
Visual dyslexia: towards an operational definition from a correlational study
Introduction
According to past literature a proportion of dyslexic children are found to be impaired in some aspects of visual processing, like contrast sensitivity (1-6), critical fusion frequency (7), motion perception (2,6-9), or visual persistence time (6,10-13). Notwithstanding, a convincing explanation of how such alterations may affect reading is missing (14-16).
Whether dyslexics suffer from reduced contrast sensitivity at low spatial frequencies is a highly controversial issue [see Skottun (14) for a critical revision of the topic]. Indeed, in this case low contrast sensitivity should affect letter recognition. Now, it has been reckoned that a reduction of contrast sensitivity by at least 1 log unit is necessary for halving the reading rate of a normal subject (17): and yet, contrast sensitivity in dyslexics is found to be lower than normal by no more than 0.1 log units for stationary stimuli, and by 0.3 log units for stimuli flickering at high temporal frequency; in addition, when present, the defect is observed at low luminance levels and not in the ordinary reading photopic conditions (2,18).
A parallel line of research provides evidence of reduced sensibility to dynamic stimuli in a significant proportion of dyslexic subjects. It has been argued that impaired motion perception is related to defective spatial encoding, since it would reflect how precisely letters are located and ordered during reading (9,19,20). According to other studies, motion perception would be more generally related to visual attention (21,22). Regardless of the interpretation, a straightforward explanation of how reduced motion sensibility affects the lexical task in dyslexic subjects is missing. The same applies to critical fusion frequency.
Finally, since the early seventies experimental evidence suggests that the interval required to discriminate two sequential stimuli is up to 100 msec longer in dyslexics than in normal readers. This finding, called increased persistence time, is expected to generate visual confusion during reading as it enables the syllables projected in the parafoveal area to interfere with the syllables on the fixation point (6,10,13). Yet, the magnitude of the phenomenon does not seem such as to justify a causal role in dyslexia: the duration of a fixation, in fact, generally is about 180 msec, and up to 450 msec (23). This is, indeed, an interval consistently longer than the visual persistence time found in dyslexic subjects.
As a matter of fact, Skottun and Skoyles stated that there is little evidence for a specifically temporal deficit (be it motion perception or visual persistence) in dyslexia (24).
Still, a proportion of dyslexics show visual alterations whose causal relation with the lexical performance seems more straightforward, namely increased spatial relationship anisotropy (SRA) and the supposedly related stronger lateral masking (25-31), unstable ocular dominance (32-37), and, as we have recently posited, abnormal interocular inhibition (38,39). It has been stated in fact, that the competitive inhibitory interactions that characterize sensory dominance promote image segmentation and grouping (40): two perceptive tasks that play an important role in decoding the written text.
This exploratory study investigates whether in a particular class of dyslexics these visual alterations provide a contribution to their reading disability.
Increased SRA and lateral masking
Crowding (or lateral masking) is the reciprocal masking effect of contiguous stimuli; this occurs if their spatial separation is below a critical spacing (41).
Stronger crowding in dyslexic children is revealed by improved performance (higher reading rate) as the distance between letters is made larger (25-31,42,43). Actually, as the space between adjoining characters widens, the amount of reciprocal lateral masking decreases. Indeed, Spinelli and colleagues found that the vocal reaction time when reading isolated words decreased when the interletter spacing was made wider in half of their dyslexic group, but not in controls (31). Zorzi and colleagues found similar results in a sample of French and Italian dyslexic children (43). In line with these findings, we observed positive correlation between reading time and interletter spacing in a group of school-age children with a formal dyslexia diagnosis, but not in normal age-matched readers (25).
We have recently hypothesized that increased lateral masking in dyslexics depends on the anisotropic contraction of the visual space along the X-axis. In this respect, it should be noted that the anisotropic contraction of the visual space would contribute to increased lateral masking not in the classical sense of “increased central (or foveal) crowding”, as it occurs in amblyopia, but in terms of its abnormal distribution across the paracentral visual field, i.e., where its main effect is exerted. The paracentral area, indeed, is crucial for the lexical task as it pre-processes each line of text during reading (26,28-30). Individuals with this contraction would experience reduced distance between letters. In support of this prediction, we found that dyslexic readers often show defective spatial relationship perception (SRP), which is the sensibility to the relative extent of a configuration along the X-, and Y-axes (the aspect ratio). In fact, a previous study reported that a proportion of dyslexics have problems to distinguish horizontal ellipses from circles, whereas their discrimination threshold for vertical ellipses was as low as that of normal readers (25).
This difference in discrimination threshold along the horizontal/vertical axis can be defined SRA. Increased SRA, therefore, is an expression of abnormal (unbalanced) SRP. In previous investigations, SRA was reported to be almost double in disabled school-age readers compared to a sample of age-matched normal subjects (25), and up to three times higher in adult dyslexics compared to normal adult readers (39).
The effect of abnormal SRA on the lexical performance of dyslexics may be revealed by improved reading fluency when the space between the letters is widened: in this case, therefore, the correlation between interletter spacing and reading rate can be considered a visual marker in dyslexic children. In other words, it suggests that a visual factor may contribute to the lexical disability.
Unstable ocular dominance
When reading, the left and right visual axes converge after each saccadic movement in order to provide exact retinal correspondence of letters/syllables. Even slight misalignments in the retinotopic location of letters in the two eyes can prevent binocular motor fusion and cause visual confusion (44).
To neutralize, or at least minimize this complication, one of the two eyes is assigned the role of “gaze director” by the visual system. Indeed, the presence of a dominant eye is stated to prevent the occurrence of fleeting slight positional differences of the two visual axes, allowing a fixed reference in binocular conditions (45-47). On the contrary, if ocular dominance alternates between the left/right eye (unstable dominance), the visual axes will oscillate around the syllable in foveal projection, hampering its recognition and its positional encoding (32,35,36,46,48).
Consistent with this assumption, unstable ocular dominance in developmental dyslexia has been reported in a substantial proportion of subjects (32-37,46,49-51), that have therefore been defined as “visual dyslexics” (45).
Yet, some studies did not confirm the prevalence of unstable dominance in the dyslexic population (47,52-54) and its association with poor lexical performance (47,55). In addition, the estimate of dominance provided by the Dunlop synoptophore test [or a modified version (56) used in these experiments] is argued to be biased by chronological and mental age, making the test unreliable, especially in non-experienced hands (47,52,57,58).
Ocular dominance investigated in the above studies is more properly called “motor ocular dominance”. A different type, namely sighting dominance [according to the classification of Evans (59)], may be more indicative in dyslexia. Motor dominance refers to the eye that shows fewer fixation shifts when the visual axes converge toward a syllable (vergence), whereas sighting dominance refers to the eye used when performing monocular tasks. Its role is optimizing the motor visual directionality by providing a fixed, reliable egocentric reference point (60-62). During reading, weak or unstable sighting dominance may determine characters and syllables position uncertainty, thereby degrading letters/syllables positional coding (58). In regard to this, it worth reminding that a link between sighting and motor dominance has been postulated (63).
Since the tests for sighting dominance provide unequivocal results, consistent across different measurement methods, reliable (55,64-67), and free from most of the drawbacks that affect the Dunlop test, there is ground for considering the measurement of sighting dominance as the soundest indicator of eye dominance: its role in reading should therefore be investigated.
Sighting dominance is commonly estimated by the Hole-in-the-Card test, considered as the most consistent among the sighting tests (68), or by similar procedures based on the alignment task (69-72). In all cases the observer is asked to look binocularly at a target aligned to a proximal stimulus, and the dominant eye is the one that continues to perceive the target (or that maintains the alignment) after the fellow eye is excluded. This way, the dominant eye can be detected to check for a crossed dominance (left arm/right eye or vice versa); yet, estimating the degree of dominance stability can be an even more indicative parameter (55). In support of this assumption, Sejias, pinpointed the importance to develop a technique able not only to identify the dominant eye, but also to quantify the magnitude of dominance (68).
To address this issue, we devised a modification of the classical hole-in-the-card test, which have called Domitest-M. This procedure aims to detect unstable sighting dominance and, in addition, to measure the displacement of the egocentric reference, so as to evaluate if this variable may be involved in the reading disability. Contrary to the Dunlop test, our technique does not require skilled operators, does not make use of a synoptophore or other expensive instruments, and is user-friendly.
