
Treatment of congenital ptosis
Introduction
Blepharoptosis, or simply known as “ptosis” (from the Greek word “to fall”), is defined as an abnormally low position of the upper eyelid, with respect to the visual axis in the primary gaze (Figure 1). It can be bilateral, unilateral or asymmetric in appearance. Furthermore, it may be an isolated finding or part of a constellation of signs of specific systemic associations. Depending on how much it interferes with the visual axis, it may be considered either as a functional (affecting vision) or mainly a cosmetic problem. In childhood, the finding of ptosis takes on a greater significance, as it may lead to deprivation amblyopia or astigmatism. The younger the child, the more urgent it is to relieve the obstruction by surgery. However, even a non-amblyogenic ptosis may need correction to avoid later psychosocial effects.
The saying that modern facial surgery has evolved into the fusion of medicine and art is true even regarding ptosis correction. The level of surgical sophistication developed over the centuries has been refined to today’s standards by parallel advances in anatomy, physiology and pathology and the development of new materials. The first documented references to rudimentary ptosis surgery date back to ancient Arabian ophthalmologists. Later during the 1st century AD in the Greek and Roman times, eyelid surgery was mentioned in the writings of Aulus C. Celsus in De Medicina (1). Later on, throughout out the ages, further attempts at ptosis correction (as well as eyelid reconstruction) were generally poorly described or undocumented, until the early 19th century. Starting with the publication of a treatise on diseases of the eye by the Italian anatomist/surgeon Antonio Scarpa in 1806, the different approaches to eyelid surgery seemed to unify. Since then, there has been a wealth of publications, which ushered in a new era of modern ptosis and eyelid surgery. In 1956, Crawford published a landmark paper on congenital ptosis correction using fascia lata and, in 1961, the Fasanella-Servat procedure was described, which has remained a popular option to this day. The founding of the American Society of Ophthalmic Plastic Surgery (ASOPRS) in 1969, marks the time when further research and publications on ptosis surgery began to truly flourish. Putterman improved on the Müller muscle-conjunctival resection (MMCR) technique by developing the Putterman clamp in 1972. Further interest was also generated in the use of different frontalis sling materials, in addition to fascia lata. As interest in anatomic studies was renewed, the age of levator aponeurosis surgery was born during the 1970’s and gained wide popularity. Championed by Anderson and others, the next decade ushered in the Age of Aponeurotic Awareness. To this day, aponeurosis repair along with frontalis sling remain as of the most common ptosis operations worldwide.
The basics: workup and diagnosis
In order to carry out an effective and focused examination, the clinician must keep in mind a short list of differential diagnoses for a child presenting with a ptotic eyelid.
Differential diagnosis of ptosis
- Isolated congenital ptosis.
- Ptosis associated with an ocular or systemic syndrome.
- Edema, cellulitis, allergy, giant papillary conjunctivitis (GPC).
- Eyelid masses (e.g., plexiform neurofibroma).
- Orbital conditions presenting with lid swelling or mass (orbital inflammation, orbital cellulitis, dacryoadenitis and tumours such as rhabdomyosarcoma).
- Pseudoptosis (contralateral lid retraction, enophthalmos, microphthalmos, hypotropia).
- Birth trauma.
Surgical anatomy
The eyelids are complex soft tissue structures covering the orbital aperture and serve the function of protecting the globe and distributing tears over the cornea. They also play an essential role in facial expressions and social interactions.
The upper eyelid retractors consist of the levator muscle with its aponeurosis (primary elevator) and the Müller muscle (aka superior tarsal muscle; secondary elevator) (Figure 2). Levator muscle originates at the orbital apex and runs anteriorly near the roof of the orbit as the muscular portion. As it reaches the opening of the orbit, it is invested by Whitnall’s ligament, and changes it course to a downward direction to transition into an aponeurosis (which is about 14–20 mm in length). It then travels inferiorly to insert into the anterior lower tarsus. Whitnall’s ligament is a horizontal band of fibrous tissue, which functions as a fulcrum and support for the levator. The tarsal plate, made of dense connective tissue, provides structural rigidity to the eyelid. Just above the point of attachment to the tarsus, the aponeurosis sends some fibres anteriorly into the orbicularis and skin, which gives rise to the eyelid crease externally. The eyelid fold of the loose preseptal skin is found above it. The orbital septum separates the orbital compartment from eyelid tissue anteriorly. It attaches to the levator aponeurosis in the upper eyelid about 2–5 mm above the tarsus. Anterior to the aponeurosis is orbital fat (the pre-aponeurotic fat pad), which is held back by the septum. This is an important anatomic landmark during ptosis repair. The septum becomes attenuated with age, but in children it is quite thick and may somewhat obscure the fat pad. In the Asian population, insertion of the septum is much lower, resulting in a lower position of the pre-aponeurotic fat pad, a lower lid crease and a fuller appearance to the eyelid. Innervation of the levator muscle is by the superior division of the third cranial nerve which also supplies the superior rectus muscle (2). At the upper edge of tarsus is the insertion of Müller’s muscle, which originates from the undersurface of the levator muscle, about 12–14 mm above tarsus. It provides about 2–3 mm of elevation of the lid via sympathetic nerve supply. Interruption of the sympathetic chain results in Horner’s syndrome, which presents as a mild ptosis along with other features.

Posteriorly, tarsus and Müllers muscle are covered by the conjunctiva which continues into the superior fornix and onto the globe. Anteriorly, all these structures are encircled by a layer of orbicularis muscle, the main protractor of both eyelids. It is supplied by the seventh cranial nerve and plays an important role in protecting the eye by closing the eyelids. The outer layer is a thin layer of subcutaneous tissue and skin. The upper eyelid has two arterial arcades, one just superior to the lash line and one near the upper edge of tarsus, which is a useful surgical landmark.
