
Ophthalmic manifestations of cat scratch disease
Introduction
Bartonella henselae is a Gram-negative intracellular bacillus, responsible for cat scratch disease (CSD). It is largely distributed among cats, with prevalence varying from 18.1% to 41.7% (1). Cats infected with Bartonella usually have asymptomatic bacteremia that can persist for several months. A transmission to humans may occur directly from a cat scratch or likely through the cat flea Ctenocephalides felix (2). The correlation between B. Henselae and CSD was established in 1989 but the first description of the disease was reported 40 years earlier, based on histopathological examination of infected lymph node biopsy (3).
The seroprevalence of Bartonella varies from 2.0% to 32.38% in Eastern China and from 12.8% to 13.7% in Brazil (4,5). The annual incidence of CSD has been recently assessed as 4.7 per 100,000 in United States (6). Although the disease can affect any age group, CSD occurs most frequently in children and adolescents.
The diagnosis of CSD is based on history of contact with a cat and high serum titers of immunoglobulin G antibodies against B. henselae. In fact, the isolation of Bartonella in culture is difficult, rarely successful and requires some weeks. The most advanced technique used to detect B. henselae in patients’ tissues is the polymerase chain reaction (PCR).
The therapeutical approach to CSD is based on patient’s immune status and severity of the disease; the prognosis in immunocompetent individuals is usually good. Many cases of CSD are asymptomatic but the most frequent clinical manifestations include fever and regional lymphadenopathy. However, extra-lymphatic manifestations, such as central nervous system (CNS) involvement, hepatosplenic disease, endocarditis, musculoskeletal involvement and skin inflammation, can also occur.
Ophthalmological manifestations of CSD include Parinaud’s oculoglandular syndrome, neuroretinitis and, less frequently, retinochoroiditis, retinal vein occlusion, retinal branch artery occlusion, macular hole, serous retinal detachment and others (7).
Knowledge of the ophthalmic manifestations of CSD and its complications can lead to a prompt diagnosis of ocular bartonellosis, which represents a continuous burden worldwide.
This article will review the current systemic and ophthalmic manifestations of CSD and its diagnosis, therapy and prognosis in order to provide the ophthalmologists with relevant data for their clinical practice.
Methods
The literature review for this article was based on a search in PubMed/Medline database to select reviews, original articles and case report published in English language from January 1950 to June 2020 about CSD and the eye. The following keywords were used: “Cat scratch disease” OR “Bartonella” OR “Bartonellosis” AND “Eye” OR “Systemic manifestations” OR “Parinaud’s oculoglandular syndrome” OR “Neuroretinitis”. The collected articles were examined by authors to review systemic manifestations, ocular manifestations, diagnosis, differential diagnosis, treatment and prognosis of CSD.
Discussion
Systemic manifestations of CSD
CSD is one of the leading causes of benign chronic lymphadenopathy in children and young adults, whereas its occurrence in the elderly is rare (8).
Most patients with CSD present with one or more 3- to 5-mm red-brown nontender papules at the site of inoculation 3 to 10 days after the introduction of bacteria. These primary lesions recede after 1–3 weeks, then regional lymphadenopathy appears. Axillary, cervical, submandibular and preauricular lymph-nodes are most frequently involved (7). Lymphadenopathy can be tender, with erythema and warm skin and generally remains regional. It can resolve within few months but may persist up to one year or rarely for longer.
Approximately up to 50% of patients also experience systemic symptoms. When this occurs, patients may experience prolonged fever, weight loss, malaise, myalgia, arthralgia or arthropathy and splenomegaly. More severe manifestations, including encephalopathy, hepatic and splenic abscesses, endocarditis, pneumonia and osteomyelitis have also been described (9).
Infection of immunocompromised patients leads to the so-called “bacillary angiomatosis-peliosis”, a condition characterized by angioproliferative lesions resembling those of Kaposi sarcoma in the skin, bone, liver, spleen and other organs.
CNS involvement
CNS manifestations are present in 5% of patients with CSD. In addition to headaches, these may include seizures, mental status changes, hemiplegia, myelitis and transient peripheral neuropathy. Encephalopathy is the most common neurological complication of CSD, which include transverse myelitis, radiculitis and cerebellar ataxia (10). Most patients are young. Encephalitis is characterized by restlessness, confusion and disorientation. Generalized headache and nuchal rigidity are often present. Symptoms may quickly progress to coma. Cranial or peripheral nerve involvement are other possible findings. Recovery from CNS involvement of CSD may be slow and can require one year or longer.
Hepatosplenic CSD
Hepatosplenic CSD is a rare form of atypical CSD found in immunocompetent individual. Patients develop daily fevers up to 104 °F (40 °C) and no other identifiable cause. Physical examination is generally normal, with occasional observation of one or more cutaneous scars secondary to cat scratches. Lymphadenopathy is present in about half of cases and abdominal discomfort is commonly reported. The diagnosis is based on typical filling defects in the liver, spleen, or both as showed by ultrasonography and/or computed tomography (CT) scanning and a positive serology for B. henselae. The liver has a characteristic nutmeg aspect with stellate necrotizing granulomata on the histologic examination. Most patients become afebrile within 48 hours after initiating antibiotic therapy, although in some cases fever can last for up to a month.
