
Diabetic retinopathy: an inflammatory disease
Introduction
Diabetic retinopathy (DR) is an important complication of both type 1 and type 2 diabetes mellitus (1) and the leading cause of preventable blindness in working age population worldwide (2). DR affects about one-third of diabetic population and 10% has sight-threatening complications such as diabetic macular edema (DME) and proliferative DR (PDR) (3). The International Clinical Diabetic Retinopathy Disease Severity Scale divides DR in five different stages based on clinical signs of vascular impairment detectable on fundus examination: no apparent DR, mild, moderate and severe non proliferative DR (NPDR), and PDR, as defined by the presence of retinal neovascularization or vitreous and/or preretinal hemorrhage (4). DR could remain silent for a long period of time and, by the time symptoms become manifest, disease may have already progressed to advanced stages. Functional changes in eyes of diabetic patients can be detected even before the appearance of initial signs of DR, as demonstrated by means of multifocal electroretinogram (mfERG) and visual evoked potentials (VEPs) (5,6). This suggests that damage in neuroretinal function in DR begins before clinically visible vascular changes occur (7-9), as confirmed by recent studies performed with spectral domain (SD)-optical coherence tomography (OCT) and OCT-angiography (10-18). Therefore, there is a need for evaluation and validation of (possibly) non-invasive biomarkers that can be used for early detection of subclinical signs of DR. Even if many previous studies focused on the role of vascular dysfunction in DR pathogenesis, diabetic microvasculopathy cannot completely explain early neuroretinal damage and thus DR could not simply be considered a pure vasculopathy. Functional alterations were demonstrated in the earliest stages of disease before the development of vascular dysfunction (5,6). Even if the exact mechanisms initiating neuroretinal damage in DR are not fully understood, actual evidence focuses on the role of chronic hyper-glycaemia-induced inflammation as critical contributing factor in DR pathogenesis (19,20) and several studies described the association between high levels of systemic and local inflammatory molecules and the development and progression of DR (21-26).
In this review we will first discuss current literature focused on the role of inflammation in the pathogenesis and progression of DR and then we will describe retinal features that may be used as non-invasive clinical inflammatory biomarkers in the everyday clinical practice that may be useful to choose the most appropriate treatment option.
Molecular basis of inflammation in DR
DR has a complex multifactorial etiology and all retinal cellular elements (vascular, neural and glial elements forming the so-called retinal neurovascular unit) are involved in the pathological process (27), even before the onset of clinical signs of DR (28-33). DR is nowadays considered a chronic low-grade inflammatory disorder involving a cascade of inflammatory mediators and adhesion molecules (19,20,34-36). Subclinical chronic inflammation contributes to diabetes occurrence and to the development of its long-term complications, including DR (37). Knowing inflammatory pathways could be helpful to create strategies to prevent and control diabetes and its complications before they cause irreversible organ damage (37).
The major factor determining retinal dysfunction in DR is represented by chronic hyper-glycaemia (19,20). Hyper-glycaemia causes the elevation of intracellular glucose and, consequently, the activation of four different cellular glucose metabolic pathways: the diacylglycerol (DAG)-protein kinase C (PKC), advanced glycation end products (AGE)/AGE receptors (RAGE), sorbitol and hexosamine pathways (38-40). This leads to cellular damage in terms of micro-vascular dysfunction, neuronal apoptosis, glial reactivity and component deposition (38-40). All these interconnected aspects are linked to upregulation of angiogenic and inflammatory mediators and to altered growth factor signaling, with recruitment and infiltration of macrophages, monocytes and neutrophils and consequent aberrant inflammatory response (28,31,32,41-43).
Endothelial dysfunction associated with inflammation determines increased vascular permeability, alteration of blood flow, oxidative stress and angiogenesis and has been related to increased expression of inflammatory adhesion molecules (ICAM-1, VCAM-1 and E-selectin) in the endothelium (44,45). Elevated levels of these molecules lead to adhesion and accumulation of leukocytes within retinal vessels, one of the very early events that occur in diabetic retina inflammation (within one week in experimental diabetes) (46-48). This results in loss of pericytes, formation of acellular capillaries and consequent break-down of the blood-retinal barrier (28,49-51) that progresses toward increased retinal vascular permeability, development of DME, and neovascularization (PDR) (41).
Retinal glial cells including astrocytes, Müller cells (MCs) and microglia play a central structural role and contribute to maintain homeostasis in the retina. Their activation is considered another critical feature involved in the initiation and amplification of inflammation in diabetic neuroretinal dysfunction (52-54). Microglia cells are thought to be the first responders to hyperglycemic stress undergoing a shift in their phenotype from “surveying microglia” to “activated microglia” (55) and migrating from the inner to the outer retinal layers (56-58). Activated microglia cells start to produce pro-inflammatory mediators such as TNF-alpha, IL-6, MCP-1 and VEGF (52,59) amplifying the inflammatory response that triggers MCs reactive gliosis (60), a process consisting of hypertrophy, cellular proliferation and increase in intermediate filament proteins such as glial fibrillary acidic protein (GFAP) (33). Glial activation occurs early in DR pathogenesis, as demonstrated by the upregulation of GFAP and aquaporin 4 (AQP4) produced by MCs both in the retinas of animal and human models of DM and in vivo in human ocular fluids and specifically in the aqueous humor of patients with DM but no DR or with early signs of DR compared to healthy controls (33,61-64).