History and examination
While the presence of ptosis may be obvious, history and physical examination are necessary to establish the exact etiology and to plan surgery. The first goal is to confirm whether the ptosis is an isolated finding or part of collection of clinical signs of an associated condition. The age of onset, character, duration, progression or intermittent nature are some of the important questions asked. Systemic, family and birth history will also help with the diagnosis, as well as any past drug or allergic reactions.
On examination, the face is first observed as a whole, looking for any abnormal head posture (e.g., chin-up position), and active recruitment of frontalis muscle. While in a young child, it may be challenging to obtain accurate measurements, we still attempt to measure the palpebral fissure (PF), margin-reflex distance (MRD1 and MRD2) and levator function (LF) (Figure 3). The PF is the mid-pupil distance between the upper and lower lid margin with the eyes in the primary position. The MRD1 is the measurement between the upper eyelid margin and the light reflex in the center, while MRD2 is the distance between the light reflex and the lower eyelid margin. LF is measured by immobilizing the brow with your thumb and the excursion between maximal upgaze and downgaze is recorded. Bell’s phenomenon and lagophthalmos are also noted. Palpation of the eyelid is done to rule out any upper eyelid or orbital masses. A slit-lamp exam looks at the status of the cornea and anterior segment. The pupils, iris colour, extraocular movements or strabismus are also noted. Lastly, a cycloplegic refraction, looking for astigmatism or anisometropia, and fundoscopy conclude the exam.
In children with ptosis, auxiliary tests (blood tests or imaging) are usually not necessary, unless there are associated neurologic findings, a new onset of Horner’s syndrome or third nerve palsy, or suspicion of a mass or recent trauma. Often parents will bring their own photographs from home, which are useful in assessment.
Congenital ptosis and systemic associations
Types of ptosis
- Isolated congenital (simple) ptosis.
- Associations/syndromes:
- Marcus-Gunn jaw-wink syndrome.
- Blepharophimosis-ptosis-epicanthus inversus syndrome (BPES).
- Congenital third cranial nerve palsy.
- Congenital Horner’s syndrome.
- Congenital fibrosis of the extraocular muscles (CFEOM).
- Congenital myasthenic syndromes.
- Chronic progressive external ophthalmoplegia (CPEO).
- Monocular elevation deficiency.
As in adults, congenital ptosis may be classified correspondingly into myogenic, neurogenic, mechanical and traumatic causes. Aponeurotic ptosis is rare in children but can occur, likely secondary to birth trauma. By far the majority of congenital ptosis is unilateral and an isolated maldevelopment of the levator muscle. Congenital ptosis can also occur in conjunction with other ophthalmic and/or systemic findings (1,3-8). Some more common syndromes are mentioned below.
Isolated congenital ptosis
Congenital ptosis is a triad of weak lid crease, downgaze lid lag and decreased LF. It is usually present at birth or manifests within the first year of life. It may be mild to severe and with a variable degree of levator dysfunction. In addition, there is also a significant lagophthalmos on downgaze with exposure of the cornea. There is often a decreased or absent lid crease (Figure 4). If the ptotic eyelid partially or fully occludes the visual axis, then the child is at great risk of developing deprivation amblyopia and urgent surgical correction is indicated. In cases where the visual axis is not fully covered, the child may adopt a chin-up head position or there may be induced astigmatism (3,4).
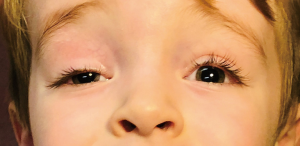
Isolated congenital ptosis occurs equally between genders and there is no racial predilection. The majority are unilateral. The exact incidence of congenital ptosis is unknown, but a large long-term study in the US reported it to be about 7.9 per 100,000 children (5). Of these, 90% were classified as isolated simple congenital ptosis. Several genes have been implicated, including autosomal dominant inheritance and X-linked dominant inheritance (6). The underlying etiology appears to be localized muscle dysgenesis. In the affected levator, normal muscle fibres are infiltrated or replaced with fibrous and adipose tissue, which results in abnormal contraction and relaxation (7,8). Intraoperatively, it feels as an inelastic muscle, which corresponds to poor function on clinical exam (9). Although it is classically considered myogenic in nature, recent reports have suggested that disordered innervation during embryonic development may be the underlying cause (10). Maldevelopment of oculomotor caudal central subnucleus motor neurons or their axons have been implicated (9,11).
Surgical implication: correction of isolated congenital ptosis is treated by direct levator muscle (or Müller muscle) resection or a frontalis suspension.
Congenital ptosis with associated anomalies
Marcus-Gunn jaw-wink syndrome
This condition usually presents as a unilateral congenital ptosis, with an associated synkinetic retraction of the ptotic eyelid associated with movement of the mandible. The most common jaw movement that triggers the eyelid elevation is lateral motion, to the contralateral side (Figure 5). It is noted at a very early age by the mother, as a retraction movement (winking) during breast feeding. The ptosis may have poor to good LF. If the ptosis is minimal with no threat to visual function, it may be appropriate to observe until older. The older child may learn to mask the synkinetic movement by avoiding certain jaw positions and it also seems to lessen with time. It is thought to be caused by an aberrant connection between the motor division of the fifth cranial nerve and the levator muscle (pterygoid-levator synkinesis), but the exact pathway is yet to be elucidated. One study detected activation of the proprioceptive receptors of the pterygoid muscles by electromyography (EMG) during levator activation (12).