Since, as reminded, weak dominance is supposed to affect letters/syllables positional coding during reading, widening the distance between letters should prevent unstable dominant readers from positional errors. Like for SRA, the effect of a weak ocular dominance (estimated with the Domitest-M) on the lexical task would therefore be revealed by the presence of the visual marker. As a confirmation, Cornelissen and associated reported that suspected dyslexics with unstable dominance are more prone to non-words errors when the text print size is reduced (therefore when the distance between the characters is made smaller), contrary to children with stable dominance (73).
Abnormal binocular sensory interaction
Since the sixties, binocular sensory interaction is found to be modulated by suppression mechanisms (74-76), and asymmetric inhibitory interocular modulation is actually regarded as the basis of sensory ocular dominance (77). The sensory dominant eye is the eye whose percept prevails when the brain receives two different visual inputs. Due to sensory dominance, two different images presented dichoptically for a given period of time generate binocular rivalry, so that one image dominates for a given interval of time, then it is inhibited and replaced by the contralateral one. This cyclic alternation of perception, called bistability, can be biased toward the left or the right eye, and the sensory dominant eye refers to the eye that shows overall shorter periods of inhibition.
Binocular inhibitory interaction is thought to be engaged in image perceptual segmentation and grouping (40): two functions, indeed, involved in decoding the written text. It is conceivable that excessive binocular inhibitory interaction affects letter positional coding during reading. So, it is worthwhile establishing whether abnormal sensory interaction contributes to the reading disability. In this study, therefore, also the interocular sensory dynamics of dyslexic children has been investigated.
The techniques commonly used to measure this function make use of rivalry paradigms (77-81): with the use of rivalry paradigms, sensory input in normal subjects is found to be substantially balanced (68,77,80,82,83).
The procedures based on dichoptic rapid serial visual presentations (RSVPs) provide a more objective measure of interocular differences in the pattern of suppression compared to rivalry paradigms (81). In the RSVP stimuli like letters, digits, or figures are presented tachistoscopically at the same location, and a target is embedded in the stream. The observer is asked to identify the target (84). By using this approach in dichoptic conditions and with alphabetical stimuli, Valle-Inclán et al. reported consistent perceptual suppression in their sample of normal readers, and a wide range of dominance values (81).
In two previous investigations (38,39), we described a RSVP-based technique similar to that presented by Valle-Inclán, but with the alphabetical stimuli replaced with checkerboard patterns, in order to make the procedure more suitable to test dyslexic subjects. By using this paradigm, we have called Domitest-S, we found that the left/right eye sensory input is substantially balanced in the majority of the normal school age population, but it is even more balanced in adult (mature readers). In addition, the distribution of the interocular inhibition is unimodal in adult (mature) readers, whereas it is bimodal and stronger in children (38) [confirming what previously reported by Li and colleagues (77)], and in dyslexic adults (39).
In this paper, we intend to evaluate if abnormal interocular inhibition as well as unbalanced sensory input affect the positional coding of letter and syllables in dyslexic readers. We postulate that the detrimental effect of this function on the readability of the characters depends on their spatial density: therefore, also in this case a correlation between reading rate and interletter spacing is expected.
In sum, the exploratory hypothesis is that there are groups of dyslexics that have at least one of the visual-perceptive alterations mentioned above: abnormal SRA putatively responsible for abnormal crowding, weak sighting dominance, and abnormal binocular interaction. In these cases, making interletter distance larger should improve the reading rate, so that positive correlation between reading rate and inter-letter distance can be taken as a marker of “visual dyslexia”.
Evidently, to investigate these assumptions it is necessary:
- To provide a cutoff of normality for the three visual-perceptive variables;
- To verify that dyslexics with the three variables below the cutoff of normality do not present the visual marker;
- To ascertain that dyslexic readers with one of the three variables above the cutoff are positive to the visual marker. This is a material point in order to maintain that the three visual parameters are causally related to the lexical task in these classes of subjects, justifying their definition of “visual dyslexics”.
Methods
Measurement of the SRA
A detailed description of the psychophysical test devised to measure SRA as well as the normative data have been reported in previous papers (25,85). The SRA estimation relies upon the difference in the discrimination threshold between vertically/horizontally oriented ellipses and circles. The variable of interest is the eccentricity of the ellipse, quantified by its focal axis, while the other parameters (luminance, contrast, size) are kept constant. The degree of eccentricity is expressed as interaxis ratio (IR):
IR = [f(x) – f(y)]/f(mx,y)
where f(x) and f(y) are, respectively, the horizontal and vertical axes, and f(mx,y) is the focal axis. This way, the IR at threshold represents the minimum eccentricity required to discriminate an elliptical from a circular stimulus. Naturally, the IR of a circumference is =0. The IR values ranged from 0.01 to 0.34.
Elliptical and circular targets (mean target size: 300 min arc, contour line 1.8 min arc wide, luminance =160 cd/m2; luminance background: 40 cd/m2) are presented centrally for 200 msec at a viewing distance of 50 cm on a 262K LCD screen (resolution: 1,280×1,024 pixels, 60 Hz) surrounded by a black sinusoidal mask (radial frequency: 0.022 c/deg: Figure 1). The exam is performed binocularly in a dim room.
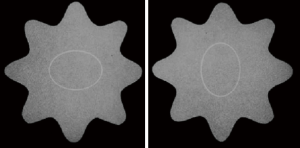
A white flickering central cross (34.2 min arc wide, 6.6 Hz) is presented at the center of the screen for 1 sec to capture the fixation of the subject before the onset of each stimulus. The subject has to recognize the target as a circle or a horizontal/vertical ellipse according to a forced triple choice procedure (3AFC).
At each trial the level of eccentricity varies according to two interleaved but independent modified staircase 4-2-1 procedures, one selecting the horizontally oriented stimuli, the other positioning the vertically oriented stimuli. This way, the discrimination threshold referred to the horizontal (X-) and the vertical (Y-) coordinate are estimated separately during the same session, and the response to one orientation does not affect the staircase run referred to the other: their difference (X-threshold-Y-threshold) quantifies the amount of SRA of the visual system under examination. In this experiment SRA (X-threshold-Y-threshold >0) expresses greater sensitivity along the vertical axis, suggesting that visual stimuli are perceived as contracted along the horizontal coordinate to a certain extent: evidently, the same anisotropy is expected to affect words when reading, so that in subjects with consistent SRA adjoining letters will be perceived closer (i.e., more crowded).
Measurement of sighting dominance
The Domitest-M, a modified version of the hole-in-the-card test, has been devised as follows: a rectangular paperboard with a horizontal graduate scale on both sides of a fixation target (a star) is placed 70 cm from the observer, who sits on a chair with the head on a chinrest. A black mask with a hole (7 mm wide) in the center is placed midway (35 cm) between the observer and the scale. The separation between each number (size: 0.4 deg) on the scale is 1 deg at the viewing distance.
The test is administered in a well illuminated room. The subject aims binocularly at the star through the hole. Then the eyes are occluded alternatively and the observer is asked to report the number seen on the graduate scale after the dominant eye is excluded.
The procedure is repeated five times, and each time the observer is asked to remove and reposition the head on the chinrest; if the same score on the same scale is recorded at every trial, dominance is considered stable. Otherwise, dominance is classified as unstable.
Based on a previous study by Barbeito & Simpson, the angular difference between the pointing direction of the dominant eye aiming at a target through the hole and the contralateral (non-dominant) eye measured after occluding the former reflects the displacement of the egocenter (61): thereby, in case of stable dominance the angular value reported by the observer is adopted to quantify the lateralization of the sighting dominance (Figure 2).
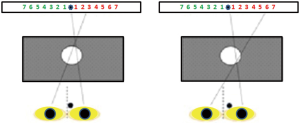
This way, the procedure provides a binary outcome of dominance (stable/unstable), and a parametric score of the degree of its lateralization.
Measurement of the asymmetry and strength of the interocular inhibition
The description of the technique devised to evaluate the sensory interaction and the amount of interocular inhibition, along with the normative data collected in a sample of more than 150 normal school-age readers (3rd, 4th and 5th grade), is reported in detail in a previous paper (38).
