
Optical coherence tomography in ischemic optic neuropathy
Introduction
Ischemic optic neuropathy is among the most common non-glaucomatous optic neuropathies, especially in patients over age 50, and it is the most common optic nerve problem causing acute vision loss in this age group, with an estimated annual incidence of 2.3–10.2/100,000 Jeny(1-5). Optic nerve ischemia may occur from a number of causes; the most common of which, non-arteritic anterior ischemic optic neuropathy (NAION) is poorly understood in terms of its precise aetiology. Vasculitic processes, such as giant cell arteritis, also may lead to optic nerve ischemia, while acute haemorrhage, intraoperative fluid shifts and haemodilution, and shock all may cause often profound hypoperfusion insults to the optic nerve, either at the optic nerve head or in the retrobulbar pathway (5). In patients with suspected ischemic optic neuropathy, optical coherence tomography (OCT) and OCT angiography (OCT-A) have been used to attempt to characterise the severity of disease as well as to identify underlying vascular anomalies that could contribute to the onset and progression of disease. Although research to date has not allowed us to develop new therapies, doing so remains one of the goals of many of the ongoing efforts that will be described.
NAION
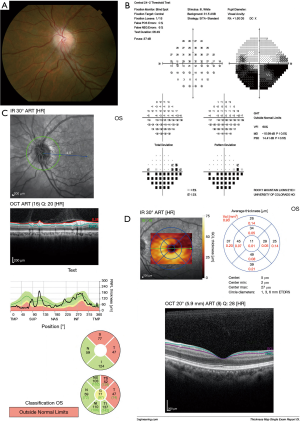
Patients with NAION typically report sudden onset of painless vision loss. Decreased visual acuity (VA) and/or visual field (VF) loss along with a relative afferent pupillary defect on the affected side (if unilateral and the fellow eye is normal) are the hallmark signs of the disease; examination in the acute phase of disease also shows optic disc edema, which is thought to occur because of ischemia from hypoperfusion of the small vessels feeding the anterior portion of the optic nerve (1,4). The fellow eye, assuming it has never experienced a similar event, usually has a small, crowded configuration often referred to as “disc at risk” with a small cup-to-disc ratio. The acute disc swelling may progress in the first 2–3 weeks after symptom onset and starts to remit by 6 weeks later. Eventually, the optic disc becomes sectorally (Figure 1) or globally pale (1). Magnetic resonance imaging (MRI) of the optic nerve is typically normal in patients with NAION and thus is not clinically indicated, although in cases where the diagnosis is uncertain, MRI orbits with contrast may be used to exclude compressive or inflammatory lesions that can mimic NAION (5).
OCT: the value of measuring peripapillary retinal nerve fibre layer (RNFL) thickness
OCT has been applied as an objective technique for diagnosis and monitoring of NAION and its differentiation from other types of optic neuropathies. Initially OCT was employed to measure peripapillary RNFL thickness. In the early stages of NAION, OCT of the RNFL may show thickening relative to the fellow eye (6-8). The RNFL thickening decreases rapidly in the subacute phase and up to 6 months after the incidence of AION. Studies with longer follow-up showed no significant decrease in thickness between month 6 and month 12 (9). The severity of RNFL thinning has been shown to correlate with the depth of VF defects and VA loss; in eyes with the typical inferior altitudinal VF loss of NAION, RNFL thickness was significantly lower in the corresponding superior quadrants and sectors of the peripapillary retina, as compared to the unaffected inferior retinal sectors as well as control eyes (10). It has also been demonstrated that for every micrometre of mean RNFL thickness lost there was a 2-decibel decrease in VF mean deviation (MD). Furthermore, VA was reduced by 1 Snellen line for every 1.6 µm of RNFL loss (7). The usefulness of OCT for measuring RNFL thickness was comparable with histologic measures in an animal (rat) model of NAION, as peripapillary inner retinal thickness measured with OCT correlated to that measured using a histologic preparation (11). Therefore, RNFL OCT can help to identify optic disc oedema acutely, particularly when subtle and be used to assess RNFL loss over time in NAION. Tracking RNFL thickness over time as the condition evolves and resolves can aid the clinician in assuring the patient that the normal or expected disease course is being followed, as ophthalmoscopic resolution of swelling may be more difficult to appreciate as pallor sets in. RNFL OCT also may be able to detect subclinical oedema of the fellow eye, which could indicate impending NAION and alert the ophthalmologist to the potential for vision loss in the near future.
