases, whereas OAL is reported to account for 12% of all EMZL cases (7). The pathogenesis and dissemination of lymphomas are found to be highly site-specific, as lymphoma tumor cells still possess most of normal lymphocytic physiological behavior (8). Lymphocytes continuously recirculate between blood, secondary lymphoid tissue, and efferent lymphatics and lymph nodes, due to tissue-specific homing mechanisms driven by adhesion molecules and chemokines (8). As the malignant counterpart of normal circulating lymphocytes, lymphoma cells are subjects to the same molecular mechanisms of tissue-specific homing, and thereby prone to arise in specific tissues (6,8). EMZL is known to frequently arise in mucosa-associated lymphoid tissue (MALT), found in numerous anatomical sites throughout the body, such as the gastrointestinal tract (7). The conjunctiva and the lacrimal gland are also considered part of the mucosa-associated lymphoid system [so-called eye-associated lymphoid tissue (EALT)], whereas the non-pathological orbital soft tissue is found not to contain lymphoid cells or lymphoid vessels (4). However, as EMZL frequently arises within the orbit, other factors must play a role in the pathogenesis of EMZL. Helicobacter pylori is known to play a role in the development of gastric EMZL (7), and chronic antigenic stimulation by an infectious organism has also been proposed to play a role in the development of OAL.
Studies on newer treatment modalities of orbital lymphoma are sparse, and treatment strategies vary between ocular oncology centers worldwide. It is generally acknowledged that localized, low-grade OAL should be treated with external beam radiation therapy (EBRT), as also pointed out in a newly published review by the American Academy of Ophthalmology (9). This is in line with our results; most of our patients with localized orbital EMZL and FL were treated with EBRT with a good disease-control. Disseminated low-grade lymphomas and high-grade lymphomas should be treated with chemoimmunotherapy (9,10). To improve the standard of treatment of patients with orbital lymphoma, prospective clinical trials on NHL treatment modalities are needed and encouraged. Also continued focus on staging of orbital lymphoma and histological subtyping based on cell origin and genetics is essential and needs likewise further investigation.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2019.05.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Olsen TG, Holm F, Mikkelsen LH, et al. Orbital Lymphoma-An International Multicenter Retrospective Study. Am J Ophthalmol 2019;199:44-57. [Crossref] [PubMed]
- Stagner AM, Zukerberg LR. Additional clinical insights into subtype-specific orbital lymphomas. Ann Eye Sci 2019;4:8. [Crossref]
- Carbone PP, Kaplan HS, Musshoff K, et al. Report of the Committee on Hodgkin's Disease Staging Classification. Cancer Res 1971;31:1860-1. [PubMed]
- Amin MB, Edge S, Greene F, et al. AJCC cancer staging manual. 8th ed. New York: Springer, 2017:1-1024.
- Bonavolontà G, Strianese D, Grassi P, et al. An analysis of 2,480 space-occupying lesions of the orbit from 1976 to 2011. Ophthalmic Plast Reconstr Surg 2013;29:79-86. [Crossref] [PubMed]
- Sjö LD. Ophthalmic lymphoma: epidemiology and pathogenesis. Acta Ophthalmol 2009;87 Thesis 1:1-20.
- Isaacson PG, Du MQ. MALT lymphoma: from morphology to molecules. Nat Rev Cancer 2004;4:644-53. [Crossref] [PubMed]
- Pals ST, de Gorter DJ, Spaargaren M. Lymphoma dissemination: the other face of lymphocyte homing. Blood 2007;110:3102-11. [Crossref] [PubMed]
- Yen MT, Bilyk JR, Wladis EJ, et al. Treatments for Ocular Adnexal Lymphoma: A Report by the American Academy of Ophthalmology. Ophthalmology 2018;125:127-36. [Crossref] [PubMed]
- Mikkelsen LH, Würtz NS, Heegaard S. Recent advances in treating extra-ocular lymphomas. Expert Rev Opthalmol 2018;13:205-17. [Crossref]
Cite this article as: Hindsø TG, Heegaard S. Letter to the Editor—subtype-specific orbital lymphomas. Ann Eye Sci 2019;4:21.


