
Evaluating visual outcomes using optical coherence tomography (OCT) in pediatric multiple sclerosis and other neuroinflammatory conditions
Background
While the use of optical coherence tomography (OCT) to generate cross sectional images of the retina was first reported in 1991, the first reports of OCT use in pediatric populations only emerged over a decade later (1). The delay in adoption of this technology in pediatrics stemmed primarily from challenges with cooperation leading to difficulty in achieving appropriate visual fixation in children, and differences between adults and children/infants in eye shape and refraction (2-4). Importantly, challenges with fixation and cooperation were addressed by the introduction, in the mid-to-late 2000s, of high speed spectral-domain OCT (SD-OCT) and the subsequent development of hand-held OCT (HH-OCT), which led to increased access to this technology for pediatric populations (5-8).
Since then, OCT-related knowledge in pediatric populations has increased rapidly, as has knowledge related to pediatric specific norms. OCT studies of visual pathway disorders of childhood have included investigations of retinopathy of prematurity (ROP) (9), diagnosis and monitoring of retinal dystrophies, retinal dysplasia and infantile nystagmus (10,11), ocular tumors (12,13), and optic nerve pathologies (14). OCT measures have also been used as surrogate markers for systemic disease in children, including chronic kidney disease (15) and congenital adrenal hyperplasia (16). The technology has been used widely to evaluate and follow idiopathic intracranial hypertension (IIH) manifestations in children (14,17).
Importantly, this technology has allowed for widespread access to minimally invasive standardized, reproducible quantified structural assessments of the optic nerve and retina. This has had important implications in pediatric optic neuropathies, patients in whom monitoring of disease activity is essential to make treatment decisions. OCT has had particular relevance for inflammatory optic neuropathies, as onset of an inflammatory optic neuropathy may herald the onset of a chronic inflammatory disorder of the central nervous system (CNS), such as multiple sclerosis (MS) and myelin oligodendroglycoprotein (MOG) or aquaporin 4 (AQP4) antibody associated neuromyelitis optica spectrum disorders (NMOSD). Specifically, diagnosis and follow up of these CNS inflammatory disorders can be aided by this tool. After reviewing pediatric-specific anatomic and practical issues pertinent to OCT, we will review knowledge related to the use of OCT in inflammatory pediatric optic neuropathies. As our focus will be exclusively on inflammatory optic neuropathies, we refer readers to other excellent publications reviewing its use for non-inflammatory optic neuropathies, retinal and other pediatric indications for information on other uses of OCT in children (18-23).
Pediatric specific OCT issues
Anatomical and developmental issues
The pediatric eye is distinguished from the adult eye by developmental changes that occur with age. These differences must be accounted for when interpreting results of OCT imaging of the eye. Gordon et al. (2) demonstrated that infants up to 6 months of age have greater corneal curvature than older age groups, and that between infancy and childhood, the axial length of the globe increases in a non-linear fashion by approximately 7 mm. The greatest increase occurs in the first 2–3 years of life, growing at a rate of about 1 mm/year. This is followed by the relatively slower pace of 0.4 mm/year until 5–6 years of age, and by about 0.2 mm/year up until age 10, beyond which no significant changes are noted.
Given the knowledge about the relationship between axial length and OCT measures, it follows that changes in axial length through childhood may affect retinal measurements using OCT. In studies of healthy adults, axial length showed a significant inverse correlation with retinal nerve fiber layer (RNFL) (24,25). Using time-domain OCT (TD-OCT) Budenz et al. (24,25) demonstrated that RNFL thickness decreases with increasing age as well as with increasing axial length in healthy adults between 18–85 years of age. The authors showed that a 1-mm increase in axial length corresponded to a 2.2-micron (95% CI, 1.1–3.4 micron) decrease in RNFL thickness. This finding was later reproduced by Bendschneider et al. (24) using SD-OCT, who demonstrated a 4.79-micron decrease in RNFL thickness with every 1-mm increase in axial length. Furthermore, these authors found that each decade increase in age correlated with a 2 micron (95% CI, 1.2–2.8) decrease in RNFL thickness (24,25). However, studies evaluating the relationship between axial length and retinal nerve fiber layer thickness (RNFLT) in pediatric populations have shown inconsistent results. Pan et al. (26) showed an inverse correlation between axial length and RNFL thickness in multivariate analysis (n=1,063), while Matalia et al. (27) found that only foveal thickness increased significantly with age and myopia in children older than 5 years (n=113). This may be due to the differences in sample sizes or differences in methodologies used: while SD-OCT was used by both groups Matalia used RTVue Premier while Pan et al. used IOLMaster (26,27).
Average RNFLT has also been shown to be significantly lower with an accompanying propensity for lower retinal ganglion cell layer (GCL) thickness in obese children compared to a healthy control population (28). Overall these results suggest a correlation between retinal thickness, axial length of the eye, and body mass index, and emphasize the need to consider multiple factors including age, axial length, degree and type of refraction errors and body mass index when interpreting OCT results.
Technical issues
OCT imaging of the pediatric eye using a tabletop machine can be challenging, as it requires patients to sit still, fixate and cooperate with instructions. Due to size considerations, younger children often have difficulty with keeping their heads against the forehead strap and chin rest on the tabletop machine. In addition, fixating on an internal or external target may be difficult for younger children. Early OCT modalities using time-domain (TD) technology entailed sequential scanning that required longer image acquisition times, but the development of SD-OCT in 2003 enabled much faster image acquisition via simultaneous scanning. The signal to noise ratio was improved by several hundred-fold in SD compared to TD (8), resulting in an increase in image acquisition speed from 2 A scans per second with TD to several million A scans per second with SD (29). Faster image acquisition has resulted in less motion artifact and an increase in sensitivity and reproducibility (8,30). The subsequent inclusion of eye-tracking technology improved image registration accuracy (31) with the potential to increase the technique’s use in younger children, in patients with intellectual disabilities, and in patients with nystagmus.