Even if VEGF is the most studied factor involved in DR pathogenesis and the main target of available therapeutic strategies, its role cannot explain alone all the events taking place in DR onset and progression. VEGF selective inhibition is not sufficient to stop the inflammatory cascade in DR and anti-VEGF therapies are frequently of transient benefit, especially in DME treatment, needing repeated injections over time (65,66) and suggesting the involvement of other molecular pathways. It has been demonstrated that a wide range of systemic and local inflammatory biomarkers are involved and act together in DR: vascular adhesion molecules (VCAM-1, ICAM-1, E-selectin, sVAP), pro-inflammatory cytokines (TNF-alpha, IL-1alpha, 1beta, 6, 8, HMGB1), anti-inflammatory cytokines (IL-10), pro-inflammatory/angiogenic chemokines (CP-1, MIF, SDF-1, fractalkine), anti-inflammatory/antiangiogenic chemokines (IP10, MIG), transcription factors (HIF-1, NF-κB), pro-inflammatory/angiogenic growth factors (VEGF, PGF, IGF1, CTGF, stem cell factor), anti-inflammatory/antiangiogenic growth factors (PEDF), anti-inflammatory/proangiogenic growth factors (EPO), and innate immune response cells (retinal endothelial cells with toll-like receptors) (67). In addition, there is some evidence suggesting that pro-inflammatory molecules’ levels increase with DR progression: in particular, serum concentration of circulating cytokines such as TNF-alpha, VEGF, IL-1beta and IL-6 seem to be associated with DR severity and not only with the presence of DR (68-73). Also elevated systemic neutrophil count was found to be associated with the presence and severity of DR, indicating that neutrophil-mediated inflammation may play a role in DR pathogenesis (74). In the eye, increased expression of vitreous IL-8 seems to correlate with worse visual acuity in diabetic patients in one study (75), while another study found an increased aqueous concentration of IL-8 in severe NPDR, suggesting that inflammation may contribute to the development of neovascularization (76). IL-6 aqueous concentration has a positive correlation with macular thickness, indicating that IL-6 may play a role in the development of DME (77). On the contrary, both circulating and aqueous levels of IL-10, a cytokine able to down-regulate T lymphocytes helper 1 response and VEGF, are decreased in DR and DME (78,79) and one recent study demonstrated a correlation between low circulating IL-10 concentration and a cystoid pattern of DME (80) (Table 1).
Table 1
| Biochemical biomarkers | Associated features | References |
|---|---|---|
| Cellular | ||
| VCAM-1, ICAM-1, selectins | Leukocyte adhesion | ( |
| TNF-alpha, IL-1, 6 and 10, MCP-1 and 2, VEGF, IFN-gamma, IP-10 | Microglia activation | ( |
| GFAP, AQP4 | MCs activation | ( |
| Clinical | ||
| TNF-alpha, VEGF, IL-1beta, IL-6 | DR progression | ( |
| IL-8 | Worse functional outcome* | ( |
| IL-6, IL-10 | DME and SND onset | ( |
*, worse visual acuity after vitrectomy performed for PDR. MCs, Müller cells; DME, diabetic macular edema; SND, serous neuroretinal detachment, PDR, proliferative diabetic retinopathy.
Therefore, there are many systemic and local (vitreous/aqueous) inflammatory molecules upregulated at any stage of DR. All of them interact to create a pro-inflammatory environment that contributes to occurrence, maintenance and progression of retinopathy.
Clinical biomarkers of inflammation in DR
A circulating molecule has to be highly specific to the retina to be a reliable biomarker of retinal diabetic disease rather than a marker of systemic disease (64). Aqueous or vitreous biomarkers are more specific but also more difficult to obtain needing an invasive procedure (in particular to collect a sample of vitreous fluid). Therefore, thanks to the great advances in retinal imaging technologies of the last years, a new concept of non-invasive “imaging biomarker” of retinal inflammation has emerged and has made its way to the study of patients with diabetes (64). Clinical research has consequently developed great interest in finding specific retinal parameters of the retinal inflammatory condition in DR and DME and there is a growing body of scientific evidence on the importance of this topic. Nowadays, proposed imaging biomarkers of inflammation in DR include subfoveal neuroretinal detachment (SND) and hyperreflective retinal spots/foci (HRS) visible on SD-OCT and increased foveal autofluorescence (iFAF) visible on fundus autofluorescence imaging (56,58,82-86) (Figure 1). In addition, an increase in thickness of the inner nuclear layer was described on SD-OCT in patients with NPDR and this may represent a clinical sign of MCs activation due to hypertrophy of these cells (11). Hyperreflective foci were described on SD-OCT also in the vitreous of patients with diabetes, with the number increasing as the stage of DR progresses (87,88). Previous studies had demonstrated in patients undergoing vitrectomy for complicated PDR that there was an increased number of lymphocytes that was correlated to disease severity (89,90). Thus, hyperreflective foci detected with SD-OCT in the vitreous of these patients were interpreted as inflammatory cells infiltrating the vitreoretinal interface and vitreous gel (87) (Table 2).