Duane’s syndrome may also occasionally exhibit similar synkinetic movement of the upper eyelid but should be easily distinguished from the classic jaw-wink syndrome. Rarely, there may be other abnormal connections between the third nerve and other cranial nerves, along with weakness of the superior rectus muscle (13).
Surgical implications: the ptosis may have poor to fair LF and is often difficult to measure due to the wink. If surgery is indicated and the wink is a significant component, then the only way to eliminate it is to disinsert the levator muscle, followed by a frontalis suspension. If the jaw-wink is not large, then a ptosis correction alone will suffice.
Blepharophimosis-ptosis-epicanthus inversus syndrome (BPES)
The BPES, also called the blepharophimosis syndrome, is an autosomal dominant congenital anomaly classically consisting of four features. Namely, severe bilateral symmetric ptosis (with poor LF), epicanthus inversus, telecanthus and shortening of both horizontal and vertical fissure distances (blepharophimosis) (Figure 6). The severity of ptosis usually results in a chin-up position. Additionally, there may be associated lower lid ectropion (with anterior lamella deficiency), hypoplasia of the superior orbital rim, ear deformities, flat nasal bridge and/or hypertelorism and few other rare findings (14). BPES is broadly divided into types I and II, with type I having associated early primary ovarian failure and infertility. The syndrome has been found to be due to a mutation on chromosome 3q23, in the FOXL2 gene, which is expressed in the developing eye and ovary. About 75% of patients have relatives with the same mutation and 25% may appear a new mutation or sporadic (14,15).
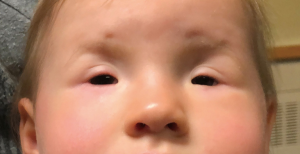
Surgical implications: due to significant risk of amblyopia, the most urgent anomaly to address is the ptosis. LF is often poor, therefore a frontalis sling is the procedure of choice. The other eyelid findings are less urgent, can be safely delayed and some may lessen with facial growth. Surgery would include correction of the telecanthus and epicanthus inversus. A variety of medial canthoplasties have been described, which generally involve combinations of flaps and Z-plasties. Occasionally, transnasal wiring is necessary.
Congenital third cranial nerve palsy
Congenital oculomotor palsy may be partial or complete and is thought be caused by either birth trauma or occur as a developmental abnormality. The presenting feature is ptosis, with varying degrees of ophthalmoplegia, consisting of an inability to fully depress, adduct and elevate. The pupil is mostly dilated, but rarely may also be constricted due to aberrant regeneration. A rare but related condition is the so-called cyclic oculomotor paresis, which involves alternating cycles of unilateral third nerve paresis and periodic spasms causing eyelid elevation, miosis, and contracture of the muscles supplied by the third nerve (16).
Surgical implications: the LF is usually severely decreased requiring a frontalis sling operation, which carries a great risk of lagophthalmos, unmasking of diplopia and exposure keratopathy due to poor supraduction. Management is usually done in conjunction with a strabismus specialist.
Congenital Horner’s syndrome
This syndrome is due to an innervational deficit of the Müller muscle, resulting in mild ptosis. Associated miosis, anhidrosis and heterochromia are some of the other findings (17). The pupillary asymmetry is best seen in low lighting. Sometime, the sympathetic denervation of the affected side of the face, may be noticed as a difference in the colour of the cheeks when the baby cries. This is sometimes referred to as a Harlequin Baby (17). Mild elevation of the lower eyelid (reverse ptosis) contributes to a decreased PF which may give the appearance of enophthalmos. The most frequent cause is likely birth trauma involving the brachial plexus. Other rarer causes are thoracic or cervical masses such as neuroblastoma or sympathetic dysgenesis. Because of interruption of sympathetic innervation to the iris during development, a hypopigmented colour of the iris (heterochromia) is often seen (17,18).
Surgical implications: because the etiology of Horner’s ptosis is due to Müller muscle denervation, surgical procedures aimed directly at the Müller muscle are considered most effective.
Congenital fibrosis of the extraocular muscles (CFEOM)
This is a non-progressive congenital condition presenting with ptosis and restrictive strabismus on all directions of gaze. A chin-up head position is a common finding (19). It appears to be caused by defective innervation of the muscles during development resulting in fibrotic changes. The underlying cause is presumed to be agenesis of ocular motor neurons in the brainstem (20).
It is classified into four phenotypes according to presentation, variability and genetics. Two of these have autosomal dominant inheritance, with CFEOM-1 exhibiting bilateral ptosis and ophthalmoplegia, while CFEOM-3 shows more variability, with unilateral ptosis, mild ophthalmoplegia and incomplete penetrance. Both are thought to be due to missense mutations in the KIF21A gene. The Tukle syndrome is basically the same as phenotype CFEOM-3, with the additional findings of oligodactyly or oligosyndactyly. The last type is CFEOM-2 and differs from the preceding by autosomal recessive inheritance and the genetic mutation in the PHOX2A gene. This syndrome falls under the umbrella term congenital cranial dysinnervation disorders (CCDD) (6), which also include a number of related conditions such as Duane’s syndrome, Möbius syndrome and hereditary congenital facial paresis (21).
Surgical implications: surgical rehabilitation requires the expertise of both a strabismologist and an oculoplastic surgeon and is a challenging condition to manage.