Fifteen pairs of sequences, each made of 10 stimuli subtending a visual angle of 1° in foveal projection, are displayed dichophtically at a viewing distance of 70 cm on a 262K LCD color monitor (1,280×1,024 pixels, 60 Hz) according to a RSVP paradigm (84). Each sequence consists of a 5×5 matrix made of 0.2° wide black and white squares (luminance: 0.3 and 240 cd/m2, respectively) arranged to form a “X” (the target) embedded in a stream made of nine matrixes arranged in pseudorandom order to form a checkerboard-like pattern (null stimuli). The luminance of the background was 240 cd/m2. The presentation time of each stimulus is 200 msec with no interstimulus interval: therefore, the duration of each stream is 2 seconds. The stimuli are projected 2 deg apart from the midline for 200 msec, the left and right sequences are presented simultaneously, and the dichoptic condition is obtained by using a rectangular cardboard mask (50.5 cm wide × 25 cm high) placed perpendicularly between the midline of the screen and the face of the observer, and aligned to the nose. At every trial the “X” is displayed at a random position (from position 2 to position 9) to the left or the right eye, so that at the end of the examination each eye is presented the same number of targets. The task is to report the detection of the target by pressing a button. In the presence of the target, no response is considered as a miss answer and the next trial occurs after 2 seconds (Figure 3).
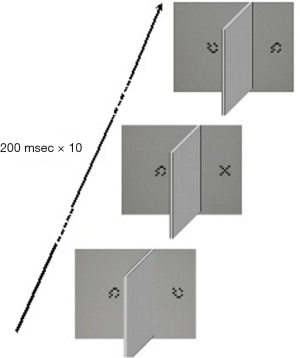
To assess the reliability of the test, false positive (FP) and false negative (FN) errors are computed. FN errors are evaluated by administering three sequences with no target. In turn, FP errors are evaluated by presenting three trials with the target displayed to both eyes in the same temporal position within the stream. Examinations with more than one type I error and more than one type II error are considered as unreliable, then discarded and repeated after a few minutes.
The difference between the right-left proportion of correct responses is assumed as the indicator of binocular input imbalance [imbalance value (IBV)], so that the higher is the IBV (taken as absolute value), the more imbalanced is the binocular sensory processing. Evidently, IBV =0 indicates complete binocular sensory balance.
To quantify the interocular inhibitory interaction, the interocular inhibitory index (III) has been computed as:
III = 2 – (Lpercent correct + Rpercent correct)
To be noted that the short presentation time of the stream avoids that sources of external noise, like eye blinks and shifts of fixation, affect the procedure. In addition, the dichoptic RSVP paradigm has been preferred to the current rivalry paradigms as tachistoscopic presentations disengage sustained attention, minimizing, especially in children, attentional drops.
Assessment of the visual marker
On the same high-resolution LCD screen adopted in the tests previously described, 11 presentations of words and 11 presentations of non-words are displayed in randomized order. To evaluate the effect of the interletter distance on the lexical as well as sublexical route, each presentation (or trial), is made of four words or five non-words (12 syllables in all) placed side by side (font free monospace). The lexical material is suitable to the age of the child: from the 3rd to the 5th grade. Words and non-words are made of dark letters 0.4 degrees in size (i.e., well above the foveal recognition threshold) displayed on a background with luminance of 85 cd/m2). Trials are presented unmodified (i.e., in their normal spatial relationship) or stretched/contracted along the horizontal axis (anisotropization) so that the interletter distance (computed center-to-center) ranged from 0.2 up to 0.51 deg (in 13 steps), with 0.4 deg taken as the reference interletter spacing. This interval has been chosen in line with two earlier studies (86,87), and it matches the interletter distance commonly adopted in the Italian lexical material.
The lexical sample presented at each trial differs from the previous one. The subject is required (without being urged to the best performance) to read each presentation, that remains visible on the screen the time required to complete the operation. Time after time the reading rate (as syllables per second) is automatically recorded via a wireless tablet activated by the technician. To make the measurement of the reading time more precise, the software tests the auditory reaction time of the operator in advance, then subtracts the obtained temporal interval from the reading time recorded at each trial.
After completion of the first set of presentations, a second set made of trials with different lexical material is administered, so as to provide two estimates for each value of anisotropization. The best estimate of each couple of trials is selected and entered in the data analysis. At the end of the test, correlation between reading rate and interletter distance is finally computed. Pearson r is considered significant for P<0.05 (25).
Sample
The records of one hundred ninety-three school-age dyslexic or retarded readers (108 males, 85 females, age 7–10 years, 3rd–5th grade of primary school) who underwent our protocol of sensory evaluation in the past 6 months have been selected from the archive of the neuro-ophthalmology service of our department by a physician not involved in the study. Children were referred to our service from neuro-psychiatrists and speech therapists interested in obtaining information about the co-occurrence of a visual-perceptual impairment.
The diagnosis of reading disability was made by a neuro-psychiatrist according to the criteria outlined by Stanley and Hall (88) that is: lexical age reduced by at least 2.5 years with reading rate, normal intellectual ability and normal or above normal IQ, normal visual acuity and no behavioral or auditory impairment. IQ was measured by the WISC-III scale, while reading rate was assessed using the MT Reading Test (89).
Exclusion criteria were pediatric ophthalmological diseases, hyperopia/myopia >2 diopters, astigmatism >1.5 diopters, natural or corrected visual acuity <50/60 in one eye, eso/exotropy, poor convergence (>8 cm from the nose), auditory impairment, behavioral problems, diagnosed attention deficit hyperactivity disorders (ADHDs), and poor collaboration. Phorias that were not considered an exclusion criterion unless they were judged as severe (exophorias higher than 13 pd and esophorias higher than 2 pd measured with adequate refractive correction). None of the children in the sample had severe phorias or poor convergence. After a preliminary ophthalmological and orthoptic examination (comprehensive of cycloplegic refraction with tropicamide 0.5% and a summary estimate of accommodation and convergence), SRA, sighting dominance, and binocular sensory interaction have been evaluated in randomized order. The correlation analysis between interletter spacing and reading rate was left until last.
In order to rule out learning effect and to help the subjects get accustomed to the test, in our clinical practice each trial is always repeated twice, and the first estimates are discarded.
Evidently, the problem of gathering reliable data is crucial when administering psychophysical tests to children. The issue is more evident with dyslexic pupils, who generally feel uncomfortable with the request of performing reading tasks, and in particular reading aloud. And yet, reading words and non-words aloud was necessary in our experiment to assess if reading speed increases as a function of the interletter distance (i.e., in order to test for the visual marker of dyslexia). In addition, the fluctuation of the performance due to attentional drops, emotional as well as other psychological factors can frustrate the search for significant and potentially interesting trends: even more so, considering the well-known high susceptibility of the regression and correlation analysis employed in this study to outliers and aberrant observations.
For all these reasons the biasing effect of unreliable estimates on the regression model adopted to test for the visual marker was our main concern. The initial sets of data on the reading rate measured as a function of the interletter spacing have therefore been filtered based on a restrictive criterion: namely, tests with the reading rate at the narrowest interletter distance higher than at the reference distance (0.4 deg) were considered unreliable, thereby discarded.
By applying this criterion the reliable dataset for the computation of the regression models dropped to 112 reliable observations with the words samples and 76 reliable observations with the non-words samples.
The Grubbs test (alpha level: 0.05) performed on the two samples of reliable observations revealed no outliers in each correlation analysis.
Cutoff assessment for the variables under consideration
The threshold of abnormality for the three parameters has been established as follows:
- SRA abnormal if >0.05. The cut off is derived from the average SRA of normal readers + 1.5 SD estimated in our previous study (25). As reported in that paper, only one normal reader (4.3%) vs. 38% of the dyslexic subjects had SRA above this threshold. This parameter is similar (even if more conservative) to the one we adopted in the abovementioned investigation (average SRA of normal readers + 1 SD) as well as to that chosen by Spinelli and colleagues (31) to define the cut-off for abnormal crowding. One percent point equals to a difference of 3.3 arc min between the x, y at the adopted viewing distance. So, subjects with an anisotropic perception above the cutoff were unable to identify ellipses with difference between the two axes lower than 16.5 min arc. In the recruited sample, the prevalence of subjects with SRA above the cutoff of normality was 22.7%.