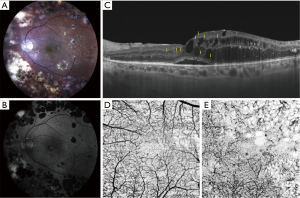
Table 2
| Clinical biomarkers | Imaging technique | References |
|---|---|---|
| Subfoveal neuroretinal detachment | OCT | ( |
| Hyper-reflective retinal spots/foci | OCT | ( |
| Increased fundus autofluorescence | FAF | ( |
| Vitreous hyper-reflective foci | OCT | ( |
| Increased INL thickness | OCT | ( |
OCT, optical coherence tomography; FAF, fundus autoflurescence in the fovea; INL, inner nuclear layer.
SND
SND is detected on OCT as a hyporeflective area beneath the neuroretina (extracellular fluid accumulation between the outer segments of photoreceptors and the retinal pigment epithelium) in approximately 15-30% of eyes with DME (85,101-104). The presence of SND has been associated with higher levels of local inflammatory molecules, in particular IL-6 (85) and in the past it was considered to correlate with a poorer visual outcome (91,92). SND is more likely to develop in the presence of increased choroidal thickness (increased choriocapillaris permeability and outer BRB impairment) (93,94), disrupted external limiting membrane (ELM) (94,101) and in association with a significantly increased number of HRS, another clinical sign of local inflammation (83,94).
Hyper-reflective retinal spots
HRS are not a specific sign of DR and have been described in other chorioretinal diseases, such as age-related macular degeneration (AMD), retinal vein occlusion and recently uveitis (56,95,96,105-110). In all these conditions HRS are visible in both inner and outer retina and also near, in the walls or inside the lumen of retinal cysts (56,95,96,105-109). Histologic evidence from human donor eyes with AMD and acquired vitelliform lesions demonstrated that HRS visualized on SD-OCT may originate from both anteriorly migrated retinal pigment epithelium cells and lipid-filled cells (thought to correspond to activated microglia) (97,111,112). HRS are increased in number in patients with diabetes (in both preclinical and early clinical DR) versus normal subjects and they are thought to represent aggregates of activated microglial cells that progressively migrate from the inner to the outer retina, confirming their role as inflammatory biomarkers (56). Further evidence supporting this hypothesis came from a recent study demonstrating a positive correlation between aqueous concentration of soluble CD14 (a cytokine associated with immune response, expressed in microglia, monocytes and macrophages) and the number of HRS in DME (98). In particular, HRS related to microglia activation seem to have specific characteristics, such as small dimension (<30 µm), reflectivity similar to nerve fiber layer, absence of back-shadowing, and location in both inner and outer retina; additionally, they do not correspond to any specific lesion on fundus examination (56,58).
Increased fundus autofluorescence in the fovea
iFAF was described in a large proportion of eyes with DME (82,84,99) and is correlated to reduced retinal sensitivity determined with microperimetry, indicating an impaired neurosensory retina function in that area (82). In DME, it is thought that areas of iFAF are caused by accumulation of oxidative products induced by activated microglial cells (100,113), thus suggesting that also iFAF may be considered as an imaging biomarker of microglial activation in DME (82).
Conclusions
Even if not all mechanisms are still fully elucidated, actual evidence highlights the important role of inflammation in DR pathogenesis and progression, and DR is considered a chronic, low-grade inflammatory disease. In particular leukocyte recruitment and adhesion to retinal vessels and glial cell activation are recognized as early events occurring in diabetic retina dysfunction, even before the onset of clinically evident signs of retinopathy.
Current available treatment options for DR (intravitreal injections of anti-VEGF or corticosteroids, laser photocoagulation, vitreoretinal surgery) are applicable only at advanced stages of disease (DME, severe NPDR, PDR) (64,114-117). In early stages the only therapeutic strategy that physicians can offer is the control of modifiable risk factors for DR such as glycaemia and systemic blood pressure (64,117). Therefore, there is an urgent need for non-invasive early-detection molecular and clinical biomarkers of subclinical and early DR that can help in DR management before irreversible damage occurs.