Hand-held SD-OCT
The table-top OCT design has many limitations for younger and disabled patients, particularly those who cannot sit in an upright position or cannot cooperate with the examination for other reasons. HH-OCT addresses these limitations (Figure 1). In the mid-to-late 2000s, several reports described modifications to table-top machines in response to these limitations and provided proof of principle that OCT images could be acquired using a portable, hand-held machine (32-35). In one case, to address an unmet need to identify progression to retinal detachment in ROP despite treatment, Patel attempted to use a portable OCT in an infant to stage the child’s ROP (32). In another instance, in 2005, Joshi and colleagues (33) modified a table-top OCT machine, placing it next to patients on the operating table with the goal of identifying subclinical changes predictive of long-term visual outcomes after vitrectomy in stage 4a ROP. In 2008, Gerth et al. (34) used SD-OCT with its sample arm replaced by a hand-held scanner developed by Bioptigen (Bioptigen Inc., Durham, NC, USA) and used this for the first time in patients with Bardet-Biedl Syndrome (BBS) to characterize differences in retinal pathology in patients with two different underlying mutations. In addition to demonstrating that the retinal pathology did not differ with the underlying genetic mutations in BBS patients, this study also showed that images of adequate quality could be obtained with HH-OCT despite poor cooperation or presence of nystagmus (34).
HH-OCT was later tested in a cohort of younger children aged 7 months to 9.9 years (n=30), of whom 10 were under 3.7 years of age. Sedation was needed only in the 10 younger patients (5). The authors were able to localize the macula via screening of the retina using the 6×6 mm volumetric scan mode on OCT, and found that macular localization was easier in conscious children than sedated children. This was explained by the absence of simultaneous fundus photography, i.e., the use of scanning light only, which created a distraction in sedated children (5). HH-OCT has been studied in infants with Shaken Baby syndrome (35,36), ROP (9,37), nystagmus (11), in children with optic pathway gliomas (38,39) and healthy children (40-42). Overall, spectral-domain HH-OCT has proven very useful in resolving technical issues that limit pediatric use, therefore allowing for in vivo and awake imaging of younger children.
OCT: normative data in pediatric patients
Given the anatomic and developmental differences noted above, it is clear that separate norms that take account of developmental trajectories in children must accompany use of OCT in pediatrics. Average RNFLT is likely higher in youth than adults. Yanni et al. (43) described retinal layer measurements in children using the Spectralis SD-OCT in a North American population of healthy children (n=83). This group found average RNFLT to be 107.6 microns [standard error of the mean (SEM) 1.2], total retinal foveal thickness to be 219.6 microns (SEM 1.7), and central macular subfield to be 271.2 microns (SEM 2.0) for children between 5–15 years. RNFLT did not differ based on age, sex or ethnicity in this cohort (43). These results were comparable to earlier reports in healthy Turkish and Taiwanese children (44,45), and to results from a healthy comparison cohort in a pediatric demyelinating disorders study (46). Importantly, these values were higher than the RNFL values reported in healthy adults (97.2±9.7 microns) using the same modality (24). The differences between the adult and pediatric studies may represent age-related decrease in RNFLT shown in adults: researchers have demonstrated RNFLT decreases of 1.90 microns per decade in an adult cohort of 170 healthy individuals aged between 20 to 78 years (24). However, it is not known when age-related decline in RNFLT actually starts as studies including healthy individuals across the continuum of age have not been published.
Evaluation of RNLFT by quadrant in children demonstrates patterns similar to the double-hump pattern (greater RNFLT in the superior and inferior quadrants followed by nasal and temporal quadrants) reported in adult cohorts (24,25,47). Three studies showed this pattern in children, with lower RNFLT in the temporal (range, 74–76 microns) and nasal quadrants (range, 71–85 microns) compared to the temporal superior (range, 138–145 microns), temporal inferior (range, 144–147 microns), nasal superior (range, 102–116 microns), and nasal inferior quadrants (range, 106–125 microns) (24,25,47). Matalia et al. (27) showed that the same pattern could be observed also in mean total retinal thickness (temporal 254±4.03 microns, nasal 264±4.04 microns, superior 268±2.26 microns, inferior 287±4.37 microns) and choroid thickness (temporal 307±4.56 microns, nasal 286±3.51 microns, superior 319±4.68 microns, inferior 320±4.74 microns).
On the other hand, in children, macular thickness shows dynamic changes through time, including significant linear increases until age 15 and reductions subsequent to this (43,48). Yanni et al. (43) demonstrated that central macular thickness measured 10 microns greater in children aged between 11–15 years compared children aged between 5–7 years (P=0.032). In another study which included older children, Matalia et al. (27) grouped healthy children in three age groups as 5–11 years (number of eyes 44), 12–14 years (number of eyes 36) and 15–17 years (number of eyes 33) and found average foveal thickness to be 209.82, 207.94 and 202.95 microns, respectively. In addition, macular thickness has been reported to decrease with increasing age in adults aged between 17–83 years (r=−0.227, P=0.002) (49). Central macular thickness may be slightly greater in boys compared to girls, although the difference has not persistently been shown to be significant across studies (43,48,50).