- Sighting dominance abnormal if unstable or with lateralization <4 deg: preliminary assessment of the stability of sighting dominance has been performed on a sample of 17 normal school-age children (9 females, 8 males, range 8–10). All subjects showed stable dominance, in line with the results obtained by Stein & Fowler with the Dunlop test (46). In addition, the egocenter displacement has been estimated in the same group. No correlation was found with interpupillary distance (r=0.09, P=0.71) or phorias (r=0.01, P=0.96). Irrespective of the direction of lateralization, median displacement of the egocenter was 7 deg (95% CI: 4–8 deg). In this respect, the lower limit of the confidence interval has been chosen as the cut-off of normality.
- IBV abnormal if >0.27, that is beyond the third quartile in the frequency distribution of a normal age-matched population, as evaluated in a previous study (38). The normal school-age population was made of 152 children (mean age 9±0.8 years) recruited from a 3rd, 4th and 5th school grade primary school. This assumption is supported by the evidence that in almost 90% of mature readers the IBV is equal to or lower than 0.27 (39): the sample of mature readers was made of 95 normal adolescent and adult subjects, ranging in age from 12 to 26 years (Figure 4, upper panels).
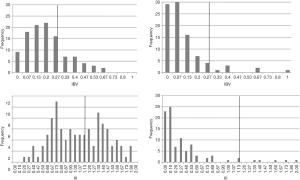 Figure 4 Upper panels: frequency distribution of the binocular sensory asymmetry expressed as IBV. Abscissa: IBV; ordinate: frequency of the observations. Lower panels: frequency distribution of the interocular inhibition (expressed as III). Left: normal children (immature readers [from Aleci and colleagues (38)]; right: mature readers [from Aleci and colleagues (39)]. Abscissa: III, ordinate: frequency of the observations. In all the cases the vertical line represents the cutoff level. IBV, imbalance value; III, interocular inhibitory index.
Figure 4 Upper panels: frequency distribution of the binocular sensory asymmetry expressed as IBV. Abscissa: IBV; ordinate: frequency of the observations. Lower panels: frequency distribution of the interocular inhibition (expressed as III). Left: normal children (immature readers [from Aleci and colleagues (38)]; right: mature readers [from Aleci and colleagues (39)]. Abscissa: III, ordinate: frequency of the observations. In all the cases the vertical line represents the cutoff level. IBV, imbalance value; III, interocular inhibitory index. - III abnormal if >1.13, that is when the interocular inhibition of the subject matches the second cluster (subjects with strong interocular inhibition) found in normal school-age readers, as evaluated in a previous study (38). The rationale of this assumption relies on the evidence that the second cluster is found absent in mature readers (39) (Figure 4, lower panels).
The cutoff levels for the continuous variables under consideration are summarized in Table 1.
Table 1
| Variables | Cutoff | Reference |
|---|---|---|
| SRA | 0.05 | Aleci |
| IBV | 0.27 | Aleci |
| III | 1.13 | Aleci |
SRA, spatial relationship anisotropy; IBV, imbalance value; III, interocular inhibitory index.
Subjects exceeding the cutoff relative to each of these variables have been selected (only one variable above the cutoff at a time). In each of the three groups the detrimental effect of the abnormal parameter on the reading performance has been verified checking for the visual marker. The presence of the visual marker is verified by performing regression analysis between interletter spacing and normalized reading rate after the latter has been normalized by dividing the value measured at each interletter distance by the value measured at the reference spacing (0.4 deg).
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional ethics board of the University of Turin as a degree Thesis (No. 754741/10.11.2016). As a retrospective study performed on an existing database, no informed consent was necessary.
Results
Of the selected sample, 12 and 37 children did not complete the reading test with words and not words, respectively. Average reading rate obtained from the remaining 181 examinations with words and 156 examinations with non-words was 2.16 syl/sec (±1.13) and 1.28 syl/sec (±0.56).
Based on the criterion reported in “Sample” section, 112 tests in the word modality and 76 in the non-word modality were judged reliable, therefore entered in the regression analysis for the assessment of the visual marker.
Thirty-three subjects [mean age: 8.78 (±1.26) years] did not exceed the cutoff of anisotropy + binocular sensory balance + interocular inhibition, and sighting dominance was stable: this group has been considered as the reference sample of “non-visual dyslexics” [non-visual sample (NVS)]. In the NVS 25 and 12 reliable reading measures were obtained in the words and non-words modalities, respectively. In both cases data were normally distributed (D: 0.15 and D: 0.19, P>0.10 at Kolmogorov-Smirnov test). Mean reading rate in the NVS was 2.55 syl/sec (±1.40) at words and 1.69 syl/sec (±0.89) at non-words.
Twenty-four subjects exceeded the threshold of anisotropy, while the other visuoperceptive indexes under evaluation were normal [anisotropic sample (AS)]; in turn, 12 children showed binocular sensory imbalance above the cutoff [binocular imbalance sample (BIS)], and 10 dyslexic readers showed abnormal interocular inhibition [interocular inhibition sample (IIS)]. Finally, 12 subjects had unstable sighting dominance [unstable dominance sample (UDS)]; in all cases the other visual parameters were normal. The five subgroups were matched for age [Kruskal-Wallis, (KW): 2.55, P=0.63], and the reading rate did not differ significantly (words: F: 2.29, P=0.06, one-way ANOVA; non-words: KW: 7.36, P=0.1, KW).
In the group showing unstable ocular dominance, IBV was lower compared to the NVS. In any other class each value referred to the visual variables not characterizing that class was not statistically different from the other classes (Table 2).
Table 2
| ANOVA | Class | SRA | IBV | III |
|---|---|---|---|---|
| a | NVS | 0.67 (±2.46) | 0.16 (±0.08)† | 0.61 (±0.33) |
| b | AS | 8.95 (±3.18) | 0.12 (±0.09) | 0.63 (±0.29) |
| c | BIS | 0.63 (±2.11) | 0.42 (±0.09) | 0.67 (±0.41) |
| d | IIS | 0.1 (±2.33) | 0.1 (±0.07) | 1.52 (±0.24) |
| e | UDS | 0.66 (±2.67) | 0.07 (±0.10)† | 0.61 (±0.28) |
In brackets is the standard deviation. ANOVA (a, c, d, e): F: 0.10, P=0.95; ANOVA (a, b, d, e): F: 3.07, P=0.03; ANOVA (a, b, c, e): F: 0.10, P=0.95. †, Tukey-Kramer: q(3.72)=3.95. SRA, spatial relationship anisotropy; IBV, imbalance value; III, interocular inhibitory index; NVS, non-visual sample; AS, anisotropic sample; BIS, binocular imbalance sample; IIS, interocular inhibition sample; UDS, unstable dominance sample.
AS
Figure 5 shows the effect of interletter spacing on reading in the NVS and in the AS.
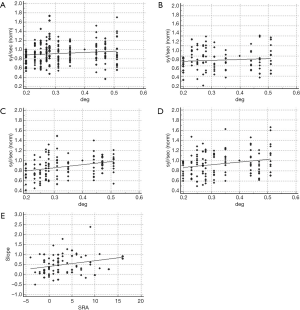
Text manipulation did not affect the reading rate of the non-visual dyslexics, irrespective of the lexical material administered (words: R2: 0.24, P=0.11; non-words: R2: 0.06, P=0.44; Figure 5A,B). Indeed, in the NVS the visual marker was positive only in two cases (words modality, FP rate: 6%).
Conversely, the dyslexic children with SRA above the cutoff showed improvement of the reading rate as a function of the distance between adjoining characters (words: R2: 0.51, P=0.01; non-words: R2: 0.43, P=0.02; Figure 5C,D). As a confirmation of the effect of SRA on the lexical visual processing, in the total population of dyslexic readers the amount of anisotropy showed a positive correlation with the slope of the regression model (R2: 0.07, P=0.02; Figure 5E).
Abnormal binocular sensory interaction
The relationship between interletter spacing and reading rate in the sample with abnormal binocular sensory interaction is shown in Figure 6. In the presence of imbalanced sensory input the coefficient of determination was not significant (R2: 0.09, P=0.35, and R2: 0.36, P=0.04 for words and non-words, respectively). Reading rate of subjects with increased interocular inhibitory pattern was related to the interletter spacing when words were administered (R2: 0.60; P=0.004), but not with non-words (R2: 0.12, P=0.28; Figure 6A,B,C,D).