Chronic progressive external ophthalmoplegia (CPEO)
While CPEO is more commonly seen in adulthood, it can also present in the first to second decade. It is a rare form of maternally transmitted myogenic ptosis with mitochondrial inheritance caused by deletions in mitochondrial DNA (22). The presentation is bilateral ptosis with limitation of ocular movements secondary to progressive weakness of the extraocular muscles, accompanied by orbicularis weakness. This is a progressive disorder eventually leading to the eyes being fixed in the primary gaze. The myopathy appears to consist of ragged red fibers and atrophy on pathology (23). When there are other findings such as pigmentary retinopathy, heart block and cerebellar ataxia, the condition is labelled Kearns-Sayre syndrome. All patients with this syndrome need cardiologic evaluation, as it can result in a fatal cardiac event.
Surgical implications: as with other myogenic causes with limitation of LF, the most common procedure of choice is a frontalis sling operation (24).
Monocular elevation deficiency
This condition, previously termed double-elevator palsy, demonstrates limitation of upward gaze with a hypotropia that is similar in adduction and abduction as well as the finding of ptosis. The underlying etiology is thought to be deficient innervation of the elevator muscles due to a monocular supranuclear gaze disorder. It is characterized by hypotropia of the involved eye with limited elevation, a chin-up head position with binocular fusion in downgaze, and ptosis or pseudoptosis. True ptosis is present in 50% of patients (25).
Surgical implications: management is provided primarily by a strabismus surgeon. The ptosis can also be repaired, but lagophthalmos may be significant due to absent Bell’s phenomenon.
Ptosis surgery
There are two main types of procedures for congenital ptosis:
- Retractor muscle surgery (resection, advancement or plication):
- Anterior approach: levator muscle resection, advancement or pexy (lid crease incision).
- Posterior approach: MMCR, ± tarsus, ± levator, posterior levatorpexy.
- Frontalis sling suspension.
Whether the ptosis is isolated or part of a syndrome, the general surgical approach to the droopy eyelid itself is the same, albeit with special considerations. Of the two main techniques, one is aimed directly at the elevators of the eyelid (levator muscle and Müller muscle) and the other transfers the function to an accessory elevator, the frontalis muscle. The decision which technique to use is guided by the measurement of the levator excursion and the severity of ptosis, as well as surgeon’s experience, preference and training. The difficulty in obtaining accurate measurements in a child, as well as elasticity of tissues while estimating the amount of resection, may affect the final result.
In the past, it has been reported that there may be higher incidence of malignant hyperthermia (MH) in patients with strabismus or ptosis, but direct association is now considered disproven (26).
Timing of surgery
The goal of ptosis surgery is to address physical, functional and psychosocial concerns. If the ptosis is minimal, without risk of amblyopia, induced astigmatism or abnormal head posture, then surgery can be delayed. In this case, we wait until the child is older, more cooperative for measurements and the facial development has progressed (27). The recommended age is 4–5 years, as other children start noticing the asymmetry, which may lead to psychosocial problems (28). Furthermore, some authors feel that the long-term cosmetic and functional results are better, if surgery is carried out before the age of 4 years (29).
On the other hand, if the visual axis is affected, especially in the very young, then surgery is done on an urgent basis to prevent amblyopia. One study showed that about 15% of childhood patients diagnosed with congenital ptosis demonstrated amblyopia (30). Another factor to keep in mind is that ptosis may not be the sole cause of amblyopia. About one-fourth of congenital ptosis may have coexisting anisometropia or strabismus and long term follow up is recommended (31). Discussion with the parents about the risks and expectations are important, as they should be aware that the result may not be perfect and may need revision. One study showed a near 20% revision rate after surgery (32), but it is likely higher in infants.
Choice of surgical procedure: levator or Müller resection vs. frontalis sling
Because of the inherent inability to assess the final eyelid position intraoperatively, the decision of how much elevation is needed, as well as the surgical approach, must be made before surgery. This is based on two important measurements: the LF and the amount of ptosis. Normal LF is considered 14 mm or better. If the LF is equal or above 5 mm, then a levator resection surgery is the most commonly performed technique. If it is equal or below 4 mm, then a frontalis sling is carried out. Posterior Müller muscle approach is usually reserved for mild ptosis with near-normal LF. Beard classified the amount of decreased LF into three groups (Table 1) (33). Over time, these number have been slightly modified, but the principal concept remains the same. The amount of LF is the overriding factor in choosing the correct approach.
Table 1
| Eyelid excursion | LF |
|---|---|
| Normal | 14+ mm |
| Good | 8–12 mm |
| Fair | 5–7 mm |
| Poor | ≤4 mm |
LF, levator function.
The amount of ptosis based on its degree has also been ranked (Table 2) (34).
Table 2
| Grading | Amount of ptosis |
|---|---|
| Mild | 2 mm or less |
| Moderate | 3 mm |
| Severe | 4 mm or more |
Good to normal LF (8 mm or more)
In this group either an anterior levator resection/advancement or posterior MMCR or levatorpexy can be effective and a satisfactory outcome is achieved. Some authors use phenylephrine 2.5% drops to assess the Müller muscle response and thus determine if a MMCR would be preferred. Others believe that an anterior levator resection is more anatomically correct and can be tailored to both good and fair LF. In addition, it has the advantage of creating a lid crease, which is often faint or absent in congenital ptosis. It is also the procedure of choice in repeat surgeries. Lastly, plication of levator can also be achieved through a posterior approach, such as a posterior levatorpexy (35), which is more powerful than MMCR and also avoids an anterior scar.
Fair LF (5–7 mm)
In this category, a levator resection has become the standard choice and the anterior open-sky technique is the most widely used procedure. This approach provides excellent exposure to the levator, allowing for greater degree resection.