The value of measuring changes in macular architecture using OCT
RNFL thickness measurement in optic neuropathies as a structural test of optic nerve integrity does present some limitations. Not all studies have found a strong correlation between superior RNFL sector thinning/change and the characteristic inferior altitudinal VF defect (10). Other publications report limited correlation between VF and a single optic disc sector (12), and an important consideration is that the peripapillary RNFL architecture does not correspond precisely to the retinotopic map that is displayed by VF testing; that is, the peripapillary RNFL map is centered on the optic disc, 15° nasal to the foveal center (where the VF is centered) (13). Furthermore, the retinal nerve fiber layer is composed of the retinal ganglion cell axons, and given that NAION often is associated with greater damage to the papillomacular bundles resulting in loss of central VA (14), assessment of the ganglion cell plus inner plexiform layer (GCIPL) and macular parameters may be a more useful method for measuring early optic nerve damage than peripapillary RNFL (15). In fact, macular thickness and superior sectoral volume have shown stronger correlations with VF sensitivity changes than RNFL parameters (12). In addition, after NAION, thinning in both the inner and outer macular sectors nasally correlates significantly with best-corrected visual acuity (BCVA) (16) and the degree of vision loss after NAION correlates directly with the severity of the damage to the papillomacular bundle (14).
With improved technology and resolution of spectral domain OCT (SD-OCT) and macular segmentation algorithms, it is possible to measure individual retinal layers, particularly ganglion cell, macular RNFL and inner plexiform layers, which contain ganglion cells bodies, their axons and dendrites, respectively. Rebolleda et al. (14) have demonstrated thinning of the ganglion cell layer in patients who had experienced NAION at least 6 months previously. They found a significant correlation between the VA and GCIPL, with the strongest correlation for the central sector of the inner plexiform layer. Larrea et al. (17) hypothesized that the ganglion cell loss could be used to detect early axonal damage in the acute phase of NAION that cannot be detected by measuring the RNFL. In a previous study (18) longitudinal measurements of the peripapillary RNFL thickness, total macular thickness, and GCIPL thickness was performed at baseline and 1, 3, and 6 months after NAION, and the results were compared to the unaffected fellow eyes. It was found that in the acute phase of NAION, there were no differences in the inner ring total macular thickness and all sector GCIPL thickness between the affected and unaffected eyes, whereas the average RNFL thickness and outer ring total macular thickness were significantly greater in the affected NAION eyes due to peripapillary RNFL edema and its extension to outer macular areas. Thinning of the GCIPL was evident in the NAION eyes at 1 month and continued at 3 months but this stabilized at 6 months. Thinning of the peripapillary RNFL and inner and outer rings total macular thickness was first evident at 3 months. Therefore, serial GCIPL thinning occurs for 1–3 months after NAION onset, which is in accordance with histological measurements of early retinal ganglion cell loss. Fard et al. (19) showed 42% loss of retinal ganglion cells with Brn3a staining starting 8 days after ischemic optic neuropathy in the rodent and continuing for 2 weeks thereafter. Of note, the total macular thickness and peripapillary RNFL thickness showed delayed thinning that occurred after GCIPL thinning. This time lag could be explained by early axonal swelling and its confounding effect or early neurodegenerative changes in the ganglion cell soma before axon (20). Kupersmith and colleagues (21) showed that marked GCIPL thinning (<5th percentile compared to normal eyes) was present with continued RNFL swelling in the subacute phase of NAION, demonstrating that RNFL measurements do not accurately reflect neuronal complex loss. Correspondingly, the degree of GCIPL thinning was directly related to VA and VF MD reduction between 1 and 6 months (21).