Measuring local and systemic biomarkers of inflammation may become a useful tool to differentiate patients with diabetes on the basis of their risk of disease progression, however, further validation is needed. In addition, the increasing understanding on inflammation involvement in DR is stimulating the interest in targeting specific inflammatory pathways to improve DR prevention and care, even if specific interventions are still not part of a routine clinical practice and further work is needed on this front.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Jay M. Stewart) for the series “Diabetic Retinopathy” published in Annals of Eye Science. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2018.09.05). The series “Diabetic Retinopathy” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- King H, Aubert RE, Hermann WH. Global burden of diabetes, 1995-2025: prevalence, numerical estimates and projections. Diabetes Care 1998;21:1414-31. [Crossref] [PubMed]
- Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet 2010;376:124-36. [Crossref] [PubMed]
- Yau JW, Rogers SL, Kawasaki RMeta-analysis for Eye Disease (META-EYE) Study Group, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012;35:556-64. [Crossref] [PubMed]
- Wilkinson CP, Ferris FL, Klein RE, et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology 2003;110:1677-82. [Crossref] [PubMed]
- Bearse MA Jr, Adams AJ, Han Y, et al. A multifocal electroretinogram model predicting the development of diabetic retinopathy. Prog Retin Eye Res 2006;25:425-48. [Crossref] [PubMed]
- Parisi V, Uccioli L. Visual electrophysiological responses in persons with type 1 diabetes. Diabetes Metab Res Rev 2001;17:12-8. [Crossref] [PubMed]
- Gardner TW, Abcouwer SF, Barber AJ, Jackson GR. An integrated approach to diabetic retinopathy research. Arch Ophthalmol 2011;129:230-5. [Crossref] [PubMed]
- Lung JC, Swann PG, Wong DSH, et al. Global flash multifocal electroretinogram: early detection of local functional changes and its correlations with optical coherence tomography and visual field tests in diabetic eyes. Doc Ophthalmol 2012;125:123-35. [Crossref] [PubMed]
- Sohn EH, van Dijk HW, Jiao C, et al. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc Natl Acad Sci USA 2016;113:E2655-E2664. [Crossref] [PubMed]
- Vujosevic S, Muraca A, Alkabes M, et al. Early microvascular and neural changes in patients with type 1 and type 2 diabetes mellitus without clinical signs of diabetic retinopathy. Retina 2017; [Epub ahead of print]. [Crossref] [PubMed]
- Vujosevic S, Midena E. Retinal layers changes in human preclinical and early clinical diabetic retinopathy support early retinal neuronal and Müller cells alterations. J Diabetes Res 2013;2013:905058 [Crossref] [PubMed]
- van Dijk HW, Verbraak FD, Kok PH, et al. Decreased retinal ganglion cell layer thickness in patients with type 1 diabetes. Invest Ophthalmol Vis Sci 2010;51:3660-5. [Crossref] [PubMed]
- van Dijk HW, Verbraak FD, Kok PH, et al. Early neurodegeneration in the retina of type 2 diabetic patients. Invest Ophthalmol Vis Sci 2012;53:2715-9. [Crossref] [PubMed]
- Srinivasan S, Pritchard N, Sampson GP, et al. Retinal tissue thickness in type 1 and type 2 diabetes. Clin Exp Optom 2016;99:78-83. [Crossref] [PubMed]
- Scarinci F, Picconi F, Virgili G, et al. Single retinal layer evaluation in patients with type 1 diabetes with no or early signs of diabetic retinopathy: the first hint of neurovascular crosstalk damage between neurons and capillaries? Ophthalmologica 2017;237:223-31. [Crossref] [PubMed]
- de Carlo TE, Chin AT, Bonini Filho MA, et al. Detection of microvascular changes in eyes of patients with diabetes but not clinical diabetic retinopathy using optical coherence tomography angiography. Retina 2015;35:2364-70. [Crossref] [PubMed]
- Dimitrova G, Chihara E, Takahashi H, et al. Quantitative retinal optical coherence tomography angiography in patients with diabetes without diabetic retinopathy. Invest Ophthalmol Vis Sci 2017;58:190-6. [Crossref] [PubMed]
- Simonett JM, Scarinci F, Picconi F, et al. Early microvascular retinal changes in optical coherence tomography angiography in patients with type 1 diabetes mellitus. Acta Ophthalmol 2017;95:e751-e755. [Crossref] [PubMed]
- Adamis AP. Is diabetic retinopathy an inflammatory disease? Br J Ophthalmol 2002;86:363-5. [Crossref] [PubMed]
- Joussen AM, Poulaki V, Le ML, et al. A central role for inflammation in the pathogenesis of diabetic retinopathy. FASEB J 2004;18:1450-2. [Crossref] [PubMed]
- Funatsu H, Noma H, Mimura T, et al. Association of vitreous inflammatory factors with diabetic macular edema. Ophthalmology 2009;116:73-9. [Crossref] [PubMed]