Poor LF (4 mm or less)
In this case, function of the eyelid elevation is transferred to the frontalis muscle with a sling procedure (36,37). The child quickly learns that raising the eyebrow will result in better vision, so it becomes a subconscious reflex. A potential problem may arise in cases of pre-existing poor vision or unilateral surgery, that there may not be enough stimulus to raise the eyebrow. Majority of surgeons agree that standard levator resection or MMCR has no role in this setting due to significant under correction.
Procedures
The following is a brief outline of important steps of the different ptosis procedures. For more comprehensive, detailed instructions, the reader is directed to many excellent instructional textbooks and publications, as well as numerous online videos on surgical techniques (38-40).
Anterior levator muscle resection
Indication
This approach is used when there is fair to good LF (5–12 mm) or better. Most popular method to estimate the amount of resection is shown in Table 3 (41,42). Another simple recommendation is to resect 4 mm of levator for every 1 mm of ptosis (6).
Table 3
| LF | Amount of resection |
|---|---|
| 8–12 mm | 10–13 mm |
| 6–8 mm | 16–18 mm |
| 4–6 mm | 22+ mm |
LF, levator function.
Technical notes
- Eyelid skin incision is done through the lid crease (or planned crease, if absent) (Figures 7,8
). - The levator muscle is resected by a pre-determined amount and then sutured directly to anterior tarsus.
- The dissection is carried up to the level of Whitnall’s ligament and fat is not resected in children.
- In children, the septum may be quite thick and obscure the preaponeurotic fat pad.
- The dysgenic levator muscle itself is often infiltrated with fat, which may make it difficult to identify
- The levator muscle is separated away from Müller muscle posteriorly before it’s shortened.
- A small strip of skin is excised from the upper edge of the wound and closed with a series of lid crease forming stitches.
- All sutures used are absorbable, to avoid stitch removal in children.
Posterior MMCR
Indication
The MMCR technique is most useful in cases where there is good to normal LF with mild to moderate ptosis, and there is good response to the phenylephrine test. The amount excised is usually between 6.5–9.5 mm (43), by the general rule of 4 mm conjunctival/Müller resection for each 1 mm of ptosis. This approach is felt to have the least chance of disrupting the lid margin contour. The drawbacks are that the lid crease is not formed, it is not repeatable, and resection of conjunctiva may worsen a dry eye. Also, it does not address the underlying anatomic problem of is levator maldevelopment (44).
Technical notes
- The eyelid is everted on a Desmarres retractor and the planned resection is marked out on the posterior side.
- The conjunctiva and the Müller muscle are clamped (e.g., Putterman ptosis clamp) and a suture is then passed under the clamp in a back and forth fashion.
- The tissues are then resected above the suture line, and the conjunctival layer closed.
- The suture can be tied and buried on the posterior side or brought out anteriorly to the skin and tied.
- If more elevation is required, a small amount of tarsus can be included in the resection (45).
- Care should be taken not to remove too much tarsus, which would destabilize the eyelid.
Although some surgeons still perform the Fasanella–Servat procedure, it involves an unnecessary excision of superior tarsal plate tissue and is not directed at correcting the actual pathology of dysgenic levator. It is reserved for management of up to 2.5 mm of congenital ptosis with normal (or near-normal) LF (46) (Figure 9).
Frontalis sling suspension
Indication
This procedure is exclusively done on patients with poor (4 mm or less) LF, which is usually accompanied by moderate to severe ptosis. The rule of thumb for intraoperative positioning is that with severe ptosis, the eyelid is set at the upper limbus and with moderate ptosis a two millimeters below (47). The main surgical decisions are which type of material is used and, secondly, which configuration of placement of the sling material is carried out (48-50) (Figures 10,11).
Technical notes
- The selected sling material is passed through pre-marked stab incisions in the eyelid and above the brow in the desired configuration.
- Most surgeons currently use either a simple rectangle or pentagon design.
- Attachment of the sling material to the tarsus can be done as an open sky procedure with a lid crease incision or a closed technique through stab incisions only (6,47).
- With the open technique, a narrow strip of skin can be excised along with creating a lid crease.
- Attention is paid to the post-septal placement of the sling, otherwise the sling may be visible and less effective.
- Both free ends exit in the brow, where the sling is tightened and tied.
- If simple rectangle does not give enough elevation, it can be converted to a pentagon by a 3rd stab incision.
- Aggressive lubrication needs to be applied in the postoperative period due to lagophthalmos, especially during sleep.
Materials
There are numerous types of materials implanted and new ones continue to be developed. The short- and long-term efficacy of the surgery, as well as the rate of complications, have been studied with respect the material (47). Due to its biocompatibility and longevity, autologous fascia lata was long considered the material of choice (51), but it has the disadvantage of a second surgical site and longer operation. Also, it cannot be used under the age of 4 due to incomplete development of fascia lata. Banked fascia lata is has been available for some time, but the long-term results appear to be less successful (52) and there is also a theoretical risk of disease transmission. Temporalis fascia and palmaris longus tendon are other examples of autologous materials that has been tried. Synthetic materials such as nylon, polyester, polypropylene, Supramid and polytetrafluoroethylene (Gore-Tex) have been used with varying good results, but silicone rod (tubing) has currently become the most popular (6). It is elastic, inert, and easy to implant or explant and adjust. Of note is that one study showed that regardless of the material used, there is a variable rate of recurrence (53) and all parents need to be aware of this.