Studies published thus far have shown no gender differences in peripapillary RNFL in children (43). Ethnic background/race may have relevance to RNFL: in one study, non-Hispanic white children were found to have lower (105.3±1.4 microns) RNFL thickness compared to children of other backgrounds including Black, Hispanic, Asian and Mixed (112.9±2.0 microns) (43).
Applications of OCT to optic nerve pathology in neuroinflammatory conditions
Differentiation of inflammatory optic neuropathies from traumatic and other optic neuropathies using OCT
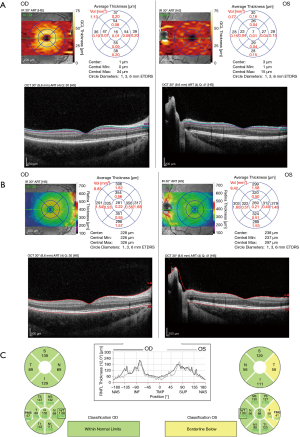
OCT is now widely used in pediatric populations to evaluate optic nerve pathology and may allow for the differentiation of inflammatory from other etiologies of optic neuropathy. Traumatic optic neuropathy, for example, may mimic inflammatory optic neuritis (ON) clinically with a positive relative afferent pupillary defect (RAPD), impairment of color vision and visual field defects (51-53), but history should distinguish the two if direct trauma is involved. However, in some cases indirect traumatic optic neuropathy may result from a minor, seemingly inconsequential blow, and, as such, differentiating the two may be difficult (Figure 2). OCT can help differentiate the two in some cases by showing posterior vitreous separation in traumatic optic neuropathies (36). The most distinct finding early after indirect traumatic optic neuropathy, however, is reduction in ganglion cell inner plexiform layer (GCIPL) thickness measurements (54).
Optic neuropathies related to inflammatory disorders of the CNS
Acquired demyelinating syndromes (ADS) of the CNS occur in approximately 1/100,000 people, of whom about 1/5 will eventually have the diagnosis of MS. Over 1/3 of youth with ADS will experience ON, and evidence of optic nerve involvement may occur in almost all youth with MS. OCT technology has led to marked increases in knowledge around visual pathway involvement in these children.
Acute ON
Acute inflammatory ON can be seen as an isolated phenomenon or in relation to known demyelinating syndromes including MS, anti-MOG associated demyelination, or NMOSD with or without AQP4 antibodies. The incidence of acute ON in the pediatric population is 1–3 per 100,000 population (55,56). ON presents with painful eye movements with or without accompanying headache, blurry vision with one or both of the eyes, and/or visual field deficits. Examination reveals varying degrees of functional impairment including loss of visual acuity (VA), color desaturation, and/or central scotomas, with visual loss occurring over hours to days with a nadir within days of onset. ON is monophasic in nearly 60% of children (57). Notably, pediatric ON is characterized by a high rate of bilateral involvement in youth under 10 (72%), with unilateral presentation predominating on youth over 10 years of age (58).
OCT studies in the acute phase of pediatric ON have demonstrated significant differences in RNFL and GCIPL thicknesses in patients in comparison to healthy subjects. In one longitudinal study of acute ON which included adults and pediatric patients, GCIPL thickness was found to decline rapidly from a baseline of 80.4±4.9 microns within 1 month (69±7.3 microns), stabilizing 2 months later (63.6±8.7 microns) (59). VA reached a nadir at baseline (−1.29±0.96 LogMAR) and improved gradually to reach a plateau (−0.18±0.19) at 2 months (59). These findings suggest a rapid and permanent loss of ganglion cells in the acute phase correlating in severity with the speed of decline. However, this study had important limitations including a mixed cohort of pediatric and adult patients (ages 8−78 years), irregular timing of follow up assessments, lack of low-contrast letter acuity (LCLA) assessments that have been shown to better capture visual impairment in pediatric ON (60), and a lack of RNFL measurements preventing the authors from commenting on the effect of acute inflammatory edema on GCIPL.
GCL thinning in affected and fellow eyes in the acute phase of ON has been shown in another study: Wilbur et al. (61) described GCL/inner plexiform layer (IPL) thinning in a cohort which included children with monophasic acute demyelinating disorders and MS. This study was limited, however, by heterogeneity of the monophasic ON group and absence of longitudinal follow-up.
OCT outside of the acute period in chronic demyelinating conditions: MS, NMOSD, MOG related disease
Mixed populations: youth with demyelination
In a cross-sectional study of a pediatric demyelinating cohort which comprised of MS (40%), acute disseminated encephalomyelitis (ADEM), ON, non-ON demyelination patients and healthy children, Yeh et al. (62) showed that both RNFL and GCIPL thicknesses were decreased in youth with demyelination compared to healthy youth. Mean RNFLT was 76.4 microns (3.67) in compared to 102.4 microns (2.11) in controls (P<0.0001). Mean GCL was 68.0 microns (2.21) in the demyelinating group compared to 84.8 microns (1.18) in the control group (P<0.0001). Significant differences in RNFL in groups with 0, 1 and >1 episode of ON were present, but these dramatic decreases were not seen in the GCIPL (62).
This group also demonstrated that RNFL measurements differed between different demyelinating subtypes (46). Average global RNFLT was 89±12 microns in the isolated-ON group, 83±12 microns in the MS group, 67±17 microns in ADEM/TM group and 50±2 microns in chronic relapsing inflammatory optic neuropathy (CRION) group, suggesting varying degrees of axonal injury in different disease phenotypes. However, this study was limited by absence of control for the number of relapses and small cohort size. There were only two patients in the CRION group which had a relapse rate of 100% while the relapse rate was only 20% in ADEM/transverse myelitis (TM) group.