Exploring pathogenesis by structural analysis of the optic nerve head
Advancements of OCT with enhanced depth imaging (EDI) allows a high-resolution visualisation of the deep optic nerve including lamina cribrosa (LC), Bruch membrane opening (BMO), as well as prelaminar tissue and laminar depth (19). Our previous observation has shown that the optic nerve head in glaucoma patients is characterized by LC thinning, in contrast with NAION eyes (22). Another study reported prelaminar tissue thickening and BMO enlargement in the acute oedematous NAION eyes compared with unaffected eyes; these changes actually reversed over time (23). EDI OCT also was employed to show detailed optic nerve head crowding in NAION, which is considered the main risk factor for the development of NAION. OCT also has changed our understanding of the possible role of the size of the scleral canal in the development of NAION. Studies using confocal scanning laser ophthalmoscopy (Heidelberg Retina Tomograph II, Heidelberg Engineering GmbH, Dossenheim, Germany) suggested that NAION eyes had smaller optic disc diameter and area as compared to control eyes (8). Subsequently, Hayreh et al. (24) hypothesized that the BMO in patients with “discs at risk” for NAION was smaller than in the general population, resulting in the characteristic optic disc crowding. However, direct comparison between clinical optic disc/fundus examination and SD-OCT imaging of the optic nerve head is inappropriate, as the information derived from each method is distinct and not comparable (25). We measured BMO area with EDI OCT in post-acute NAION and their fellow eyes and compared these measurements with normal eyes. Of note, BMO area was similar in three groups (26). Similarly, several time-domain OCT studies also have shown that BMO is not smaller in patients with NAION than in age-matched controls (6,27). Another EDI OCT study also determined that BMO area in NAION and control subjects was not significantly different (28). We also measured prelaminar tissue (perpendicular distance between the internal limiting membrane (ILM) surface and anterior laminar surface) in post-acute NAION eyes. We found prelaminar tissue thickening in both NAION eyes and unaffected fellow eyes of NAION patients, as compared to eyes in normal control subjects. Given that we identified thickened prelaminar structures in both the affected and unaffected eyes of patients with NAION (Figure 2), we suggest that this change might be pathogenic and not a consequence of the disease (26). Another study also found thicker (but not significant) prelaminar tissue in NAION fellow eyes compared to controls (29). Further and even more importantly, though, EDI OCT allowed us to measure peripapillary choroidal thickness. Our group has found a thicker peripapillary choroidal structure in both the affected and unaffected fellow eyes of NAION subjects when compared to control subject eyes (30). Studies by Pérez-Sarriegui et al. (31), and Nagia et al. (28) confirmed our results in finding a bilaterally thicker choroid in patients with NAION. It seems that apparent thick prelaminar neural tissues have been pushed anteriorly by a thickened peripapillary choroid, and both features contribute to the appearance of a “crowded” disc in NAION.
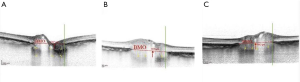
The Spectralis OCT Glaucoma Module Premium (GMP) Edition provides another method of optic nerve head analysis using BMO as the anatomical border of the rim (32,33) which is called BMO-minimum rim width (BMO-MRW). This parameter may be a more accurate reflection of axons passing through the optic nerve head opening than peripapillary RNFL thickness because it is obtained perpendicular to the axis of the neural tissue (33). BMO-MRW measurements were thinned in NAION eyes compared with the unaffected fellow eyes and control eyes. Of note, BMO-MRW was significantly thicker in contralateral unaffected NAION eyes than in a control group, again reflecting thickened prelaminar tissue in NAION fellow eyes (29). In addition, BMO-MRW could help to discriminate between chronic NAION and glaucoma. While both glaucoma and NAION patients showed similar reduction of peripapillary RNFL thickness values, only patients with glaucoma showed significantly smaller BMO-MRW values compared to NAION eyes (34).
OCT-A in NAION: what can we learn?
While a number of research tools have been used for measuring optic nerve head perfusion in acute and late NAION (35,36), advent of OCT-A enables us quantitative assessment of the circulation of peripapillary and retinal vasculature vessels in different diseases such as glaucoma, diabetic mellitus, acute rise in intra ocular pressure, and thyroid eyes disease (37-40). Peripapillary vessel density measurement by OCT-A could analyze the vessels located between the ILM and the posterior boundary of the RNFL (radial peripapillary capillaries; RPC) in addition to total peripapillary retinal vessel density and peripapillary choroidal microvasculature (39,40).
Several studies have reported findings of peripapillary OCT-A in eyes following NAION both in acute and post-acute stages. Both a reduced RPC density in acute NAION and a progressive vessel reduction within 3 months, which correlates with GCIPL thinning, have been shown (41,42). Wright Mayes et al. (43) also showed flow impairment in the RPC corresponding to structural OCT deficits of the RNFL in 80% of eyes. In eyes with optic disc swelling we employed customized image analysis with major vessel removal to measure peripapillary capillary density (PCD). We showed lower PCD in eyes with disc swelling due to NAION compared with papilledema (Figure 3) (44).