Configuration of sling placement
Since the modern description of the frontalis sling technique by Crawford, multiple variations have emerged with respect subcutaneous placement. This may take the shape of a pentagon, single- or double- triangle, or even a simple rectangle or trapezoid. A successful result can likely be achieved with many of these designs. The pentagon or simple rectangle (trapezoid) seem to be currently the most favoured configurations (Figure 10). In the neonate, because of small anatomy, most surgeons will use a monofilament suture (2-0 or 3-0) in a simple rectangle fashion with good results.
Complications
The most common complications of congenital ptosis surgery are undercorrection and recurrence. Overcorrection in children is very rare, and it is more frequently found in adults. Some of the other post-operative complications include crease asymmetry, altered lid contour, PF asymmetry, tarsal eversion, prolapse of conjunctiva and lagophthalmos. Implanted materials for slings can undergo extrusion, infection, rejection, inflammation and pyogenic granuloma formation. If the sling is placed too shallow, it can result in external visibility or poor tarsal apposition (54). As in all surgeries, wound dehiscence, infection and scarring may occur. Attention also needs to be paid to pre-existing dry eye, corneal surface disease, seventh cranial nerve weakness (e.g., Möbius syndrome) and extraocular muscle abnormalities. All these may predispose the eye to a higher risk of lagophthalmos and corneal compromise.
Special considerations
Unilateral ptosis with poor LF
This situation presents a special challenge, as any type of procedure will likely not result in a perfect symmetrical appearance. The reason is that the operated side uses the frontalis muscle to elevate the eyelid, whereas the other side still has normal levator elevation. Several surgical options have been developed and are described briefly below. As mentioned, such discussion with parents is essential.
Unilateral sling. In this case, only the affected eye undergoes the operation and the normal side is left untouched. The postoperative symmetry is acceptable in the primary position, but becomes more noticeable with eye movement, especially on down gaze. With age, the asymmetry seems to decrease, and the child learns to camouflage the blemish. However in practice, this seem to be a common choice with satisfactory results (51).
Bilateral sling with disinsertion of levator muscle on the normal side. To achieve bilateral symmetry, an iatrogenic ptosis can be induced on the normal side and then a bilateral sling carried out (55). The disinserted levator is sometimes sutured to the orbital roof to completely disable it. Thereafter, both sides undergo a frontalis sling operation, which will result in greater symmetry (14). The downside is that the initially normal eyelid has to undergo surgery, and this is often resisted by parents.
Bilateral sling without levator muscle surgery on normal side. The same operation as above, with the exception of not disinserting the levator on the normal side. In other words, we introduce a sling into a normally functioning eyelid. The results are also more symmetrical than a unilateral sling, but may not be quite as good as levator extirpation (56).
Whitnall’s sling and supramaximal levator resection
Both of these techniques are basically extended levator resections. In the first case, the levator is resected up to Whitnall’s ligament, which is then directly sutured to the superior tarsus (57). In the second one, the levator dissection is carried well beyond the ‘normal’ range of large (25–30 mm) and beyond Whitnall’s ligament (58).
The Whitnall’s sling and supramaximal levator resection are usually reserved for situations, where a standard frontalis suspension is not desirable or expected to fail. The most common setting is due to frontalis weakness, such the Möbius and other deinnervation syndromes affecting the seventh nerve. In both of these techniques, one can also expect significant lagophthalmos with exposure.
Frontalis muscle flap
In place of a frontalis sling procedure, a unique technique of transplanting a flap of frontalis muscle into the eyelid has also been described (59). It involves raising an innervated flap from the frontalis muscle and passing through the septum towards the tarsus. It is indicated in severe ptosis with poor LF, as an alternative to a sling procedure. It avoids the need for implanted sling material and seems to achieve satisfactory results in select cases. However, there may be some problems with forehead anesthesia, brow asymmetry, lagophthalmos and overcorrection.
Summary
Management of congenital ptosis can be challenging and rewarding at the same time. While most congenital ptosis occurs in isolation, some have complex associations that stretch the creativity of our surgical options. The effect of ptosis can range from a severe amblyogenic potential to negative psychosocial effect. Furthermore, the fact that our examination is in many ways limited by age and cooperation, and there is a lack of intraoperative feedback under general anesthetic, it makes a good outcome that much more satisfactory. While techniques and materials have evolved over time, the main progress is now being made in the advances of embryology and genetics of underlying causes. What we used to assume was an isolated muscle anomaly or maldevelopment, is in many cases now being studied as a central disinnervation effect and it makes our standard classification little more blurred. A lot of the surgical techniques we use have been developed over centuries and the future challenge is to improve upon them. New materials for frontalis suspension are continually being developed and tested. But the real progress likely will be found in better understanding of embryology, neurophysiology and molecular genetics. While successful repair of ptosis usually results in a satisfactory outcome in majority of cases, still more work, improvement and progress lie in the years ahead.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Eye Science for the series “Eyelid Surgery”. The article has undergone external peer review.
Conflicts of Interest: The author has completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes-20-102). The series “Eyelid Surgery” was commissioned by the editorial office without any funding or sponsorship. VK served as the unpaid Guest Editor of the series. The author has no other conflicts of interest to declare.
Ethical Statement: The author is accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Gonzalez MO, Durairaj VD. The history of ptosis surgery. In: Cohen A, Weinberg D. editors. Evaluation and management of blepharoptosis. New York: Springer, 2011.