- Elner SG, Elner VM, Jaffe GJ, et al. Cytokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Curr Eye Res 1995;14:1045-53. [Crossref] [PubMed]
- Mitamura Y, Takeuchi S, Matsuda A, et al. Monocyte chemotactic protein-1 in the vitreous of patients with proliferative diabetic retinopathy. Ophthalmologica 2001;215:415-8. [Crossref] [PubMed]
- Abu El-Asrar AM, Struyf S, Kangave D, et al. Chemokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Eur Cytokine Netw 2006;17:155-65. [PubMed]
- Muramatsu D, Wakabayashi Y, Usui Y, et al. Correlation of complement fragment C5a with inflammatory cytokines in the vitreous of patients with proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 2013;251:15-7. [Crossref] [PubMed]
- Wakabayashi Y, Usui Y, Okunuki Y, et al. Correlation of vascular endothelial growth factor with chemokines in the vitreous in diabetic retinopathy. Retina 2010;30:339-44. [Crossref] [PubMed]
- Antonetti DA, Klein R, Gardner TW, et al. Diabetic Retinopathy. N Engl J Med 2012;366:1227-39. [Crossref] [PubMed]
- Joussen AM, Poulaki V, Qin W, et al. Retinal vascular endothelial growth factor induces intercellular adhesion molecule-1 and endothelial nitric oxide synthase expression and initiates early diabetic retinal leukocyte adhesion in vivo. Am J Pathol 2002;160:501-9. [Crossref] [PubMed]
- Martin PM, Roon P, Van Ells TK, et al. Death of retinal neurons in streptozotocin-induced diabetic mice. Invest Ophthalmol Vis Sci 2004;45:3330-6. [Crossref] [PubMed]
- Lecleire-Collet A, Tessier LH, Massin P, et al. Advanced glycation end products can induce glial reaction and neuronal degeneration in retinal explants. Br J Ophthalmol 2005;89:1631-3. [Crossref] [PubMed]
- Antonetti DA, Barber AJ, Bronson SKJDRF Diabetic Retinopathy Center Group, et al. Diabetic retinopathy: seeing beyond glucose-induced microvascular disease. Diabetes 2006;55:2401-11. [Crossref] [PubMed]
- Vujosevic S, Micera A, Bini S, et al. Proteome analysis of retinal glial cells-related inflammatory cytokines in the aqueous humour of diabetic patients. Acta Ophthalmol 2016;94:56-64. [Crossref] [PubMed]
- Vujosevic S, Micera A, Bini S, et al. Aqueous humor biomarkers of Müller cell activation in diabetic eyes. Invest Ophthalmol Vis Sci 2015;56:3913-8. [Crossref] [PubMed]
- Yuuki T, Kanda T, Kimura Y, et al. Inflammatory cytokines in vitreous fluid and serum of patients with diabetic vitreoretinopathy. J Diabetes Complications 2001;15:257-9. [Crossref] [PubMed]
- Grant MB, Afzal A, Spoerri P, et al. The role of growth factors in the pathogenesis of diabetic retinopathy. Expert Opin Investig Drugs 2004;13:1275-93. [Crossref] [PubMed]
- Khalfaoui T, Lizard G, Ouertani-Meddeb A. Adhesion molecules (ICAM-1 and VCAM-1) and diabetic retinopathy in type 2 diabetes. J Mol Histol 2008;39:243-9. [Crossref] [PubMed]
- Lontchi-Yimagou E, Sobngwi E, Matsha TE, et al. Diabetes mellitus and inflammation. Curr Diab Rep 2013;13:435-44. [Crossref] [PubMed]
- Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 2005;54:1615-25. [Crossref] [PubMed]
- Bhagat N, Grigorian RA, Tutela A, et al. Diabetic macular edema: pathogenesis and treatment. Surv Ophthalmol 2009;54:1-32. [Crossref] [PubMed]
- Zhang X, Zeng H, Bao S, et al. Diabetic macular edema: new concepts in patho-physiology and treatment. Cell Biosci 2014;4:27. [Crossref] [PubMed]
- Rübsam A, Parikh S, Fort PE. Role of Inflammation in Diabetic Retinopathy. Int J Mol Sci 2018;19:942. [Crossref] [PubMed]
- Rangasamy S, McGuire PG, Das A. Diabetic retinopathy and inflammation: novel therapeutic targets. Middle East Afr J Ophthalmol 2012;19:52. [Crossref] [PubMed]
- Grigsby JG, Cardona SM, Pouw CE, et al. The role of microglia in diabetic retinopathy. J Ophthalmol 2014;2014:705783 [Crossref] [PubMed]
- Limb GA, Hickman-Casey J, Hollifield RD, et al. Vascular adhesion molecules in vitreous from eyes with proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci 1999;40:2453-7. [PubMed]
- Shram MT, Chaturvedi N, Schalkwijk C, et al. Vascular risk factors and markers of endothelial function as determinants of inflammatory markers in type 1 diabetes; the EURODIAB Prospective Complications Study. Diabetes Care 2003;26:2165-73. [Crossref] [PubMed]
- Miyamoto K, Hiroshiba N, Tsujikawa A, et al. In vivo demonstration of increased leukocyte entrapment in retinal microcirculation of diabetic rats. Invest Ophthalmol Vis Sci 1998;39:2190-4. [PubMed]
- Miyamoto K, Khosrof S, Bursell SE, et al. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc Natl Acad Sci USA 1999;96:10836-41. [Crossref] [PubMed]