Pediatric onset MS
Pediatric MS is a demyelinating disorder of the CNS which comprises up to 10% of all cases of MS, with a relapsing course in almost all children (63,64). Up to 40% of children presenting with an initial episode of ON eventually develop MS (65-67). As noted above, OCT studies have demonstrated distinctive patterns of RNFL and GCIPL thinning in youth with demyelination (46,61,62). Similar to adults with MS, pediatric onset MS patients have lower RNFLT compared to healthy controls. Average global RNFLT ranges between 82–103 microns, and average temporal RNFLT ranges between 58–60 in pediatric MS cohorts (46,60,68,69). Both RNFLT and GCIPL are lower in MS patients with history of clinical episodes of ON (RNFLT range, 75–86 microns, GCIPL 68.4 microns) and in patients with no clinical history of ON (RNFLT range, 95–109 microns, GCIPL 83.4 microns) compared to healthy controls (RNFLT range, 109–98 microns, GCIPL 90 microns) (46,60,68-70). Only one study has reported absence of RNFL thinning in children with MS with no history of ON (60). In addition to RNFLT {109 [11] vs. 109 [9] microns in ON-negative MS and healthy control eyes, respectively}, functional visual metrics including high-contrast letter acuity (HCLA) {62 [6] vs. 64 [4] in ON-negative MS and healthy control eyes, respectively), LCVA {31 [8] vs. 33 [8] in ON-negative MS and healthy control eyes, respectively}, and Pelli-Robson log score {1.66 [0.09] vs. 1.64 [0.06] in ON-negative MS and healthy control eyes, respectively} were found to be similar between ON-negative MS patients and healthy controls. RNFL thinning correlated with 1.25% LCLA measurements in patients with history of ON demonstrating persistent functional abnormalities after ON in MS (60).
Sex differences in RNFL thickness may exist in youth with MS. In a pediatric MS cohort of 53 patients, Graves et al. (69), demonstrated that boys had a 9.4 microns lower global RNFLT in comparison to girls (P=0.022), while there were no sex-related differences in GCIPL. Whether this finding is due to greater retinal axonal injury in boys and better recovery in girls, or larger macular volumes in boys which compensate for the GCIPL measurements remains to be addressed in prospective longitudinal studies.
Investigation of macular volume in pediatric MS cohorts showed that macular volume is lower in the affected as well as the unaffected eyes of pediatric MS patients (46,68). This finding was also seen in children with other forms of CNS demyelination including ADEM/TM and CRION with lower values in their affected eyes. Yilmaz et al. (68), noted that macular volume was lower at 6 months (6.30 mm3) and more so at 12 months (6.09 mm3) of follow-up compared to baseline (6.46 mm3) in pediatric MS patients and healthy controls (6.91 mm3) (68).
In the cross-sectional study noted above, Graves et al. (69) studied pediatric MS patients (n=53) in comparison to healthy controls (n=19) and found that lower total macular volume and RNFLT were associated with longer disease duration. Furthermore, this group found RNFLT in MS patients with no clinical ON events to be significantly different from a healthy control population, suggesting subclinical degeneration in afferent visual pathways (46).
Other studies have shown reduction in GCIPL beyond 6 months of onset in MS, suggesting progressive ganglion cell loss in this population (62,69). Structural OCT measurements have also been investigated in relation to cognitive impairment and markers of subclinical disease progression in MS in various reports. In a pediatric cohort of 20 pediatric MS patients and 22 healthy controls, Datta et al. (71) found no relationship between RNFL and GCIPL thickness and global and visual cortex cortical mantle thickness using 3T MRI.
NMOSD/AQP-4 disease
NMOSD is a clinical syndrome characterized by optic nerve and spinal cord involvement, which is associated with antibodies to AQP4. ON is seen in about 80% of children with AQP4+ NMOSD and is the incident neuroinflammatory episode in 50–75% (72,73). Studies in adults with NMOSD have demonstrated more severe GCL and RNFL thinning in a global rather than temporal pattern in NMOSD compared to MS patients (74,75). In pediatrics, OCT comparisons have been limited to antibody positive and seronegative ON. Global RNFLT ranged between 64–89 microns in AQP4 positive children (70,76). Chen et al. (76) compared the clinical characteristics in MOG-positive, AQP4-positive and seronegative Asian children with NMOSD and found that the severity of axonal degeneration evidenced by RNFLT was similar in MOG (58.03±8.73 microns) and AQP4-positive patients (64.34±12.88 microns) and worse than in seronegative patients (78.12±13.34 microns). However, the groups were not balanced in terms of the number of clinical relapses; the seronegative group had a lower number of relapses compared to the other groups, potentially confounding the results. In a better-balanced cohort in terms of relapse rate Narayan et al. (77) found that median global RNFLT was 89 microns (range, 26–120 microns; IQR, 40 microns) in AQP4-positive children compared to 58 microns (range, 34–97 microns; IQR, 23 microns) in MOG-positive and 82.5 microns (range, 32–116 microns; IQR, 36 microns) in seronegative children. Similar to the other studies, Narayan et al. included only a limited number of AQP4+ patients (n=9) all with history of ON.
MOG-related disease
More recently, the NMOSD phenotype has been described in association with antibodies to MOG. The role of MOG in CNS demyelination has been studied extensively in the context of experimental autoimmune encephalomyelitis (EAE) models, but it was not until recently that anti-MOG antibodies were accepted to be associated with clinical disease. ON associated with MOG antibodies has been reported in 27–47% of children with acute ON (57,78). MOG-ON presents in younger children and is often bilateral. MRI of the brain frequently shows multifocal cortical and white matter lesions. Patients frequently respond rapidly to pulse steroid treatment with nearly complete functional recovery (76,78,79).