- Ng SK, Chan W, Marcet MM, et al. Levator palpebrae superioris: an anatomic update. Orbit 2013;32:76-84. [Crossref] [PubMed]
- Lin LK, Uzcategui N, Chang EL. Effect of surgical correction of congenital ptosis on amblyopia. Ophthalmic Plast Reconstr Surg 2008;24:434-6. [Crossref] [PubMed]
- Takahashi Y, Leibovitch I, Kakizaki H. Frontalis suspension surgery in upper eyelid blepharoptosis. Open Ophthalmol J 2010;4:91-7. [Crossref] [PubMed]
- Griepentrog GJ, Diehl NN, Mohney BG. Incidence and demographics of childhood ptosis. Ophthalmology 2011;118:1180-3. [Crossref] [PubMed]
- SooHoo JR. Congenital ptosis. Surv Ophthalmol 2014;59:483-92. [Crossref] [PubMed]
- Yoon JS, Lee SY. Long-term functional and cosmetic outcomes after frontalis suspension using autologous fascia lata for pediatric congenital ptosis. Ophthalmology 2009;116:1405-14. [Crossref] [PubMed]
- Wabbels B, Schroeder JA, Voll B, et al. Electron microscopic findings in levator muscle biopsies of patients with isolated congenital or acquired ptosis. Graefes Arch Clin Exp Ophthalmol 2007;245:1533-41. [Crossref] [PubMed]
- Berke RN, Wadsworth JA. Histology of levator muscle in congenital and acquired ptosis. AMA Arch Ophthalmol 1955;53:413-28. [Crossref] [PubMed]
- McMullan TFW, Robinson DO, Tyers AG. Towards an understanding of congenital ptosis. Orbit 2006;25:179-84. [Crossref] [PubMed]
- Lemagne JM, Colonval S, Moens B, et al. Anatomical modification of the levator muscle of the eyelid in congenital ptosis. Bull Soc Belge Ophtalmol 1992;243:23-7. [PubMed]
- Demirci H, Frueh BR, Nelson CC. Marcus Gunn jaw-winking synkinesis: clinical features and management. Ophthalmology 2010;117:1447-52. [Crossref] [PubMed]
- Beard C. A new treatment for severe unilateral congenital ptosis and for ptosis with jaw-winking. Am J Ophthalmol 1965;59:252-8. [Crossref] [PubMed]
- Allen CE, Rubin PA. Blepharophimosis-ptosis-epicanthus inversus syndrome (BPES): clinical manifestation and treatment. Int Ophthalmol Clin 2008;48:15-23. [Crossref] [PubMed]
- Crisponi L, Deiana M, Loi A, et al. The putative forkhead transcription factor FOXL2 is mutated in blepharophimosis/ptosis/epicanthus inversus syndrome. Nat Genet 2001;27:159-66. [Crossref] [PubMed]
- Yazici B, Unal M, Koksal M, et al. Oculomotor palsy with cyclic spasms. Orbit 2000;19:129-33. [Crossref] [PubMed]
- Mirzai H, Baser E. Congenital Horner’s syndrome and the usefulness of the apraclonidine test in its diagnosis. Indian J Ophthalmol 2006;54:197-9. [Crossref] [PubMed]
- Zafeiriou DI, Economou M, Koliouskas D, et al. Congenital Horner's syndrome associated with cervical neuroblastoma. Eur J Paediatr Neurol 2006;10:90-2. [Crossref] [PubMed]
- Heidary G, Engle EC, Hunter DG. Congenital fibrosis of the extraocular muscles. Semin Ophthalmol 2008;23:3-8. [Crossref] [PubMed]
- DeRespinis PA, Caputo AR, Wagner RS, et al. Duane’s retraction syndrome. Surv Ophthalmol 1993;38:257-88. [Crossref] [PubMed]
- Miller NR, Kiel SM, Green WR, et al. Unilateral Duane’s retraction syndrome (type 1). Arch Ophthalmol 1982;100:1468-72. [Crossref] [PubMed]
- Lee AG, Brazis PW. Chronic progressive external ophthalmoplegia. Curr Neurol Neurosci Rep 2002;2:413-7. [Crossref] [PubMed]
- Caballero PE, Candela MS, Alvarez CI, et al. Chronic progressive external ophthalmoplegia: a report of 6 cases and a review of the literature. Neurologist 2007;13:33-6. [Crossref] [PubMed]
- Allen RC, Zimmerman MB, Watterberg EA, et al. Primary bilateral silicone frontalis suspension for good levator gunction ptosis in oculopharyngeal muscular dystrophy. Br J Ophthalmol 2012;96:841-5. [Crossref] [PubMed]
- Kim JH, Hwang JM. Congenital monocular elevation deficiency. Ophthalmology 2009;116:580-4. [Crossref] [PubMed]
- O’Hara M, Goff MJ, Woods DJ, et al. Malignant hyperthermia in strabismus surgery: a survey of AAPOS members. J AAPOS 2013;17:E24 [Crossref]
- Stein A, Kelly JP, Weiss AH. Congenital eyelid ptosis: onset and prevalence of amblyopia, associations with systemic disorders, and treatment outcomes. J Pediatr 2014;165:820-4.e2. [Crossref] [PubMed]
- Mathews D. Congenital ptosis. In: Rycroft PV. editor. Corneo-plastic surgery. Oxford: Pergamon, 1969:15-29.