- Barouch FC, Miyamoto K, Allport JR, et al. Integrin-mediated neutrophil adhesion and retinal leukostasis in diabetes. Invest Ophthalmol Vis Sci 2000;41:1153-8. [PubMed]
- Ai H, Song HP. Different expression pattern of serum soluble intercellular adhesion molecules-1 and neutrophilic expression of CD18 in patients with diabetic retinopathy. Int J Ophthalmol 2012;5:202-7. [PubMed]
- Barouch FC, Miyamoto K, Allport JR, et al. Integrin-mediated neutrophil adhesion and retinal leukostasis in diabetes. Invest Ophthalmol Vis Sci 2000;41:1153-8. [PubMed]
- Spijkerman AM, Gall MA, Tarnow L, et al. Endothelial dysfunction and low-grade inflammation and the progression of retinopathy in Type 2 diabetes. Diabet Med 2007;24:969-76. [Crossref] [PubMed]
- Abcouwer SF. Muller cell-microglia cross talk drives neuroinflammation in diabetic retinopathy. Diabetes 2017;66:261-3. [Crossref] [PubMed]
- Arroba AI, Valverde AM. Modulation of microglia in the retina: new insights into diabetic retinopathy. Acta Diabetol 2017;54:527-33. [Crossref] [PubMed]
- Altmann C, Schmidt MHH. The role of microglia in diabetic retinopathy: inflammation, microvasculature defects and neurodegeneration. Int J Mol Sci 2018;19:E110 [Crossref] [PubMed]
- Hanisch UK, Kettenmann H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 2007;10:1387-94. [Crossref] [PubMed]
- Vujosevic S, Bini S, Midena G, et al. Hyperreflective intraretinal spots in diabetics without and with non proliferative diabetic retinopathy: an in vivo study using spectral domain OCT. J Diabetes Res 2013;2013:491835 [Crossref] [PubMed]
- Zeng HY, Green WR, Tso MO. Microglial activation in human diabetic retinopathy. Arch Ophthalmol 2008;126:227-32. [Crossref] [PubMed]
- Vujosevic S, Bini S, Torresin T, et al. Hyperreflective retinal spots in normal and diabetic eyes: b-scan and en face spectral domain optical coherence tomography evaluation Retina 2017;37:1092-103. [Crossref] [PubMed]
- Langmann T. Microglia activation in retinal degeneration. J Leukoc Biol 2007;81:1345-51. [Crossref] [PubMed]
- Sorrentino FS, Allkabes M, Salsini G, et al. The importance of glial cells in the homeostasis of the retinal microenvironment and their pivotal role in the course of diabetic retinopathy. Life Sci 2016;162:54-9. [Crossref] [PubMed]
- Barber AJ, Antonetti DA, Gardner TW. Altered expression of retinal occludin and glial fibrillary acidic protein in experimental diabetes. Invest Ophthalmol Vis Sci. 2000;41:3561-8. [PubMed]
- Mizutani M, Gerhardinger C, Lorenzi M. Müller cell changes in human diabetic retinopathy. Diabetes 1998;47:445-9. [Crossref] [PubMed]
- El-Asrar AMA, Dralands L, Missotten L, et al. Expression of apoptosis markers in the retinas of human subjects with diabetes. Invest Ophthalmol Vis Sci 2004;45:2760-6. [Crossref] [PubMed]
- Vujosevic S, Simó R. Local and systemic inflammatory biomarkers of diabetic retinopathy: an integrative approach. Invest Ophthalmol Vis Sci 2017;58:BIO68-BIO75. [Crossref] [PubMed]
- Sang DN, D’Amore PA. Is blockade of vascular endothelial growth factor beneficial for all types of diabetic retinopathy? Diabetologia 2008;51:1570-3. [Crossref] [PubMed]
- Gonzalez VH, Campbell J, Holekamp NM, et al. Early and long-term responses to anti-vascular endothelial growth factor therapy in diabetic macular edema: Analysis of protocol I data. Am J Ophthalmol 2016;172:72-9. [Crossref] [PubMed]
- Jenkins AJ, Joglekar MV, Hardikar AA, et al. Biomarkers in diabetic retinopathy. Rev Diabet Stud 2015;12:159. [Crossref] [PubMed]
- Koleva-Georgieva DN, Sivkova NP, Terzieva D. Serum inflammatory cytokines IL-1beta, IL-6, TNF-alpha and VEGF have influence on the development of diabetic retinopathy. Folia Med (Plovdiv) 2011;53:44-50. [Crossref] [PubMed]
- Boss JD, Singh PK, Pandya HK, et al. Assessment of neurotrophins and inflammatory mediators in vitreous of patients with diabetic retinopathy. Invest Ophthalmol Vis Sci 2017;58:5594-603. [Crossref] [PubMed]
- Kuo JZ, Guo X, Klein R, et al. Systemic soluble tumor necrosis factor receptors 1 and 2 are associated with severity of diabetic retinopathy in Hispanics. Ophthalmology 2012;119:1041-6. [Crossref] [PubMed]
- Kaul K, Hodgkinson A, Tarr JM, et al. Is inflammation a common retinal-renal-nerve pathogenic link in diabetes? Curr Diabetes Rev 2010;6:294-303. [Crossref] [PubMed]
- Gustavsson C, Agardh E, Bengtsson B, et al. TNF-alpha is an independent serum marker for proliferative retinopathy in type 1 diabetic patients. J Diabetes Complications 2008;22:309-16. [Crossref] [PubMed]