In a retrospective study of 36 pediatric NMOSD patients Narayan et al. (77) identified MOG antibodies in 19 patients, of whom 14 had at least one episode of ON. Among these, the median RNFLT was 58 microns (range, 34–97 microns; IQR, 23 microns) in the affected eyes of MOG positive patients (number of eyes 23). RNFLT was lower in the eyes with no clinical events in MOG-positive patients (number of eyes 4) compared to the AQP4-positive and seronegative patients, suggesting subclinical involvement in these patients (77). However, this study was limited by both its cross-sectional design and the very low number of unaffected eyes studied. The authors also demonstrated that despite greater axonal injury, MOG positive patients had better functional outcomes. Sub-group analysis by RNFLT ranges demonstrated that in the lowest RNFL ranges (<50 microns) MOG-positive patients had significantly better VA on 2.5% LCLA testing compared to the other patient groups (77).
In a similar study Eyre et al. (70) found that median RNFLT was 73 microns (54.1–84.2 microns) in the affected eyes of MOG-antibody positive children (number of eyes 29), while it was 78 microns (68.8–85.6 microns) in MS patients. RNFLT in eyes with at least one episode of ON was inversely correlated with the degree of visual impairment evaluated using logMAR high-contrast visual acuity (HCVA); however, overall there was no correlation between the number of ON episodes and the RNFL thickness or the degree of functional impairment (logMAR HCVA). Longitudinal observations were performed in a very small subgroup of nine patients (9 MOG-ON and 6 MS-ON eyes) that showed no significant decline in mean RNFLT over a median interval of 23 months. In particular, change in mean RNFLT from baseline was −4.4 (6.9) microns in ON-positive eyes of MOG positive patients and −0.83 (3) microns in ON-positive eyes of MS patients (70). Based on these, authors suggested that the first ON episode might be responsible for the degree of permanent injury rather than the accumulating effect of subsequent episodes; however, they did not account for the number of relapses in each group. Furthermore, the MOG antibody positive ON and MS groups were unbalanced in terms of the number of relapses in the large group comparisons.
Conclusions and future directions
Knowledge regarding OCT use in youth, and in particular, youth with demyelinating syndromes, has expanded tremendously over the past decade. Work establishing norms in healthy youth suggests that RNFL and GCIPL may be higher in children than adults, but the timing of the onset of physiologic decreases in RNFL and GCIPL through time is unknown. Furthermore, OCT has proven to be of great utility in identifying anterior visual pathway abnormalities and visual outcomes in youth with demyelinating syndromes. MS patients and patients positive for AQP4 antibodies and MOG antibodies have distinct patterns of abnormality acutely and in the chronic phase, both in the presence and absence of clinical episodes of ON. Studies have demonstrated that children with demyelinating disorders, like adults, may experience progressive degeneration of both the RNFL and GCIPL, and that sex differences may exist in these populations. Finally, a number of studies have shown that structural metrics derived from OCT studies correlate with functional outcomes in this population. Further work is needed to evaluate the utility of this tool, in combination with functional studies, for use in future clinical trials and as a means by which to make earlier diagnoses in children with demyelinating disorders.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Fiona Costello and Steffen Hamann) for the series “The Use of OCT as a Biomarker in Neuro-ophthalmology” published in Annals of Eye Science. The article has undergone external peer review.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes.2020.02.03). The series “The Use of OCT as a Biomarker in Neuro-ophthalmology” was commissioned by the editorial office without any funding or sponsorship. BCK is supported by funds from the National MS Society; EAY has received funds from NMSS, CIHI, CIHR, OIRM, MS Society of Canada, Mario Battaglia Foundation, SickKids Foundation, CBMH Innovation Fund, CMSC, Rare Diseases Foundation and Guthy Jackson Foundation. She serves as a relapse adjudicator for ACI. She has served on a scientific advisory panel for Juno Therapeutics and has received a speaker’s honorarium from Novartis. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Meyer CH, Lapolice DJ, Freedman SF. Foveal hypoplasia in oculocutaneous albinism demonstrated by optical coherence tomography. Am J Ophthalmol 2002;133:409-10. [Crossref] [PubMed]
- Gordon RA, Donzis PB. Refractive development of the human eye. Arch Ophthalmol 1985;103:785-9. [Crossref] [PubMed]
- Salchow DJ, Hutcheson KA. Optical coherence tomography applications in pediatric ophthalmology. J Pediatr Ophthalmol Strabismus 2007;44:335-49. [Crossref] [PubMed]