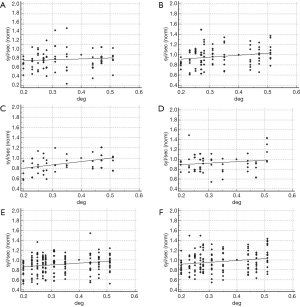
In order to describe the binocular sensory interaction in a more comprehensive way, a vector Vsi, combining the amount of interocular inhibition (III) and the degree of imbalance of the sensory dominance (IBV) has been computed as:
Vsi = 3 × (1 – IBV) + III
Based on the IBV and III cutoffs (IBV: 0.27, III: 1.13, see “Cutoff assessment for the variables under consideration” section), the limit of normality for Vsi, is 3.32, as derived from the equation.
The coefficient of determination of the regression model computed in the sample with Vsi >3.32 was R2: 0.47, P=0.02 (words), and R2: 0.57, P=0.007 (non-words) (Figure 6E,F).
Sighting dominance
Twenty-seven subjects (14%) had unstable sighting dominance. In 12 cases the other variables were normal, while in the remaining cases dominance instability was associated with one, two or three of the other abnormal visual parameters (Table 3).
Table 3
| Class | Unstable dominants (nb) |
|---|---|
| NVS | 0 |
| UDS | 12 |
| UDS + AS | 3 |
| UDS + BIS | 4 |
| UDS + IIS | 4 |
| UDS + AS + BIS | 1 |
| UDS + AS + IIS | 3 |
| Total unstable ocular dominants | 27 |
NVS, non-visual sample; UDS, unstable dominance sample; AS, anisotropic sample; BIS, binocular imbalance sample; IIS, interocular inhibition sample.
The binary outcome of dominance obtained at Domitest-M (stable/unstable) was not related to the age of the observer (chi-squared: 1.70, P=0.19). In average, the unstable dominant dyslexics did not read slower than the stable dominant dyslexics when presented with words (t: 0.35, P=0.95) and with non-words (t: 1.73, P=0.09).
The coefficient of determination of the regression analysis between normalized reading rate and interletter spacing in the 12 children with unstable dominance was R2: 0.13 (P=0.26) at words, and R2: 0.43 (P=0.02) at non-words (Figure 7).
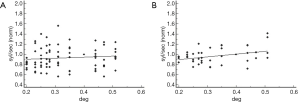
Finally, 32 dyslexics had the egocenter displacement under the lower limit (4 deg). Of these, in eight cases the other visual parameters were normal. In this group no correlation was found between egocenter displacement and words/not words reading rate (P>0.05 in both cases).
Discussion
Among the visuoperceptive alterations supposed to affect dyslexics, increased crowding and unstable fixation due to unstable ocular dominance seem to provide a straightforward explanation of why dyslexic children fail to read fluently. As we have posited (38,39), abnormal interocular inhibition could play a causal role as well.
A number of studies, indeed, showed that widening the distance between letters improves reading speed in unselected samples of dyslexics by counterbalancing the abnormal lateral masking between adjoining letters. Recently we postulated that the source of increased crowding in dyslexics relies upon an anisotropic SRP of the visual space (25); in addition, we have posited that larger intervals between letters help minimize the positional errors also in subjects with unstable ocular (sighting) dominance and with abnormal binocular sensory interaction (39).
The basic assumption is that the detrimental effect of these three visual anomalies on the lexical task can be postulated only if positive dyslexics prove to be sensitive to the distance between characters: this is the visual marker, in fact, that provides the basis for defining subjects showing one or more of these visuoperceptive problems as “visual dyslexics”.
Indeed, after having set the cut-off of normality for each of these visual parameters, the results obtained in this exploratory study suggest that reading is sensitive to interletter spacing (the visual marker) in the groups of dyslexics above the threshold of anisotropy, but not in those whose visuoperceptive parameters remain within the limit of normality.
As recalled, dyslexics in average perform better when the distance between characters is made larger. The same behavior, ascribed to abnormal crowding, is less evident or does not apply at all to normal readers (25,31,43,90-92). In this respect, Chung was the first to report that in general reading speed does not benefit from increased letter spacing in normal adult subjects (90).
However, Atkinson (27) and Spinelli and colleagues (31) showed that, when considering the individual cases, only a minority of disabled readers suffer from increased lateral masking. The criterion for abnormal crowding adopted by Spinelli was the difference between vocal reaction time of crowded and isolated words, and the cutoff was computed as this average difference measured in the normal population + 1 SD (31). The authors found that only 32% of the recruited dyslexics (7 out of 22 subjects) were affected by abnormal lateral masking. A similar proportion (about 30%, i.e., 4 out of 13 cases) has been reported by Atkinson.
The criterion adopted in the present study relies upon the measure of SRA, assuming that the higher is the anisotropy, the more evident is the effect that increased interletter spacing has on reading. We have suggested, therefore, that the anisotropic perception of the visual space affects lateral masking (25). Adopting a cutoff of normality similar to that used by Spinelli, in our larger sample 23% of the disabled readers (44 out of 194 subjects) fell beyond the cutoff of anisotropy, whereas about half of the subjects (49%) showed anisotropic levels below the normal average value of 0.02 computed in a previous study (25). Our finding, therefore, confirms that only a subgroup of dyslexic children suffer from abnormal lateral masking. As a matter of fact, the anisotropic group showed improved reading rate when interletter distance was increased across a range of 0.37 degrees, and the coefficient of the linear model correlated with the degree of anisotropy in the overall sample of dyslexics when the sublexical route was adopted (letter-to-letter reading). Evidently, the specific impairment of the visual channel in these subjects plays a consistent role in their reading disability, so that these cases could be defined “visual dyslexics”.
On the contrary, the ameliorative effect was absent in the non-visual dyslexics, that are dyslexics who did not show abnormal anisotropy (as well as alterations in the other visual variables considered).
In summary, our data suggest that dyslexics who show anisotropy beyond the normal range tend to read more fluently when the interletter distance is made wider, while dyslexics with anisotropy below the cutoff level do not. Based on this finding, the former (but not the latter) suffer from this specific impairment of the visual channel we have posited to lead to reinforced crowding: this way, increased SRA, rather than being an epiphenomenon, is proved to play an active, causal role in determining the reading disability. It is worth recalling that, according to previous studies, SRA in young adults (mean age 29.3±11.6 years) was half the value found in non-dyslexic children (mean age 9.2±2.0 years, SRA: 1.13±2.19 vs. 2.36±1.75) (25,85). It is therefore arguable that SRP matures through development, and this maturation may be a requisite for normal reading acquisition.
The correlation between reading rate and interletter spacing is not limited to reveal dyslexics with abnormal SRA, but can be regarded as a more general marker of visual-perceptive impairment. Indeed, as pointed out by Perea and colleagues (91) and Perea (92), the ameliorative effect of larger distances between characters in dyslexics relies on improved letters position coding within words [see Davis (93) and Gomez and colleagues (94) for two models]. As a matter of fact, we have found that the lexical performance of dyslexics with normal crowding but unstable sighting dominance and, more evidently, in those with abnormal interocular interaction tend to improve if the spatial density of the letters is reduced.
In this study only a minority of dyslexic children had unstable sighting dominance, isolated or associated with other visual-perceptive alterations. The proportion is far lower than that found by Stein and Fowler with a modified version of the Dunlop test (52% to 63%) (46), and by Bigelow and McKenzie with a Dunlop test-derived portable device (tranaglyph: 64%) (48). Probably, as already suggested, different procedures provide different estimates (62,67): as a matter of fact, in a study of Zeri and colleagues motor dominance (Dunlop test) was unstable in 65% of cases whereas no dyslexics with unstable sighting dominance were reported (55). Indeed, it has been recalled that the Dunlop test (and presumably the tranaglyph) is more demanding compared to the hole—in the card-based sighting tests, as in the former a fusional effort is required during the examination (95): on the basis of this, it is expected that a higher proportion of dyslexic subjects fail the Dunlop test compared to the sighting tests.