- Myśliwiec M, Balcerska A, Zorena K, et al. The assessment of the correlation between vascular endothelial growth factor (VEGF), tumor necrosis factor (TNF-alpha), interleukin 6 (IL-6), glycemic control (HbA1c) and the development of the diabetic retinopathy in children with diabetes mellitus type 1. Klin Oczna 2007;109:150-4. [PubMed]
- Woo SJ, Ahn SJ, Ahn J, et al. Elevated systemic neutrophil count in diabetic retinopathy and diabetes: a hospital-based cross-sectional study of 30.793 Korean subjects. Invest Ophthalmol Vis Sci 2011;52:7697-703. [Crossref] [PubMed]
- Petrovič MG, Korosec P, Kosnik M, et al. Association of preoperative vitreous IL-8 and VEGF levels with visual acuity after vitrectomy in proliferative diabetic retinopathy. Acta Ophthalmol 2010;88:e311-6. [Crossref] [PubMed]
- Yoshimura T, Sonoda KH, Sugahara M, et al. Comprehensive analysis of inflammatory immune mediators in vitreoretinal diseases. PLoS One 2009;4:e8158 [Crossref] [PubMed]
- Oh IK, Kim SW, Oh J, et al. Inflammatory and angiogenic factors in the aqueous humor and the relationship to diabetic retinopathy. Curr Eye Res 2010;35:1116-27. [Crossref] [PubMed]
- Dong N, Xu B, Chu L, et al. Study of 27 Aqueous Humor Cytokines in Type 2 Diabetic Patients with or without Macular Edema. PLoS One 2015;10:e0125329 [Crossref] [PubMed]
- Dong N, Xu B, Wang B, et al. Study of 27 aqueous humor cytokines in patients with type 2 diabetes with or without retinopathy. Mol Vis 2013;19:1734-46. [PubMed]
- Figueras-Roca M, Molins B. Peripheral blood metabolic and inflammatory factors as biomarkers to ocular findings in diabetic macular edema. PLoS One 2017;12:e0173865 [Crossref] [PubMed]
- Dong N, Chang L, Wang B, et al. Retinal Neuronal Mcp-1 Induced by Ages Stimulates TNFα Expression in Rat Microglia Via P38, Erk, and Nf-κB Pathways. Mol Vis 2014;20:616-28. [PubMed]
- Vujosevic S, Casciano M, Pilotto E, et al. Diabetic macular edema: fundus autofluorescence and functional correlations. Invest Ophthalmol Vis Sci 2011;52:442-8. [Crossref] [PubMed]
- Vujosevic S, Berton M, Bini S, et al. Hyperreflective retinal spots and visual function after anti-vascular endothelial growth factor treatment in center-involving diabetic macular edema. Retina 2016;36:1298-308. [Crossref] [PubMed]
- Vujosevic S, Torresin T, Bini S, et al. Imaging retinal inflammatory biomarkers after intravitreal steroid and anti‐VEGF treatment in diabetic macular oedema. Acta Ophthalmol 2017;95:464-71. [Crossref] [PubMed]
- Sonoda S, Sakamoto T, Yamashita T, et al. Retinal morphologic changes and concentrations of cytokines in eyes with diabetic macular edema. Retina 2014;34:741-8. [Crossref] [PubMed]
- Madeira MH, Boia R, Santos PF, et al. Contribution of microglia-mediated neuroinflammation to retinal degenerative diseases. Mediators Inflamm 2015;2015:673090 [Crossref] [PubMed]
- Mizukami T, Hotta Y, Katai N. Higher numbers of hyperreflective foci seen in the vitreous on spectral-domain optical coherence tomographic images in eyes with more severe diabetic retinopathy. Ophthalmologica 2017;238:74-80. [Crossref] [PubMed]
- Korot E, Comer G, Steffens T, et al. Algorithm for the measure of vitreous hyperreflective foci in optical coherence tomographic scans of patients with diabetic macular edema. JAMA Ophthalmol 2016;134:15-20. [Crossref] [PubMed]
- Urbančič M, Kloboves Prevodnik V, Petrovič D, et al. A flow cytometric analysis of vitreous inflammatory cells in patients with proliferative diabetic retinopathy. Biomed Res Int 2013;2013:251528 [Crossref] [PubMed]
- Cantón A, Martinez-Cáceres EM, Hernández C, et al. CD4-CD8 and CD28 expression in t cells infiltrating the vitreous fluidin patients with proliferative diabetic retinopathy: A flow cytometric analysis. Arch Ophthalmol 2004;122:743-9. [Crossref] [PubMed]
- Otani T, Kishi S. Tomographic assessment of vitreous surgery for diabetic macular edema. Am J Ophthalmol 2000;129:487-94. [Crossref] [PubMed]
- Seo KH, Yu SY, Kim M, et al. Visual and morphologic outcomes of intravitreal ranibizumab for diabetic macular edema based on optical coherence tomography patterns. Retina 2016;36:588-95. [Crossref] [PubMed]
- Campos A, Campos EJ, Martins J, et al. Viewing the choroid: where we stand, challenges and contradictions in diabetic retinopathy and diabetic macular oedema. Acta ophthalmol 2017;95:446-59. [Crossref] [PubMed]
- Vujosevic S, Torresin T, Berton M, et al. Diabetic macular edema with and without subfoveal neuroretinal detachment: two different morphologic and functional entities. Am J Ophthalmol 2017;181:149-55. [Crossref] [PubMed]
- Uji A, Murakami T, Nishijima K, et al. Association between hyperreflective foci in the outer retina, status of photoreceptor layer, and visual acuity in diabetic macular edema. Am J Ophthalmol 2012;153:710-717, 717.e1.