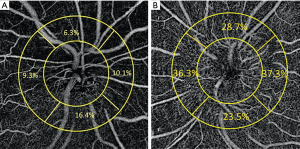
Peripapillary vessel density loss in post-acute NAION has also been reported in several studies (45-48), but it does not mean that OCT-A directly shows the optic nerve ischemia in NAION. First of all, NAION is believed to result from acute infarction of the retrolaminar segments of the optic nerve head, which are supplied primarily by flow through the short posterior ciliary arteries (49). OCT-A clearly is not able to show those deep vessels with current technology. Further and even more importantly, however, a decrease in the peripapillary vessel density in post-acute NAION is found in locations that match the VF defect positions and also correlates with the severity of peripapillary RNFL thinning. This pattern of change further matches the observations of previous investigations into VF loss in patients with glaucoma (45,50,51). This fact indicates that the dropout of RPCs is not specific to glaucomatous optic neuropathy or NAION, and that it may be secondary to peripapillary RNFL loss. In another study, we compared peripapillary RPC density in glaucoma and NAION eyes and found that the extent of RPC loss in these two conditions were not distinctive from each other (52). Notably, we utilised subject eyes with moderate or severe glaucoma with matched RNFL thickness, since it has been shown that greater PCD loss occurs with greater RNFL loss in glaucoma (51). We concluded that the loss of RPC density did not seem to be specific to either disease (glaucoma or NAION) and might instead be a consequence of RNFL damage (45,52). Similar vessel density attenuation was found between optic neuritis and NAION eyes, after adjustment for severity of optic nerve injury according to mean RNFL thickness (53).
Few studies have reported macular vessel OCT-A findings in NAION. Retinal layers were automatically segmented to the superficial and deep capillary plexuses (54). OCT-A in the macular superficial and deep capillary plexuses showed an overall rarefaction of the capillary mesh that predominates near larger vessels in post-acute NAION (48), in contrast with earlier work showing no macular vascular changes (55). However, in those studies segmentation of deep capillary plexus was subject to projection artifact, which is duplication of superficial vessels in macular deep images. In another study we imaged macular vessels in glaucoma and post-acute NAION eyes after resolving projection artifacts. While we found lower superficial macular vasculature in NAION eyes than in control eyes, deep vessels were not affected in those eyes. Thus, in a manner that resembles our observations regarding RPC loss in the peripapillary area, the loss of superficial macular vessels in NAION may be secondary to ganglion cell complex (GCC) loss and not necessarily causative/primary. In line with this hypothesis, we noted a strong correlation between superficial vasculature density and corresponding GCC integrity and VF MD (56). Such correlation was not found for parafoveal ganglion cells and deep macular vessel density. Using macular OCT-A in acute NAION showed different results. Our finding in acute NAION is contrary to secondary microvascular change theory in post-acute NAION. In early acute NAION, macular vessels density values demonstrated a significant reduction compared to control eyes, even though GCC in the macula and parafovea was not affected (57). This difference might relate to the stage of NAION, and further studies are needed.
Arteritic anterior ischemic optic neuropathy (AAION)
While giant-cell arteritis is the most common cause of AAION, other vasculitides may cause ischemic optic neuropathy (5,58). Visual loss is often more severe in AAION than in NAION (5). The affected swollen optic nerve is often pale in giant cell arteritis, and a disc at risk is not necessary for AAION. Danesh-Meyer et al. used Heidelberg Retina Tomography to compare optic disc structure after NAION and AAION and found significant excavation of optic cup after an event of AAION but not NAION (59). The finding of associated retinal or choroidal ischemia is highly suggestive of giant cell arteritis (5). In contrast to NAION, OCT has not been used extensively in the acute or chronic phases of AAION. In a recent study, optic nerve head and macular OCT of patients with AAION and NAION were used to measure choroidal vascular index. Macular choroidal vascular index was markedly and significantly lower in patients with AAION compared to patients with NAION. Conversely, no significant difference in macular choroidal vascular index was found between patients with NAION and controls. Furthermore, peripapillary choroidal vascular index was significantly lower in patients with AAION compared to patients with NAION (60). The decrease of the choroid vascular index in patients with AAION may reflect the choroidal hypoperfusion secondary to vasculitis of the posterior ciliary arteries (60). Similar to NAION, defects of the RPC were also demonstrated in AAION by using OCT-A (61,62) and OCT-A studies revealed retinal capillary perfusion defects corresponding to VF loss in AAION. However, OCT-A laminar analysis did not highlight the choroidal/choriocapillaris perfusion defects seen on fluorescein angiography, suggesting that fluorescein angiography is more sensitive in detecting choroidal perfusion defects, and subtle but potentially visually significant defects in choroidal perfusion might be missed using OCT-A (62).