In our study the reading rate of unstable dominants is not worse than stable dominants, in support of the lack of correlation between unstable ocular dominance and reading performance previously found by Newman (47) and Zeri (55). Yet, unstable dominants showed improved reading speed when the distance between letters of non-words was made larger: probably fixation instability affects mainly the sublexical route, that is syllable-by-syllable decoding. It would be interesting to know if normal readers with unstable dominance show the visual marker as well, revealing itself as FP.
Finally, according to our definition binocular sensory interaction is determined by the degree of left/right sensory imbalance and by the amount of interocular inhibition. Even if on average the binocular input is substantially balanced in the normal population (68), in individual cases it is biased to the left or right side to a certain degree. Kommerell and colleagues used the Freiburg Ocular Prevalence Test and provided a nominal scale to quantify the amount of perceived displacement of two targets presented at stereo-disparity of 7’ in the midline between the two eyes (82). The scale ranged from 0% (no prevalence, i.e., perfect balance) to 100%. The authors found evident imbalance in 65% of cases. Similarly, the Domitest-S provides a quantitative scale of sensory asymmetry ranging from 0.00 (perfect balance) to 1.00 (complete imbalance). In line with the finding reported by Kommerell and colleagues, in previous investigations we found that the sensory input was substantially imbalanced (IBV ≥0.2) in 64% of normal children, but only in 20% of normal adult (“mature”) readers. Yet, we found that in a consistent number of cases the degree of this asymmetry was small, with IBV (irrespective of the L/R side) lower than 0.1 in 25% of normal children, increasing up to 63% in adults (38,39). The lower sensory input asymmetry in adult (“mature”) readers leads us to believe that sensory imbalance tends toward reduction with the sensory development, so that its persistence above a certain level could be a detrimental factor for reading fluently.
Forty-five percent of our dyslexics showed substantial sensory imbalance. The proportion is not higher compared to the normal school-age children, suggesting that the imbalanced sensory input is not a common trait of the dyslexic population, but characterizes a specific group of subjects. Dyslexics who show consistent imbalance of their binocular visual input are found to benefit from larger interletter spacing when non-words are presented. At the moment it is difficult to explain why the correlation is not significant for words. It is possible, that, as hypothesized for unstable dominant dyslexics, the effect of the sensory imbalance is mild enough to be detectable only upon exclusive activation of the sublexical route. The sublexical route, in fact, requires sequential letter-by-letter or syllable-by-syllable processing, so that the reader cannot rely upon the anticipatory content of high frequency words.
Likewise, there is reason to believe that the strength of interocular suppression enhances the effect of the sensory imbalance in affecting the reading performance. When IBV and III are combined into a vector Vsi, the change of the interletter spacing on average accounts up to 57% of the variance of the reading rate of the sample with Vsi above the cutoff. To be noted that in the study of Cornelissen and colleagues suspected dyslexics who failed the Dunlop test read less accurately in binocular conditions compared to monocular viewing (73,96): this suggests, indeed, that interocular interference between the left/right sensory input (as well as unfixed reference due to unstable dominance) may affect reading.
In summary, our study suggests an effect of abnormal visual processing on reading fluency in terms of populations of dyslexics. Yet, the regression model-based procedure adopted in this investigation seems unsuitable to detect the individual cases, especially within the clinical setting: the reading rate of words or non-words, in fact, was sensitive to interletter spacing in no more than 23% of cases with abnormal SRA, 12% of cases with abnormal binocular sensory interaction (Vsi above the cutoff), and 50% of cases with unstable sighting dominance when non-words were presented. In turn, the positive marker was found in only one non-visuoperceptive case (4%).
Evidently, the variability in the reading performance of dyslexic children due to difference in environmental and psychological factors is not so high to affect the significant trends provided by the regression model applied on a population scale, but makes the trends unreliable on the individual scale. It follows that if our results support the existence of a class of visual dyslexics, a different approach is required to detect single cases of visual dyslexics with an acceptable degree of sensitivity and specificity.
In a recent paper of Zorzi and associates and of Perea and colleagues subtle changes in the inter-letter spacing helped dyslexic subjects read better (43,91). It is likely that the effect is due to the presence of “visual dyslexics” in their unselected samples of dyslexic readers. As a matter of fact, the “non-visual dyslexics” of our sample did not benefit from larger interletter distance, mimicking the behavior of the normal readers (25).
A last consideration is worth to be made about the relationship between what reported in this study and the so-called “letter position dyslexia” (LPD). LPD is a selective deficit in letter position encoding occurring at a pre-lexical stage of the orthographic visual analysis, in absence of phonological problems. This deficit manifests itself as word-interior letter migration (97). The diagnosis of LPD relies on the accuracy, that is on the number of positional errors. Actually, the effect of the visual functions considered in this paper is based on the reading rate, so that it is difficult to establish if there is a relation between LPD and the visual-perceptive alterations discussed here. Anyway, unstable fixation due to unstable dominance might lead to positional errors. Similarly, if anisotropy of the visual space affects crowding, hampered letter decoding will be expected causing reduced accuracy. The same could stem from abnormal interocular inhibition. In all cases widening the distance between characters could (at least partly) prevent from positional errors due to letter migration. Yet, a distinctive trait of LPD is that the migration does not affect all the characters of the word, but involves selectively the interior letters: evidently, this is a feature that cannot be addressed simply under the visual-perceptive perspective considered in this paper.
Conclusions
This exploratory study suggests, with due caution, the existence of a type of dyslexia whose lexical impairment is causally related to a variety of specific and quantifiable defects of the visual channel, namely increased SRA, abnormal binocular temporal sensory interaction and, to a less extent, unstable ocular dominance. This finding helps explain why remediation protocols based on treatment of phonological deficits are not entirely successful (98,99).
Without doubt, our preliminary results need confirmation, and other visual factors are expected to be causally involved in the reading disability: of these, visual attention has been extensively studied in the past years (100,101) and deserves particular consideration.
As reading disability is considered a multidisciplinary condition, the relationship between these visual impairments and dyslexia is not necessarily bi-univocal: so, it cannot be assumed that most people with the visual alterations like those considered in this study are necessarily dyslexics.
Even if our line of investigation seems promising to delineate trends of visuoperceptive impairment in terms of classes of dyslexics, the detection of the individual cases in clinical practice requires a different approach. Additional research, therefore, is needed to better differentiate between visual dyslexics and non-visual dyslexics, as well as to detect individual cases of visual dyslexia with acceptable sensitivity and specificity. Within a practical frame, this distinction is crucial for orienting specific rehabilitation programs.
This study, evidently, does not pretend to draw definitive conclusions, but opens up new directions of investigation, advancing a novel approach to address the issue of vision and dyslexia and suggesting a tentative operational definition of “visual dyslexic”.
Acknowledgments
Funding: None.