- Framme C, Schweizer P, Imesch M, et al. Behavior of SDOCT-detected hyperreflective foci in the retina of antiVEGF-treated patients with diabetic macular edema. Invest Ophthalmol Vis Sci 2012;53:5814-8. [Crossref] [PubMed]
- Chen KC, Jung JJ, Curcio CA, et al. Intraretinal hyperreflective foci in acquired vitelliform lesions of the macula: clinical and histologic study. Am J Ophthalmol 2016;164:89-98. [Crossref] [PubMed]
- Lee H, Jang H, Choi YA, et al. Association Between Soluble CD14 in the Aqueous Humor and Hyperreflective Foci on Optical Coherence Tomography in Patients With Diabetic Macular Edema. Invest Ophthalmol Vis Sci 2018;59:715-21. [Crossref] [PubMed]
- Pece A, Isola V, Holz F, et al. Autofluorescence imaging of cystoid macular edema in diabetic retinopathy. Ophthalmologica 2010;224:230-5. [Crossref] [PubMed]
- Schmitz-Valckenberg S, Holz FG, Bird AC, Spaide RF. Fundus autofluorescence imaging: and perspectives. Retina 2008;28:385-409. [Crossref] [PubMed]
- Otani T, Kishi S, Maruyama Y. Patterns of diabetic macular edema with optical coherence tomography. Am J Ophthalmol 1999;127:688-93. [Crossref] [PubMed]
- Gaucher D, Sebah C, Erginay A, et al. Optical coherence tomography features during the evolution of serous retinal detachment in patients with diabetic macular edema. Am J Ophthalmol 2008;145:289-96. [Crossref] [PubMed]
- Catier A, Tadayoni R, Paques M, et al. Characterization of macular edema from various etiologies by optical coherence tomography. Am J Ophthalmol 2005;140:200-6. [Crossref] [PubMed]
- Ozdemir H, Karacorlu M, Karacorlu S. Serous macular detachment in diabetic cystoid macular oedema. Acta Ophthalmol Scand 2005;83:63-6. [Crossref] [PubMed]
- Coscas G, De Benedetto U, Coscas F, et al. Hyperreflective dots: a new spectral-domain optical coherence tomography entity for follow-up and prognosis in exudative age-related macular degeneration. Ophthalmologica 2013;229:32-7. [Crossref] [PubMed]
- Bolz M, Schmidt-Erfurth U, Deak G, et al. Optical coherence tomographic hyperreflective foci: a morphologic sign of lipid extravasation in diabetic macular edema. Ophthalmology 2009;116:914-20. [Crossref] [PubMed]
- Ogino K, Murakami T, Tsujikawa A, et al. Characteristics of optical coherence tomographic hyperreflective foci in retinal vein occlusion. Retina 2012;32:77-85. [Crossref] [PubMed]
- Deák GG, Bolz M, Kriechbaum K, et al. Diabetic Retinopathy Research Group Vienna. Effect of retinal photocoagulation on intraretinal lipid exudates in diabetic macular edema documented by optical coherence tomography. Ophthalmology 2010;117:773-9. [Crossref] [PubMed]
- De Benedetto U, Sacconi R, Pierro L, et al. Optical coherence tomographic hyperreflective foci in early stages of diabetic retinopathy. Retina 2015;35:449-53. [Crossref] [PubMed]
- Berasategui B, Fonollosa A, Artaraz J, et al. Behavior of hyperreflective foci in non-infectious uveitic macular edema, a 12-month follow-up prospective study. BMC Ophthalmology 2018;18:179. [Crossref] [PubMed]
- Zanzottera EC, Messinger JD, Ach T, et al. The Project macula retinal pigment epithelium grading system for histology and optical coherence tomography in age-related macular degeneration. Invest Ophthalmol Vis Sci 2015;56:3253-68. [Crossref] [PubMed]
- Pang CE, Messinger JD, Zanzottera EC, et al. The onion sign in neovascular age-related macular degeneration represents cholesterol crystals. Ophthalmology 2015;122:2316-26. [Crossref] [PubMed]
- Xu H, Chen M, Manivannan A, et al. Age-dependent accumulation of lipofuscin in perivascular and subretinal microglia in experimental mice. Aging Cell 2008;7:58-68. [Crossref] [PubMed]
- Simó R, Hernández C. Novel approaches for treating diabetic retinopathy based on recent pathogenic evidence. Prog Retin Eye Res 2015;48:160-80. [Crossref] [PubMed]
- Stitt AW, Curtis TM, Chen M, et al. The progress in understanding and treatment of diabetic retinopathy. Prog Retin Eye Res 2016;51:156-86. [Crossref] [PubMed]
- Tan GS, Cheung N, Simó R, et al. Diabetic macular oedema. Lancet Diabetes Endocrinol 2017;5:143-55. [Crossref] [PubMed]
- Wong TY, Cheung CM, Larsen M, et al. Diabetic retinopathy. Nat Rev Dis Primers 2016;2:16012. [Crossref] [PubMed]
Cite this article as: Vujosevic S, Toma C. Diabetic retinopathy: an inflammatory disease. Ann Eye Sci 2018;3:52.