Endocarditis
About 3% of all cases of endocarditis are attributable to Bartonella species (3). Bartonella infection should be considered in patients with endocarditis and negative blood culture results who have regular contact with cats. Patients with Bartonella endocarditis may need valve replacement.
Musculoskeletal involvement
Osteomyelitis secondary to B. henselae is rare but is being reported with increased frequency. CT scanning, magnetic resonance imaging (MRI) and scintigraphy can all be used to show bone involvement. Immunosuppression does not represent a risk factor for the development of osteomyelitis. Generally, affected patients respond well to treatment with no significant long-term effect.
Dermatologic manifestations
As seen above, the disease usually begins with erythematous papule, single or multiple, at the site of inoculation. Typically, the papule are followed 1 to 3 weeks later by regional lymphadenopathy. Other dermatologic manifestations, such as erythema nodosum, macular, maculopapular, morbilliform, and petechial rashes occur in approximatively 5% of patients and are more likely individuals with more severe or atypical disease.
Ocular manifestations of CSD
Ocular involvement occurs in 5% to 10% of patients with CSD (11). The eye may represent the first site of inoculation, resulting in the so-called “Parinaud’s oculoglandular syndrome” or by haematogenous spread, leading to a large variety of other ocular manifestations. Most common symptoms include redness, ocular discomfort, unilateral of bilateral visual loss and scotoma.
Parinaud’s oculoglandular syndrome (POGS)
First described by Henri Parinaud in 1889, POGS is characterized by unilateral granulomatous conjunctivitis associated with locoregional lymphadenopathy, predominantly involving the preauricular or submandibular lymph-nodes, and fever. Its association with CSD was first pointed out in 1953 (12). In 1990 Jawad and Amen reported two patients with POGS, father and daughter, both scratched by the same cat (13).
In 1999, Bartonella henselae was first recovered from conjunctival scraping of a patient affected by POGS, providing strong evidence for its role as causative agent of CSD (14).
Considering that presentation may be atypical, diagnosis of POGS is often challenging.
Neuroretinitis
Originally described by Leber in 1916 as a “stellate maculopathy”, neuroretinitis is an inflammation of neural retina and optic nerve. The term neuroretinitis was proposed by Gass in 1977, citing that disc edema precedes macular exudates.
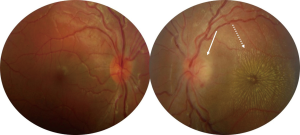
In 1970, was first found a correlation between the appearance of neuroretinitis and CSD (15). Neuroretinitis is the most common posterior segment complication of the disease. CSD is one of the most common infectious cause of optic disc edema with a macular star of exudates. Other infectious causes include syphilis, Lyme disease, Rocky Mountain Spotted Fever, toxoplasmosis, toxocariasis, histoplasmosis, leptospirosis and many others. Neuroretinitis may also be idiopathic and recurrent (16). Papilledema and malignant hypertension may produce a similar ophthalmoscopic feature and should be excluded when neuroretinitis is bilateral. However, more often, in malignant hypertension the macular star is limited to the temporal side of the optic nerve (Figure 1). Neuroretinitis is typically characterized by reduced visual acuity, optic disc inflammation associated to a partial or complete ‘star-shaped’ exudates around the macula (Figure 2) in one eye. Bilateral involvement can also be observed, especially in immunocompromised patients. A mild vitritis may also be present. The macular star may be absent at initial presentation, becoming visible up to two weeks after the onset of disease, by which time optic disc inflammation may have reduced. The macular star vanishes after about a month but may persist for a year. Optic disc inflammation, on the contrary, may take 2–3 months to completely disappear. Thus, neuroretinitis enters the differential diagnosis of isolated optic disc edema when macular star has not yet formed. The macular star is characterized by the leakage of lipid-rich fluid from the optic disc vasculature into the outer plexiform layer of the macula. The passage of the aqueous phase through the external membrane to beneath the neurosensory retina results in deposition of lipids in a star arrangement around the macula (17). Optical coherence tomography (OCT) is helpful in the diagnosis and monitoring of patients with CSD neuroretinitis detecting subretinal fluid, even when this cannot be easily seen on ophthalmoscopy (18) (Figure 3). OCT is also able to show retinal thickening, fluid or exudates and even a serous retinal detachment before macular star is appeared. Macular exudates are visible as hyperreflective lesions within the outer plexiform layer (Henle’s layer). Fundus autofluorescence (FAF) may also assist in detecting retinal exudates, which appear as hypoautofluorescent lesions. Zatreanu et al. (19) described 4 patients with neuroretinitis who presented several distinguishing and novel features on spectral-domain i.e.,: “epipapillary infiltrates” and an atypical pattern of inner retinal folds. They concluded that the recognition of these findings on OCT may lead to an early diagnosis of neuroretinitis. Visual prognosis of neuroretinitis is generally good with over 80% of cases achieving 20/40 or better vision (20).