Footnote
Data Sharing Statement: Available at http://dx.doi.org/10.21037/aes-20-86
Peer Review File: Available at http://dx.doi.org/10.21037/aes-20-86
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes-20-86). Dr. CA reports personal fees from Eynext SRL for consulting and lectures, personal fees from TiPubblica, Lafeltrinelli, Milan, Italy for copyright as author of two books on dyslexia and vision, outside the submitted work. The other author has no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional ethics board of the University of Turin as a degree Thesis (No. 754741/10.11.2016). As a retrospective study performed on an existing database, no informed consent was necessary.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Borsting E, Ridder WH, Dudeck K, et al. The presence of a magnocellular defect depends on the type of dyslexia. Vis Res 1996;36:1047-53. [Crossref] [PubMed]
- Cornelissen P, Richardson A, Mason A, et al. Contrast sensitivity and coherent motion detection measured at photopic luminance levels in dyslexics and controls. Vision Res 1995;35:1483-94. [Crossref] [PubMed]
- Lovegrove WJ, Bowling A, Badcock D, et al. Specific reading difficulty: differences in contrast sensitivity as a function of spatial frequency. Science 1980;210:439-40. [Crossref] [PubMed]
- Lovegrove W, Martin F, Bowling A, et al. Contrast sensitivity functions and specific reading disability. Neuropsychologia 1982;20:309-15. [Crossref] [PubMed]
- Lovegrove WJ, Martin F, Slaghuis W. A theoretical and experimental case for a visual deficit in specific reading disability. Cogn Neuropshychol 1986;3:225-67. [Crossref]
- Slaghuis WL, Ryan JF. Spatio-temporal contrast sensitivity, coherent motion, and visible persistence in developmental dyslexia. Vis Res 1999;39:651-68. [Crossref] [PubMed]
- Talcott JB, Hansen PC, Willis-Owen C, et al. Visual magnocellular impairment in adult developmental dyslexics. Neuro-Ophthalmology 1998;20:187-201. [Crossref]
- Hansen PC, Stein JF, Orde SR, et al. Are dyslexics’ visual deficits limited to measures of dorsal stream function? Neuroreport 2001;12:1527-30. [Crossref] [PubMed]
- Talcott JB, Witton C, McLean MF, et al. Dynamic sensory sensitivity and children's word decoding skills. Proc Natl Acad Sci U S A 2000;97:2952-7. [Crossref] [PubMed]
- Badcock DR, Lovergrove WJ. The effect of contrast, stimulus duration and spatial frequency on visible persistence in normal and specifically disabled readers. J Exp Psychol Hum Percept Perform 1981;7:495-505. [Crossref]
- Di Lollo V, Hanson D, McIntyre JS. Initial stages of visual information processing in dyslexia. J Exp Psychol Hum Percept Perform 1983;9:923-35. [Crossref] [PubMed]
- Slaghuis WL, Lovegrove W. Flicker masking of spatial-frequency-dependent visible persistence and specific reading disability. Perception 1984;13:527-34. [Crossref] [PubMed]
- Winters RL, Patterson R, Shontz W. Visual persistence and adult dyslexia. J Learn Disabil 1989;22:641-5. [Crossref] [PubMed]
- Skottun BC. The magnocellular deficit theory of dyslexia: the evidence from contrast sensitivity. Vis Res 2000;40:111-27. [Crossref] [PubMed]
- Olulade OA, Napoliello EM, Eden GF. Abnormal visual motion processing is not a cause of dyslexia. Neuron 2013;79:180-90. [Crossref] [PubMed]
- Taroyan NA, Nicolson RI, Buckley D. Neurophysiological and behavioural correlates of coherent motion perception in dyslexia. Dyslexia 2011;17:282-9. [Crossref] [PubMed]
- Legge GE, Rubin GS, Luebker A. Psychophysics of reading--V. The role of contrast in normal vision. Vision Res 1987;27:1165-77. [Crossref] [PubMed]
- Martin F, Lovegrove WJ. The effects of field size and luminance on contrast sensitivity differences between specifically reading disabled and normal children. Neuropsychologia 1984;22:73-7. [Crossref] [PubMed]
- Cornelissen PL, Hansen PC, Gilchrist I, et al. Coherent motion detection and letter position encoding. Vision Res 1998;38:2181-91. [Crossref] [PubMed]
- Talcott JB, Hansen PC, Assoku EL, et al. Visual motion sensitivity in dyslexia: evidence for temporal and energy integration deficits. Neuropsychologia 2000;38:935-43. [Crossref] [PubMed]
- Iles J, Walsh V, Richardson A. Visual search performance in dyslexia. Dyslexia 2000;6:163-77. [Crossref] [PubMed]
- Raymond JE. Attentional modulation of visual motion perception. Trends Cogn Sci 2000;4:42-50. [Crossref] [PubMed]
- McConkie GW, Zola D. Some characteristics in readers’ eyes movements. In: von Euler C, Lundberg I, Lennerstrand G. editors. Brain and reading. London: Macmillian Press, 1989.
- Skottun BC, Skoyles JR. Dyslexia and rapid visual processing: a commentary. J. Clin Exp Neuropsychol 2008;30:666-73. [Crossref] [PubMed]
- Aleci C, Piana G, Piccoli M, et al. Developmental dyslexia and spatial relationship perception. Cortex 2012;48:466-76. [Crossref] [PubMed]
- Atkinson J. Review of human visual development: crowding and dyslexia. In: Stein JF. editor. Vision and visual dyslexia. London: MacMillian Press, 1991:44-77.
- Atkinson J. Vision in dyslexics: Letter recognition, acuity, visual crowding, contrast sensitivity, accommodation, convergence and sight reading music. In: Wright SF, Groner R. editors. Facets of dyslexia and its remediation. Amsterdam: Elsevier, 1993:125-38.
- Bouma H, Legein CHP. Foveal and parafoveal recognition of letters and words by dyslexics and by average readers. Neuropsychologia 1977;15:69-80. [Crossref] [PubMed]
- Geiger G, Lettvin JY. Peripheral vision in persons with dyslexia. N Engl J Med 1987;316:1238-43. [Crossref] [PubMed]
- Martelli M, Di Filippo G, Spinelli D, et al. Crowding, reading, and developmental dyslexia. J Vis 2009;9:14.1-18.
- Spinelli- D, De Luca M, Judica A, et al. Crowding effects on word identification in developmental dyslexia. Cortex 2002;38:179-200. [Crossref] [PubMed]
- Riddell P, Fowler MS, Stein JF. A comparison of sighting dominance and the Dunlop Test reference eye in reading disabled children. Br Orthopt J 1987;44:64-9.
- Stein JF, Riddell PM, Fowler S. Fine binocular control in dyslexic children. Eye 1987;1:433-8. [Crossref] [PubMed]
- Stein JF, Fowler S. Visual dyslexia. Trends Neurosci 1981;4:77-80. [Crossref]
- Stein JF, Fowler S. Unstable binocular control in children with specific reading retardation. J Res Read 1993;16:30-45. [Crossref]
- Stein JF, Riddell PM, Fowler S. Disordered vergence eye movement control in dyslexic children. Br J Ophthalmol 1988;72:162-6. [Crossref] [PubMed]
- Stein J, Walsh V. To see but not to read; the magnocellular theory of dyslexia. Trends Neurosci 1997;20:147-52. [Crossref] [PubMed]
- Aleci C, Cavaglià L, Piana G, et al. Domitest-S: a novel dichoptic technique to assess ocular sensory dominance in children. a population study. Br J Med Med Res 2014;4:1371-82. [Crossref]
- Aleci C, Blanc M, Canavese L. Detecting visuoperceptive defects in adult disabled readers with the TETRA Analyzer™. Normative data and test-retest reliability. Ophthalmology Research: An International Journal 2017;7:1-13.
- Blake R, Logothetis N. Visual competition. Nat Rev Neurosci 2002;3:13-21. [Crossref] [PubMed]
- Bouma H. Interaction effects in parafoveal letter recognition. Nature 1970;226:177-8. [Crossref] [PubMed]
- Hawelka S, Wimmer H. Impaired visual processing of multi-element arrays is associated with increased number of eye movements in dyslexic reading. Vis Res 2005;45:855-63. [Crossref] [PubMed]
- Zorzi M, Barbiero C, Facoetti A, et al. Extra-large letter spacing improves reading in dyslexia. Proc Natl Acad Sci USA 2012;109:11455-9. [Crossref] [PubMed]
- Vernet M, Kapoula Z. Binocular motor coordination during saccades and fixations while reading: a magnitude and time analysis. J Vis 2009;9:2. [Crossref] [PubMed]
- Stein JF. Visual dyslexia. Trends Neurosci 1981;4:77-80. [Crossref]
- Stein JF, Fowler S. Diagnosis of dyslexia by means of a new indicator of eye dominance. Br J Ophthalmol 1982;66:332-6. [Crossref] [PubMed]
- Newman SP, Wadsworth JF, Archer R, et al. Ocular dominance, reading, and spelling ability in school children. Br J Ophthalmol 1985;69:228-32. [Crossref] [PubMed]
- Bigelow ER, McKenzie B. Unstable ocular dominance and reading ability. Perception 1985;14:329-35. [Crossref] [PubMed]
- Dunlop P, Banks EM. New binocular factors in reading disability. Australian Orthoptic Journal 1974;37:11-5.
- Jainta S, Kapoula Z. Dyslexic children are confronted with unstable binocular fixation while reading. PLoS One 2011;6:e18694 [Crossref] [PubMed]
- Mason A, Cornelissen P, Fowler S, et al. Contrast sensitivity, ocular dominance and specific reading disability. Clin Vis Sci 1993;8:345-53.
- Bishop DV, Jancey C, Steel AM. Orthoptic Status and reading disability. Cortex 1979;15:659-66. [Crossref] [PubMed]
- Cornelissen P. Fixation, contrast sensitivity and children’s reading. In: Wright SF, Groner R. editors. Facets of dyslexia and its remediation. Amsterdam: Elsevier, 1993:139-62.