Conclusions
OCT and OCT-A have allowed for a greater understanding of the structural changes that occur in ischemic optic neuropathies, and in particular, the evolution of changes in optic disc, peripapillary, and macular structures as disease resolution occurs. With these advanced imaging methods, we have recognized that there may be different risk factors for disorders such as NAION than previously thought, and further research into the contribution of observed structural changes to the onset of this disease may lead to better therapeutic interventions.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Fiona Costello and Steffen Hamann) for the series “The Use of OCT as a Biomarker in Neuro-ophthalmology” published in Annals of Eye Science. The article has undergone external peer review.
Conflicts of Interest: The authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2019.12.05). The series “The Use of OCT as a Biomarker in Neuro-ophthalmology” was commissioned by the editorial office without any funding or sponsorship. PSS reports grants and personal fees from Quark Pharmaceuticals, outside the submitted work. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hayreh SS. Ischemic optic neuropathies - where are we now? Graefes Arch Clin Exp Ophthalmol 2013;251:1873-84. [Crossref] [PubMed]
- Hattenhauer MG, Leavitt JA, Hodge DO, et al. Incidence of nonarteritic anterior ischemic optic neuropathy. Am J Ophthalmol 1997;123:103-7. [Crossref] [PubMed]
- Preechawat P, Bruce BB, Newman NJ, et al. Anterior ischemic optic neuropathy in patients younger than 50 years. Am J Ophthalmol 2007;144:953-60. [Crossref] [PubMed]
- . Characteristics of patients with nonarteritic anterior ischemic optic neuropathy eligible for the Ischemic Optic Neuropathy Decompression Trial. Arch Ophthalmol 1996;114:1366-74. [Crossref] [PubMed]
- Biousse V, Newman NJ. Ischemic Optic Neuropathies. N Engl J Med 2015;372:2428-36. [Crossref] [PubMed]
- Contreras I, Rebolleda G, Noval S, et al. Optic disc evaluation by optical coherence tomography in nonarteritic anterior ischemic optic neuropathy. Invest Ophthalmol Vis Sci 2007;48:4087-92. [Crossref] [PubMed]
- Contreras I, Noval S, Rebolleda G, et al. Follow-up of nonarteritic anterior ischemic optic neuropathy with optical coherence tomography. Ophthalmology 2007;114:2338-44. [Crossref] [PubMed]
- Saito H, Tomidokoro A, Tomita G, et al. Optic disc and peripapillary morphology in unilateral nonarteritic anterior ischemic optic neuropathy and age- and refraction-matched normals. Ophthalmology 2008;115:1585-90. [Crossref] [PubMed]
- Dotan G, Goldstein M, Kesler A, et al. Long-term retinal nerve fiber layer changes following nonarteritic anterior ischemic optic neuropathy. Clin Ophthalmol 2013;7:735-40. [Crossref] [PubMed]
- Deleón-Ortega J, Carroll KE, Arthur SN, et al. Correlations between retinal nerve fiber layer and visual field in eyes with nonarteritic anterior ischemic optic neuropathy. Am J Ophthalmol 2007;143:288-94. [Crossref] [PubMed]
- Maekubo T, Chuman H, Kodama Y, et al. Evaluation of inner retinal thickness around the optic disc using optical coherence tomography of a rodent model of nonarteritic ischemic optic neuropathy. Jpn J Ophthalmol 2013;57:327-32. [Crossref] [PubMed]
- Papchenko T, Grainger BT, Savino PJ, et al. Macular thickness predictive of visual field sensitivity in ischaemic optic neuropathy. Acta Ophthalmol 2012;90:e463-9. [Crossref] [PubMed]