- Skarmoutsos F, Sandhu SS, Voros GM, et al. The use of optical coherence tomography in the management of cystoid macular edema in pediatric uveitis. J AAPOS 2006;10:173-4. [Crossref] [PubMed]
- Gerth C, Zawadzki RJ, Heon E, et al. High-resolution retinal imaging in young children using a handheld scanner and Fourier-domain optical coherence tomography. J AAPOS 2009;13:72-4. [Crossref] [PubMed]
- Wojtkowski M, Bajraszewski T, Targowski P, et al. Real-time in vivo imaging by high-speed spectral optical coherence tomography. Opt Lett 2003;28:1745-7. [Crossref] [PubMed]
- Yun S, Tearney G, Bouma B, et al. High-speed spectral-domain optical coherence tomography at 1.3 mum wavelength. Opt Express 2003;11:3598-604. [Crossref] [PubMed]
- de Boer JF, Cense B, Park BH, et al. Improved signal-to-noise ratio in spectral-domain compared with time-domain optical coherence tomography. Opt Lett 2003;28:2067-9. [Crossref] [PubMed]
- Chavala SH, Farsiu S, Maldonado R, et al. Insights into advanced retinopathy of prematurity using handheld spectral domain optical coherence tomography imaging. Ophthalmology 2009;116:2448-56. [Crossref] [PubMed]
- Chong GT, Farsiu S, Freedman SF, et al. Abnormal foveal morphology in ocular albinism imaged with spectral-domain optical coherence tomography. Arch Ophthalmol 2009;127:37-44. [Crossref] [PubMed]
- Lee H, Proudlock F, Gottlob I. Is handheld optical coherence tomography reliable in infants and young children with and without nystagmus? Invest Ophthalmol Vis Sci 2013;54:8152-9. [Crossref] [PubMed]
- Shields CL, Mashayekhi A, Luo CK, et al. Optical coherence tomography in children: analysis of 44 eyes with intraocular tumors and simulating conditions. J Pediatr Ophthalmol Strabismus 2004;41:338-44. [Crossref] [PubMed]
- Rootman DB, Gonzalez E, Mallipatna A, et al. Hand-held high-resolution spectral domain optical coherence tomography in retinoblastoma: clinical and morphologic considerations. Br J Ophthalmol 2013;97:59-65. [Crossref] [PubMed]
- El-Dairi MA, Holgado S, O'Donnell T, et al. Optical coherence tomography as a tool for monitoring pediatric pseudotumor cerebri. J AAPOS 2007;11:564-70. [Crossref] [PubMed]
- Balmforth C, van Bragt JJ, Ruijs T, et al. Chorioretinal thinning in chronic kidney disease links to inflammation and endothelial dysfunction. JCI Insight 2016;1:e89173 [Crossref] [PubMed]
- Onal H, Kutlu E, Aydin B, et al. Assessment of retinal thickness as a marker of brain masculinization in children with congenital adrenal hyperplasia: a pilot study. J Pediatr Endocrinol Metab 2019;32:683-7. [Crossref] [PubMed]
- Hedges TR 3rd, Legge RH, Peli E, et al. Retinal nerve fiber layer changes and visual field loss in idiopathic intracranial hypertension. Ophthalmology 1995;102:1242-7. [Crossref] [PubMed]
- Gospe SM, Bhatti MT, El-Dairi MA. Emerging Applications of Optical Coherence Tomography in Pediatric Optic Neuropathies. Semin Pediatr Neurol 2017;24:135-42. [Crossref] [PubMed]
- Banc A, Stan C, Florian IS. Optical coherence tomography as a marker of vision in children with optic pathway gliomas. Childs Nerv Syst 2018;34:51-60. [Crossref] [PubMed]
- Gise R, Gaier ED, Heidary G. Diagnosis and Imaging of Optic Nerve Head Drusen. Semin Ophthalmol 2019;34:256-63. [Crossref] [PubMed]
- Chan NCY, Chan CKM. The use of optical coherence tomography in neuro-ophthalmology. Curr Opin Ophthalmol 2017;28:552-7. [Crossref] [PubMed]
- Khan M, Ehlers JP. Clinical utility of intraoperative optical coherence tomography. Curr Opin Ophthalmol 2016;27:201-9. [Crossref] [PubMed]
- Huang LL, Hirose T. Portable optical coherence tomography in management of vitreoretinal diseases: current developments, indications, and implications. Semin Ophthalmol 2012;27:213-20. [Crossref] [PubMed]
- Bendschneider D, Tornow RP, Horn FK, et al. Retinal nerve fiber layer thickness in normals measured by spectral domain OCT. J Glaucoma 2010;19:475-82. [Crossref] [PubMed]
- Budenz DL, Anderson DR, Varma R, et al. Determinants of normal retinal nerve fiber layer thickness measured by Stratus OCT. Ophthalmology 2007;114:1046-52. [Crossref] [PubMed]
- Pan CW, Qian YX, Zhong H, et al. Distribution and Determinants of Peripapillary Retinal Nerve Fiber Layer Thickness and Its Association with Sleep Quality in Chinese Teenagers. J Ophthalmol 2019;2019:6510203 [Crossref] [PubMed]
- Matalia J, Anegondi NS, Veeboy L, et al. Age and myopia associated optical coherence tomography of retina and choroid in pediatric eyes. Indian J Ophthalmol 2018;66:77-82. [Crossref] [PubMed]
- Baran RT, Baran SO, Toraman NF, et al. Evaluation of intraocular pressure and retinal nerve fiber layer, retinal ganglion cell, central macular thickness, and choroidal thickness using optical coherence tomography in obese children and healthy controls. Niger J Clin Pract 2019;22:539-45. [Crossref] [PubMed]