- Evans BJW, Drasdo N, Richards IL. Investigation of accommodative and binocular function in dyslexia. Ophthalmic Physiol Opt 1994;14:5-19. [Crossref] [PubMed]
- Zeri F, De Luca M, Spinelli D, et al. Ocular dominance stability and reading skill: A controversial relationship. Optom Vis Sci 2011;88:1353-62. [Crossref] [PubMed]
- Dunlop P. Dyslexia. The orthoptic approach. Australian Orthoptic Journal 1972;12:16-20.
- Bishop DV. Unfixed reference, monocular occlusion, and developmental dyslexia-a critique. Br J Ophthalmol 1989;73:209-15. [Crossref] [PubMed]
- Stein JF. The Dunlop test and reading in primary school children. Br J Ophthalmol 1986;70:317-20. [Crossref] [PubMed]
- Evans BJW. Dyslexia and Vision. London: Whurr Publishers, 2001.
- Barbeito R. Sighting dominance: An explanation based on the processing of visual direction in tests of sighting dominance. Vis Res 1981;21:855-60. [Crossref] [PubMed]
- Barbeito R, Simpson TL. The relationship between eye position and egocentric visual direction. Percept Psychophys 1991;50:373-82. [Crossref] [PubMed]
- Mapp AP, Ono H, Barbeito R. What does the dominant eye dominate? A brief and somewhat contentious review. Percept Psychophys 2003;65:310-7. [Crossref] [PubMed]
- van Leeuwen AF, Collewijn H, Erkelens CJ. Dynamics of horizontal vergence movements: interaction with horizontal and vertical saccades and relation with monocular preferences. Vis Res 1998;38:3943-54. [Crossref] [PubMed]
- Coren S, Kaplan CP. Patterns of ocular dominance. Am J Optom Arch Am Acad Optom 1973;50:283-92. [Crossref] [PubMed]
- Gronwall DMA, Sampson H. Ocular dominance: a test of two hypotheses. Br J Psychol 1971;62:175-85. [Crossref] [PubMed]
- Porac C, Coren S. The dominant eye. Psychol Bull 1976;83:880-97. [Crossref] [PubMed]
- Rice ML, Leske DA, Smestad CE, et al. Results of ocular dominance testing depend on assessment method. J AAPOS 2008;12:365-9. [Crossref] [PubMed]
- Seijas O, Gómez de Liaño P, Gómez de Liaño R, et al. Ocular dominance diagnosis and its influence in monovision. Am J Ophthalmol 2007;144:209-16. [Crossref] [PubMed]
- Gould G. A method of determining ocular dominance. JAMA 1910;55:360-70.
- Miles WR. Ocular dominance demonstrated by unconscious sighting. J Exp Psychol 1929;12:113-26. [Crossref]
- Miles WR. Ocular dominance in human adults. J Gen Psychol 1930;3:412-29. [Crossref]
- Walls GL. A theory of ocular dominance. AMA Arch Ophthalmol 1951;45:387-412. [Crossref] [PubMed]
- Cornelissen P, Bradley L, Fowler S, et al. What children see affects how they read. Dev Med Child Neurol 1991;33:755-62. [Crossref] [PubMed]
- Baker DH, Meese TS, Summers RJ. Psychophysical evidence for two routes to suppression before binocular summation of signals in human vision. Neuroscience 2007;146:435-48. [Crossref] [PubMed]
- Fox R, Rasche F. Binocular rivalry and reciprocal inhibition. Percept Psychophys 1969;5:215-7. [Crossref]
- Hess RF, Hutchinson CV, Ledgeway T, et al. Binocular influences on global motion processing in the human visual system. Vis Res 2007;47:1682-92. [Crossref] [PubMed]
- Li J, Lam CS, Yu M, et al. Quantifying sensory eye dominance in the normal visual system: a new technique and insights into variation across traditional tests. Invest Ophthalmol Vis Sci 2010;51:6875-81. [Crossref] [PubMed]
- Handa T, Mukuno K, Uozato H, et al. Effects of dominant and nondominant eyes in binocular rivalry. Optom Vis Sci 2004;81:377-83. [Crossref] [PubMed]
- Mitchell JF, Stoner GR, Reynolds JH. Object-based attention determines dominance in binocular rivalry. Nature 2004;429:410-3. [Crossref] [PubMed]
- Ooi TL, He ZJ. Sensory eye dominance. Optometry 2001;72:168-78. [PubMed]
- Valle-Inclán F, Blanco MJ, Soto D, et al. A new method to assess eye dominance. Psicológica 2008;29:55-64.
- Kommerell G, Schmitt C, Kromeier M, et al. Ocular prevalence versus ocular dominance. Vis Res 2003;43:1397-403. [Crossref] [PubMed]
- Yang E, Blake R, McDonald JE. A new interocular suppression technique for measuring sensory eye dominance. Invest Ophthalmol Vis Sci 2010;51:588-93. [Crossref] [PubMed]
- Raymond JE, Shapiro KL, Arnell KM. Temporary suppression of visual processing in an RSVP task: An attentional blink? J Exp Psychol Hum Percept Perform 1992;18:849-60. [Crossref] [PubMed]
- Aleci C, Piana G, Anselmino F. Evaluation of spatial anisotropy by curvature analysis of elliptical targets. Open Ophthalmol J 2010;4:15-21. [Crossref] [PubMed]
- Huckauf A, Heller D, Nazir TA. Lateral masking: limitations of the feature interaction account. Percept Psychophys 1999;61:177-89. [Crossref] [PubMed]
- Huckauf A, Heller D. What various kinds of errors tell us about lateral masking effects. Vis Cogn 2002;9:889-910. [Crossref]
- Stanley G, Hall R. Short term visual processing in dyslexics. Child Dev 1973;44:841-4. [PubMed]
- Cornoldi C, Colpo G. La verifica dell’apprendimento della lettura. Firenze: Organizzazioni Speciali, 1981.
- Chung STL. The effect of letter spacing on reading speed in central and peripheral vision. Invest Ophthalmol Vis Sci 2002;43:1270-6. [PubMed]
- Perea M, Panadero V, Moret-Tatay C, et al. The effect of inter-letter spacing in visual-word recognition: Evidence with young normal readers and developmental dyslexics. Learn Instr 2012;22:420-30. [Crossref]
- Perea M, Gomez P. Increasing interletter spacing facilitates encoding of words. Psychon Bull Rev 2012;19:332-8. [Crossref] [PubMed]
- Davis CJ. The spatial coding model of visual word identification. Psychol Rev 2010;117:713-58. [Crossref] [PubMed]
- Gomez P, Ratcliff R, Perea M. The overlap model: a model of letter position coding. Psychol Rev 2008;115:577-600. [Crossref] [PubMed]
- Cornelissen P, Munro N, Fowler S, et al. The stability of binocular fixation during reading in adults and children. Dev Med Child Neurol 1993;35:777-87. [Crossref] [PubMed]
- Cornelissen P, Bradley L, Fowler S, et al. Covering one eye affects how some children read. Dev Med Child Neurol 1992;34:296-304. [Crossref] [PubMed]
- Friedmann N, Gvion A, Nisim R. Insights from letter position dyslexia on morphological decomposition in reading. Front Hum Neurosci 2015;9:143. [Crossref] [PubMed]
- Lyon GR, Moats LC. Critical conceptual and methodological considerations in reading intervention research. J Learn Disabil 1997;30:578-88. [Crossref] [PubMed]
- Torgesen JK. Individual differences in response to early interventions in reading: The lingering problem of treatment resisters. Learning disabilities Research & Practice 2000;15:55-64. [Crossref]
- Moores E, Tsouknida E, Romani C. Adults with dyslexia can use cues to orient and constrain attention but have a smaller and weaker attention spotlight. Vis Res 2015;111:55-65. [Crossref] [PubMed]
- Roach NW, Hogben JH. Spatial cueing deficits in dyslexia reflect generalised difficulties with attentional selection. Vis Res 2008;48:193-207. [Crossref] [PubMed]
Cite this article as: Aleci C, Belcastro E. Visual dyslexia: towards an operational definition from a correlational study. Ann Eye Sci 2020;5:32.