- Aghsaei Fard M, Fakhree S, Ameri A. Posterior Pole Retinal Thickness for Detection of Structural Damage in Anterior Ischaemic Optic Neuropathy. Neuroophthalmology 2013;37:183-91. [Crossref] [PubMed]
- Rebolleda G, Sanchez-Sanchez C, Gonzalez-Lopez JJ, et al. Papillomacular bundle and inner retinal thicknesses correlate with visual acuity in nonarteritic anterior ischemic optic neuropathy. Invest Ophthalmol Vis Sci 2015;56:682-92. [Crossref] [PubMed]
- Kardon RH. Role of the macular optical coherence tomography scan in neuro-ophthalmology. J Neuroophthalmol 2011;31:353-61. [Crossref] [PubMed]
- Fernández-Buenaga R, Rebolleda G, Muñoz-Negrete FJ, et al. Macular thickness. Ophthalmology 2009;116:1587.e1-3. [PubMed]
- Larrea BA, Iztueta MG, Indart LM, et al. Early axonal damage detection by ganglion cell complex analysis with optical coherence tomography in nonarteritic anterior ischaemic optic neuropathy. Graefes Arch Clin Exp Ophthalmol 2014;252:1839-46. [Crossref] [PubMed]
- Akbari M, Abdi P, Fard MA, et al. Retinal Ganglion Cell Loss Precedes Retinal Nerve Fiber Thinning in Nonarteritic Anterior Ischemic Optic Neuropathy. J Neuroophthalmol 2016;36:141-6. [Crossref] [PubMed]
- Fard MA, Ebrahimi KB, Miller NR. RhoA activity and post-ischemic inflammation in an experimental model of adult rodent anterior ischemic optic neuropathy. Brain Res 2013;1534:76-86. [Crossref] [PubMed]
- Chauhan BC, Stevens KT, Levesque JM, et al. Longitudinal in vivo imaging of retinal ganglion cells and retinal thickness changes following optic nerve injury in mice. PloS One 2012;7:e40352 [Crossref] [PubMed]
- Kupersmith MJ, Garvin MK, Wang JK, et al. Retinal ganglion cell layer thinning within one month of presentation for optic neuritis. Mult Scler 2016;22:641-8. [Crossref] [PubMed]
- Fard MA, Afzali M, Abdi P, et al. Optic Nerve Head Morphology in Nonarteritic Anterior Ischemic Optic Neuropathy Compared to Open-Angle Glaucoma. Invest Ophthalmol Vis Sci 2016;57:4632-40. [Crossref] [PubMed]
- Rebolleda G, García-Montesinos J, De Dompablo E, et al. Bruch's membrane opening changes and lamina cribrosa displacement in non-arteritic anterior ischaemic optic neuropathy. Br J Ophthalmol 2017;101:143-9. [Crossref] [PubMed]
- Hayreh SS, Zimmerman MB. Nonarteritic anterior ischemic optic neuropathy: refractive error and its relationship to cup/disc ratio. Ophthalmology 2008;115:2275-81. [Crossref] [PubMed]
- Chauhan BC, Burgoyne CF. From clinical examination of the optic disc to clinical assessment of the optic nerve head: a paradigm change. Am J Ophthalmol 2013;156:218-227.e2. [Crossref] [PubMed]
- Moghimi S, Afzali M, Akbari M, et al. Crowded optic nerve head evaluation with optical coherence tomography in anterior ischemic optic neuropathy. Eye (Lond) 2017;31:1191-8. [Crossref] [PubMed]
- Chan CK, Cheng AC, Leung CK, et al. Quantitative assessment of optic nerve head morphology and retinal nerve fibre layer in non-arteritic anterior ischaemic optic neuropathy with optical coherence tomography and confocal scanning laser ophthalmoloscopy. Br J Ophthalmol 2009;93:731-5. [Crossref] [PubMed]
- Nagia L, Huisingh C, Johnstone J, et al. Peripapillary pachychoroid in nonarteritic anterior ischemic optic neuropathy. Invest Ophthalmol Vis Sci 2016;57:4679-85. [Crossref] [PubMed]
- Rebolleda G, Pérez-Sarriegui A, Díez-Álvarez L, et al. Lamina cribrosa position and Bruch’s membrane opening differences between anterior ischemic optic neuropathy and open-angle glaucoma. Eur J Ophthalmol 2019;29:202-9. [Crossref] [PubMed]