- de Boer JF, Leitgeb R, Wojtkowski M. Twenty-five years of optical coherence tomography: the paradigm shift in sensitivity and speed provided by Fourier domain OCT Biomed Opt Express 2017;8:3248-80. [Invited]. [Crossref] [PubMed]
- Choma M, Sarunic M, Yang C, et al. Sensitivity advantage of swept source and Fourier domain optical coherence tomography. Opt Express 2003;11:2183-9. [Crossref] [PubMed]
- Ferguson RD, Hammer DX, Paunescu LA, et al. Tracking optical coherence tomography. Opt Lett 2004;29:2139-41. [Crossref] [PubMed]
- Patel CK. Optical coherence tomography in the management of acute retinopathy of prematurity. Am J Ophthalmol 2006;141:582-4. [Crossref] [PubMed]
- Joshi MM, Trese MT, Capone A Jr. Optical coherence tomography findings in stage 4A retinopathy of prematurity: a theory for visual variability. Ophthalmology 2006;113:657-60. [Crossref] [PubMed]
- Gerth C, Zawadzki RJ, Werner JS, et al. Retinal morphology in patients with BBS1 and BBS10 related Bardet-Biedl Syndrome evaluated by Fourier-domain optical coherence tomography. Vision Res 2008;48:392-9. [Crossref] [PubMed]
- Scott AW, Farsiu S, Enyedi LB, et al. Imaging the infant retina with a hand-held spectral-domain optical coherence tomography device. Am J Ophthalmol 2009;147:364-373.e2. [Crossref] [PubMed]
- Muni RH, Kohly RP, Sohn EH, et al. Hand-held spectral domain optical coherence tomography finding in shaken-baby syndrome. Retina 2010;30:S45-50. [Crossref] [PubMed]
- Muni RH, Kohly RP, Charonis AC, et al. Retinoschisis detected with handheld spectral-domain optical coherence tomography in neonates with advanced retinopathy of prematurity. Arch Ophthalmol 2010;128:57-62. [Crossref] [PubMed]
- Avery RA, Cnaan A, Schuman JS, et al. Intra- and inter-visit reproducibility of ganglion cell-inner plexiform layer measurements using handheld optical coherence tomography in children with optic pathway gliomas. Am J Ophthalmol 2014;158:916-23. [Crossref] [PubMed]
- Avery RA, Cnaan A, Schuman JS, et al. Reproducibility of circumpapillary retinal nerve fiber layer measurements using handheld optical coherence tomography in sedated children. Am J Ophthalmol 2014;158:780-787.e1. [Crossref] [PubMed]
- Maldonado RS, O'Connell RV, Sarin N, et al. Dynamics of human foveal development after premature birth. Ophthalmology 2011;118:2315-25. [Crossref] [PubMed]
- Cabrera MT, Maldonado RS, Toth CA, et al. Subfoveal fluid in healthy full-term newborns observed by handheld spectral-domain optical coherence tomography. Am J Ophthalmol 2012;153:167-75.e3. [Crossref] [PubMed]
- Cabrera MT, O'Connell RV, Toth CA, et al. Macular findings in healthy full-term Hispanic newborns observed by hand-held spectral-domain optical coherence tomography. Ophthalmic Surg Lasers Imaging Retina 2013;44:448-54. [Crossref] [PubMed]
- Yanni SE, Wang J, Cheng CS, et al. Normative reference ranges for the retinal nerve fiber layer, macula, and retinal layer thicknesses in children. Am J Ophthalmol 2013;155:354-360.e1. [Crossref] [PubMed]
- Turk A, Ceylan OM, Arici C, et al. Evaluation of the nerve fiber layer and macula in the eyes of healthy children using spectral-domain optical coherence tomography. Am J Ophthalmol 2012;153:552-559.e1. [Crossref] [PubMed]
- Tsai DC, Huang N, Hwu JJ, et al. Estimating retinal nerve fiber layer thickness in normal schoolchildren with spectral-domain optical coherence tomography. Jpn J Ophthalmol 2012;56:362-70. [Crossref] [PubMed]
- Yeh EA, Weinstock-Guttman B, Lincoff N, et al. Retinal nerve fiber thickness in inflammatory demyelinating diseases of childhood onset. Mult Scler 2009;15:802-10. [Crossref] [PubMed]
- Varma R, Skaf M, Barron E. Retinal nerve fiber layer thickness in normal human eyes. Ophthalmology 1996;103:2114-9. [Crossref] [PubMed]
- Gürağaç FB, Totan Y, Guler E, et al. Normative Spectral Domain Optical Coherence Tomography Data in Healthy Turkish Children. Semin Ophthalmol 2017;32:216-22. [Crossref] [PubMed]
- Song WK, Lee SC, Lee ES, et al. Macular thickness variations with sex, age, and axial length in healthy subjects: a spectral domain-optical coherence tomography study. Invest Ophthalmol Vis Sci 2010;51:3913-8. [Crossref] [PubMed]
- Nigam B, Garg P, Ahmad L, et al. OCT Based Macular Thickness in a Normal Indian Pediatric Population. J Ophthalmic Vis Res 2018;13:144-8. [Crossref] [PubMed]
- Shokunbi T, Agbeja A. Ocular complications of head injury in children. Childs Nerv Syst 1991;7:147-9. [Crossref] [PubMed]
- Mueller I, Mast H, Sabel BA. Recovery of visual field defects: a large clinical observational study using vision restoration therapy. Restor Neurol Neurosci 2007;25:563-72. [PubMed]
- Gise R, Truong T, Parsikia A, et al. Visual Pathway Injuries in Pediatric Ocular Trauma-A Survey of the National Trauma Data Bank From 2008 to 2014. Pediatr Neurol 2018;85:43-50. [Crossref] [PubMed]
- Lee JY, Cho K, Park KA, et al. Analysis of Retinal Layer Thicknesses and Their Clinical Correlation in Patients with Traumatic Optic Neuropathy. PLoS One 2016;11:e0157388 [Crossref] [PubMed]