- Quaranta-Leoni FM, Sposato S, Leonardi A, et al. Timing of surgical correction for the treatment of unilateral congenital ptosis. Orbit 2017;36:382-7. [Crossref] [PubMed]
- Griepentrog GJ, Diehl N, Mohney BG. Amblyopia in childhood eyelid ptosis. Am J Ophthalmol 2013;155:1125-8.e1. [Crossref] [PubMed]
- Srinagesh V, Simon JW, Meyer DR, et al. The association of refractive error, strabismus, and amblyopia with congenital ptosis. J AAPOS 2011;15:541-4. [Crossref] [PubMed]
- Berry-Brincat A, Willshaw H. Paediatric blepharoptosis: a 10-year review. Eye 2009;23:1554-9. [Crossref] [PubMed]
- Beard C. The surgical treatment of blepharoptosis: a quantitative approach. Trans Am Ophthalmol Soc 1966;64:401-87. [PubMed]
- Beard C. A new classification of blepharoptosis. Int Ophthalmol Clin 1989;29:214-6. [Crossref] [PubMed]
- Feldman I, Brusasco L, Malhotra R. Improving outcomes of posterior approach levatorpexy for congenital ptosis with reduced levator function. Ophthalmic Plast Reconstr Surg 2018;34:460-2. [PubMed]
- Crawford JS. Ptosis: is it correctable, and how? Ann Ophthalmol 1971;3:452-6. [PubMed]
- Philandrianos C, Galinier P, Salazard B, et al. Long-term outcome of frontalis suspension using autogenous temporal fascia or fascia lata in children. J Plast Reconstr Aesthet Surg 2010;63:782-6. [Crossref] [PubMed]
- Levine M, Allen C. Manual of oculoplastic surgery. 5th ed. Cham: Springer Publishing, 2018.
- Gladstone G, Nesi G, Black E. Oculoplastic surgery atlas. Cham: Springer Publishing, 2018.
- Leatherbarrow B. Oculoplastic surgery. 3rd ed. New York: Thieme Medical Publishers, 2020.
- Beard C. Ptosis. St. Louis: Mosby Company, 1976.
- Berke RN. Results of resection of the levator muscle through a skin incision in congenital ptosis. AMA Arch Ophthalmol 1959;61:177-201. [Crossref] [PubMed]
- Putterman AM, Urist MJ. Müller muscle-conjunctiva resection. Technique for treatment of blepharoptosis. Arch Ophthalmol 1975;93:619-23. [Crossref] [PubMed]
- Anderson RL. Predictable ptosis procedures: do not go to the dark side. Ophthal Plast Reconstr Surg 2012;28:239-41. [Crossref] [PubMed]
- Fasanella RM, Servat J. Levator resection for minimal ptosis: another simplified operation. Arch Ophthalmol 1961;65:493-6. [Crossref] [PubMed]
- Samimi DB, Erb MH, Lane CJ, et al. The Modified Fasanella-Servat Procedure: description and Quantified Analysis. Ophthal Plast Reconstr Surg 2013;29:30-4. [Crossref] [PubMed]
- Ahmadi AJ, Sires B. Ptosis in infants and children. Int Ophthalmol Clin 2002;42:15-29. [Crossref] [PubMed]
- Lee MJ, Oh JY, Choung HK, et al. Frontalis sling operation using silicone rod compared with preserved fascia lata for congenital ptosis a three-year follow-up study. Ophthalmology 2009;116:123-9. [Crossref] [PubMed]
- Patel RM, Aakalu VK, Setabutr P, et al. Efficacy of Müller’s muscle and conjunctival resection with or without tarsectomy for the treatment of severe involutional blepharoptosis. Ophthalmic Plast Reconstr Surg 2017;33:273-8. [Crossref] [PubMed]
- Woo KI, Kim YD, Kim YH. Surgical treatment of severe congenital ptosis in patients younger than two years of age using preserved fascia lata. Am J Ophthalmol 2014;157:1221-6.e1. [Crossref] [PubMed]
- Crawford JS. Repair of ptosis using frontalis muscle and fascia. Trans Am Acad Ophthalmol Otolaryngol 1956;60:672-8. [PubMed]
- Bansal RK, Sharma S. Results and complications of silicone frontalis sling surgery for ptosis. J Pediatr Ophthalmol Strabismus 2015;52:93-7. [Crossref] [PubMed]
- Ben Simon GJ, Macedo AA, Schwarcz RM, et al. Frontalis suspension for upper eyelid ptosis: evaluation of different surgical designs and suture material. Am J Ophthalmol 2005;140:877-85. [Crossref] [PubMed]
- Mokhtarzadeh A, Harrison AR. Controversies and advances in the management of congenital ptosis. Expert Review of Ophthalmology 2015;10:59-63. [Crossref] [PubMed]
- Kersten RC, Bernardini FP, Khouri L, et al. Unilateral frontalis sling for the surgical correction of unilateral poor-function ptosis. Ophthalmic Plast Reconstr Surg 2005;21:412-6; discussion 416-7.
- Callahan A. Correction of unilateral blepharoptosis with bilateral eyelid suspension. Am J Ophthalmol 1972;74:321-6. [Crossref] [PubMed]
- Anderson RL, Jordan DR, Dutton JJ. Whitnall’s sling for poor function ptosis. Arch Ophthalmol 1990;108:1628-32. [Crossref] [PubMed]
- Cruz AA, Akaishi PM, Mendonça AK, et al. Supramaximal levator resection for unilateral congenital ptosis: cosmetic and functional results. Ophthalmic Plast Reconstr Surg 2014;30:366-71. [Crossref] [PubMed]
- Ramirez OM, Pena G. Frontalis muscle advancement: a dynamic strucrure for the treatment of severe congenital ptosis. Plast Reconstr Surg 2004;113:1841-9. [Crossref] [PubMed]
Cite this article as: Kratky V. Treatment of congenital ptosis. Ann Eye Sci 2020;5:37.