- Fard MA, Abdi P, Kasaei A, et al. Peripapillary choroidal thickness in nonarteritic anterior ischemic optic neuropathy. Invest Ophthalmol Vis Sci 2015;56:3027-33. [PubMed]
- Pérez-Sarriegui A, Muñoz-Negrete FJ, Noval S, et al. Automated Evaluation of Choroidal Thickness and Minimum Rim Width Thickness in Nonarteritic Anterior Ischemic Optic Neuropathy. J Neuroophthalmol 2018;38:7-12. [Crossref] [PubMed]
- Chauhan BC, Danthurebandara VM, Sharpe GP, et al. Bruch's membrane opening minimum rim width and retinal nerve fiber layer thickness in a normal white population: a multicenter study. Ophthalmology 2015;122:1786-94. [Crossref] [PubMed]
- Reis AS, O'Leary N, Yang H, et al. Influence of clinically invisible, but optical coherence tomography detected, optic disc margin anatomy on neuroretinal rim evaluation. Invest Ophthalmol Vis Sci 2012;53:1852-60. [Crossref] [PubMed]
- Resch H, Mitsch C, Pereira I, et al. Optic nerve head morphology in primary open‐angle glaucoma and nonarteritic anterior ischaemic optic neuropathy measured with spectral domain optical coherence tomography. Acta ophthalmol 2018;96:e1018-24. [Crossref] [PubMed]
- Traversi C, Bianciardi G, Tasciotti A, et al. Fractal analysis of fluoroangiographic patterns in anterior ischaemic optic neuropathy and optic neuritis: a pilot study. Clin Exp Ophthalmol 2008;36:323-8. [Crossref] [PubMed]
- Collignon-Robe NJ, Feke GT, Rizzo JF III. Optic nerve head circulation in nonarteritic anterior ischemic optic neuropathy and optic neuritis. Ophthalmology 2004;111:1663-72. [Crossref] [PubMed]
- Jia Y, Wei E, Wang X, et al. Optical coherence tomography angiography of optic disc perfusion in glaucoma. Ophthalmology 2014;121:1322-32. [Crossref] [PubMed]
- Shin YI, Nam KY, Lee SE, et al. Peripapillary microvasculature in patients with diabetes mellitus: An optical coherence tomography angiography study. Sci Rep 2019;9:15814. [Crossref] [PubMed]
- Moghimi S. Changes in Optic Nerve Head Vessel Density After Acute Primary Angle Closure Episode. Invest Ophthalmol Vis Sci 2019;60:552-8. [Crossref] [PubMed]
- Jamshidian Tehrani M, Mahdizad Z, et al. Early macular and peripapillary vasculature dropout in active thyroid eye disease. Graefes Arch Clin Exp Ophthalmol 2019;257:2533-40. [Crossref] [PubMed]
- Sharma S, Ang M, Najjar RP, et al. Optical coherence tomography angiography in acute non-arteritic anterior ischaemic optic neuropathy. Br J Ophthalmol 2017;101:1045-51. [Crossref] [PubMed]
- Rebolleda G, Diez-Alvarez L, Garcia Marin Y, et al. Reduction of Peripapillary Vessel Density by Optical Coherence Tomography Angiography from the Acute to the Atrophic Stage in Non-Arteritic Anterior Ischaemic Optic Neuropathy. Ophthalmologica 2018;240:191-9. [Crossref] [PubMed]
- Wright Mayes E, Cole ED, Dang S, et al. Optical Coherence Tomography Angiography in Nonarteritic Anterior Ischemic Optic Neuropathy. J Neuroophthalmol 2017;37:358-64. [Crossref] [PubMed]
- Fard MA, Jalili J, Sahraiyan A, et al. Optical coherence tomography angiography in optic disc swelling. Am J Ophthalmol 2018;191:116-23. [Crossref] [PubMed]
- Hata M, Oishi A, Muraoka Y, et al. Structural and Functional Analyses in Nonarteritic Anterior Ischemic Optic Neuropathy: Optical Coherence Tomography Angiography Study. J Neuroophthalmol 2017;37:140-8. [Crossref] [PubMed]
- Song Y, Min JY, Mao L, et al. Microvasculature dropout detected by the optical coherence tomography angiography in nonarteritic anterior ischemic optic neuropathy. Lasers Surg Med 2018;50:194-201. [Crossref] [PubMed]