- Choi DD, Park MS, Park KA. Incidence of optic neuritis in Korean children and adolescents: A Nationwide survey and National Registry Analysis. J Neurol Sci 2020;408:116554 [Crossref] [PubMed]
- Kinnunen E. The incidence of optic neuritis and its prognosis for multiple sclerosis. Acta Neurol Scand 1983;68:371-7. [Crossref] [PubMed]
- Jonzzon S, Suleiman L, Yousef A, et al. Clinical Features and Outcomes of Pediatric Monophasic and Recurrent Idiopathic Optic Neuritis. J Child Neurol 2020;35:77-83. [Crossref] [PubMed]
- Waldman AT, Stull LB, Galetta SL, et al. Pediatric optic neuritis and risk of multiple sclerosis: meta-analysis of observational studies. J AAPOS 2011;15:441-6. [Crossref] [PubMed]
- Fukuchi M, Kishi S, Li D, et al. Acute ganglion cell loss during rapid visual recovery in optic neuritis. Graefes Arch Clin Exp Ophthalmol 2016;254:2355-60. [Crossref] [PubMed]
- Waldman AT, Hiremath G, Avery RA, et al. Monocular and binocular low-contrast visual acuity and optical coherence tomography in pediatric multiple sclerosis. Mult Scler Relat Disord 2013;3:326-34. [Crossref] [PubMed]
- Wilbur C, Reginald YA, Longoni G, et al. Early neuroaxonal injury is seen in the acute phase of pediatric optic neuritis. Mult Scler Relat Disord 2019;36:101387 [Crossref] [PubMed]
- Yeh EA, Marrie RA, Reginald YA, et al. Functional-structural correlations in the afferent visual pathway in pediatric demyelination. Neurology 2014;83:2147-52. [Crossref] [PubMed]
- Renoux C, Vukusic S, Mikaeloff Y, et al. Natural history of multiple sclerosis with childhood onset. N Engl J Med 2007;356:2603-13. [Crossref] [PubMed]
- Boiko A, Vorobeychik G, Paty DUniversity of British Columbia MSCN, et al. Early onset multiple sclerosis: a longitudinal study. Neurology 2002;59:1006-10. [Crossref] [PubMed]
- Kriss A, Francis DA, Cuendet F, et al. Recovery after optic neuritis in childhood. J Neurol Neurosurg Psychiatry 1988;51:1253-8. [Crossref] [PubMed]
- Lana-Peixoto MA, Andrade GC. The clinical profile of childhood optic neuritis. Arq Neuropsiquiatr 2001;59:311-7. [Crossref] [PubMed]
- Parkin PJ, Hierons R, McDonald WI. Bilateral optic neuritis. A long-term follow-up. Brain 1984;107:951-64. [Crossref] [PubMed]
- Yilmaz Ü, Gucuyener K, Erin DM, et al. Reduced retinal nerve fiber layer thickness and macular volume in pediatric multiple sclerosis. J Child Neurol 2012;27:1517-23. [Crossref] [PubMed]
- Graves JS, Chohan H, Cedars B, et al. Sex differences and subclinical retinal injury in pediatric-onset MS. Mult Scler 2017;23:447-55. [Crossref] [PubMed]
- Eyre M, Hameed A, Wright S, et al. Retinal nerve fibre layer thinning is associated with worse visual outcome after optic neuritis in children with a relapsing demyelinating syndrome. Dev Med Child Neurol 2018;60:1244-50. [Crossref] [PubMed]
- Datta R, Sollee JR, Lavery AM, et al. Effects of Optic Neuritis, T2 Lesions, and Microstructural Diffusion Integrity in the Visual Pathway on Cortical Thickness in Pediatric-Onset Multiple Sclerosis. J Neuroimaging 2019;29:760-70. [Crossref] [PubMed]
- Collongues N, Marignier R, Zephir H, et al. Long-term follow-up of neuromyelitis optica with a pediatric onset. Neurology 2010;75:1084-8. [Crossref] [PubMed]
- Banwell B, Tenembaum S, Lennon VA, et al. Neuromyelitis optica-IgG in childhood inflammatory demyelinating CNS disorders. Neurology 2008;70:344-52. [Crossref] [PubMed]
- Ratchford JN, Quigg ME, Conger A, et al. Optical coherence tomography helps differentiate neuromyelitis optica and MS optic neuropathies. Neurology 2009;73:302-8. [Crossref] [PubMed]
- Park KA, Kim J, Oh SY. Analysis of spectral domain optical coherence tomography measurements in optic neuritis: differences in neuromyelitis optica, multiple sclerosis, isolated optic neuritis and normal healthy controls. Acta Ophthalmol 2014;92:e57-65. [Crossref] [PubMed]
- Chen Q, Zhao G, Huang Y, et al. Clinical Characteristics of Pediatric Optic Neuritis With Myelin Oligodendrocyte Glycoprotein Seropositive: A Cohort Study. Pediatr Neurol 2018;83:42-9. [Crossref] [PubMed]
- Narayan RN, McCreary M, Conger D, et al. Unique characteristics of optical coherence tomography (OCT) results and visual acuity testing in myelin oligodendrocyte glycoprotein (MOG) antibody positive pediatric patients. Mult Scler Relat Disord 2019;28:86-90. [Crossref] [PubMed]
- Lechner C, Baumann M, Hennes EM, et al. Antibodies to MOG and AQP4 in children with neuromyelitis optica and limited forms of the disease. J Neurol Neurosurg Psychiatry 2016;87:897-905. [Crossref] [PubMed]
- Fernandez-Carbonell C, Vargas-Lowy D, Musallam A, et al. Clinical and MRI phenotype of children with MOG antibodies. Mult Scler 2016;22:174-84. [Crossref] [PubMed]
Cite this article as: Ciftci-Kavaklioglu B, Yeh EA. Evaluating visual outcomes using optical coherence tomography (OCT) in pediatric multiple sclerosis and other neuroinflammatory conditions. Ann Eye Sci 2020;5:16.