- Liu CH, Kao LY, Sun MH, et al. Retinal vessel density in optical coherence tomography angiography in optic atrophy after nonarteritic anterior ischemic optic neuropathy. J Ophthalmol 2017;2017:9632647 [Crossref] [PubMed]
- Augstburger E, Zeboulon P, Keilani C, et al. Retinal and Choroidal Microvasculature in Nonarteritic Anterior Ischemic Optic Neuropathy: An Optical Coherence Tomography Angiography Study. Invest Ophthalmol Vis Sci 2018;59:870-7. [Crossref] [PubMed]
- Olver JM, Spalton DJ, McCartney AC. Microvascular study of the retrolaminar optic nerve in man: the possible significance in anterior ischaemic optic neuropathy. Eye (Lond) 1990;4:7-24. [Crossref] [PubMed]
- Ghahari E, Bowd C, Zangwill LM, et al. Association of Macular and Circumpapillary Microvasculature with Visual Field Sensitivity in Advanced Glaucoma. Am J Ophthalmol 2019;204:51-61. [Crossref] [PubMed]
- Yarmohammadi A, Zangwill LM, Diniz-Filho A, et al. Relationship between Optical Coherence Tomography Angiography Vessel Density and Severity of Visual Field Loss in Glaucoma. Ophthalmology 2016;123:2498-508. [Crossref] [PubMed]
- Fard MA, Suwan Y, Moghimi S, et al. Pattern of peripapillary capillary density loss in ischemic optic neuropathy compared to that in primary open-angle glaucoma. PloS One 2018;13:e0189237 [Crossref] [PubMed]
- Fard MA, Yadegari S, Ghahvechian H, et al. Optical coherence tomography angiography of a pale optic disc in demyelinating optic neuritis and ischemic optic neuropathy. J Neuroophthalmol 2019;39:339-44. [Crossref] [PubMed]
- Campbell JP, Zhang M, Hwang TS, et al. Detailed Vascular Anatomy of the Human Retina by Projection-Resolved Optical Coherence Tomography Angiography. Sci Rep 2017;7:42201. [Crossref] [PubMed]
- Liu CH, Wu WC, Sun MH, et al. Comparison of the Retinal Microvascular Density Between Open Angle Glaucoma and Nonarteritic Anterior Ischemic Optic Neuropathy. Invest Ophthalmol Vis Sci 2017;58:3350-6. [Crossref] [PubMed]
- Fard MA, Fakhraee G, Ghahvechian H, et al. Macular Vascularity in Ischemic Optic Neuropathy Compared to Glaucoma by Projection-Resolved Optical Coherence Tomography Angiography. Am J Ophthalmol 2020; [Crossref] [PubMed]
- Fard MA, Ghahvechian H, Sahrayan A, et al. Early Macular Vessel Density Loss in Acute Ischemic Optic Neuropathy Compared to Papilledema: Implications for Pathogenesis. Transl Vis Sci Technol 2018;7:10. [Crossref] [PubMed]
- Fard MA, Nozarian Z, Veisi A. Arteritic anterior ischaemic optic neuropathy with unusual systemic manifestations. Neuroophthalmology 2019; [Crossref]
- Danesh-Meyer H, Savino PJ, Spaeth GL, et al. Comparison of arteritis and nonarteritic anterior ischemic optic neuropathies with the Heidelberg Retina Tomograph. Ophthalmology 2005;112:1104-12. [Crossref] [PubMed]
- Pellegrini M, Giannaccare G, Bernabei F, et al. Choroidal Vascular Changes in Arteritic and Nonarteritic Anterior Ischemic Optic Neuropathy. Am J Ophthalmol 2019;205:43-9. [Crossref] [PubMed]
- Balducci N, Morara M, Veronese C, et al. Optical coherence tomography angiography in acute arteritic and non-arteritic anterior ischemic optic neuropathy. Graefes Arch Clin Exp Ophthalmol 2017;255:2255-61. [Crossref] [PubMed]
- Gaier ED, Gilbert AL, Cestari DM, et al. Optical coherence tomographic angiography identifies peripapillary microvascular dilation and focal non-perfusion in giant cell arteritis. Br J Ophthalmol 2018;102:1141-6. [PubMed]
Cite this article as: Fard MA, Ghahvehchian H, Subramanian PS. Optical coherence tomography in ischemic optic neuropathy. Ann Eye Sci 2020;5:6.