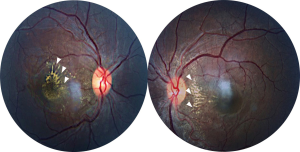
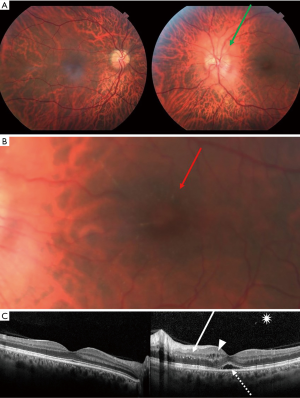
Retinochoroiditis
Chorioretinal involvement is another common manifestation of CSD. Small or large retinal lesions consistent with inner retinitis or retinochoroiditis with a typical juxtavascular location may be observed (11). These infiltrates may be associated or not with neuroretinitis. Telangiectasia or angiomatous-like proliferation of retinal capillaries, more clearly detectable with fluorescein angiography, may also accompany retinochoroiditis.
Chorioretinal manifestations may often lead to a worse visual prognosis than neuroretinitis. In a recent case series in which retinal infiltrates and angiomatous lesions were the most common presentation of CSD, 56.8% of affected eyes presented with vision worse than 20/200 and 43.2% with visual acuity better than 20/200 (21). After treatment, visual acuity improved in most of eyes with only 13.4% having vision worse than 20/200.
Other ophthalmic manifestations of Bartonella infection
While CSD has a predilection to cause neuroretinitis and other form of uveitis, it may also be responsible of a wide number of other ophthalmic complications, albeit more rarely. Retinal vascular occlusions are known as effect of Bartonella infection (22). CSD related retinal vascular occlusion could be the result of mechanical occlusion by an inflammatory lesion or bacteria invasion to the vascular endothelium causing obliterative or thrombogenic vasculopathy (23). Habot-Wilner et al. (18) reviewed ocular manifestation of 86 patients among a cohort of 141 cases of CSD with ophthalmic complications. Branch retinal vein occlusion was found in one patient only (1.2%). However, signs of retinal vascular occlusion were present in 7% of eyes. All eyes with vessel occlusions, except for one eye with vein occlusion, displayed swollen disc as well. Therefore, isolated vein occlusion is a rare but possible manifestation of CSD and should be suspected when other risk factors are absent. Branch retinal artery occlusion was present in 4% of eyes and mixed (arterial and vein) occlusion in 1% of patients. In another report by Solley et al. (24), vasculopathic effect of Bartonella infection of the posterior segment was evident in 8 out of 24 cases (33%). Peripapillary angiomatosis is another known but rare manifestation of retinal and optic nerve involvement. It may be described as an elevated and edematous optic disc, with dilated capillaries and an adjacent yellow-orange subretinal mass.
Other ocular complications of CSD include vitreitis, vitreous hemorrhage, intermediate uveitis, retinal vasculitis, inflammatory retinal mass, subretinal mass associated with abnormal vascular network among patients with human immunodeficiency virus infection, choroidal granuloma, white dot syndrome, serous macular detachment, retina-retinal anastomosis, macular edema, macular hole, optic nerve head mass, panuveitis mimicking Vogt-Koyanagi-Harada disease, acute endophthalmitis and choroidal neovascularization (11,25-27).
Diagnosis
Historically, the diagnosis of CSD was made when three of the following criteria were present: history of contact with a cat, positive skin test in response to CSD antigen, characteristic histopathological changes in lymph-node biopsy and no other identified causes of lymphadenopathy.
Because of the culture isolation of B. henselae from blood patient is difficult and requires a time of incubation up to six weeks, it is not widely used for diagnosis of CSD (17).
Alternatively, serologic testing using either indirect fluorescence assay (IFA) or enzyme-linked immunosorbent assay (ELISA) have been developed for detecting anti-Bartonella immunoglobulin M (IgM) and immunoglobulin G (IgG). The IFA test seems to be the most reliable method with higher specificity than ELISA and represents the more largely used test for B. henselae (11).
IgM positivity indicates acute infection whereas if IgG are found the titers can help to diagnosis. Titers 1:64 or less suggest inactive infection, titers between 1:64 and 1:256 indicate possible CSD and testing should be repeated after 10–14 days; lastly, titers exceeding 1:256 establish diagnosis of CSD.
PCR for the detection of B. henselae 16S ribosomal RNA gene on tissue samples such as lymph nodes is another useful tool for CSD diagnosis when serology is negative but clinical findings are suggestive for the disease. The other two main approaches to use PCR are amplification of the gltA gene and amplification of htrA gene of B. henselae DNA (3). Unfortunately, PCR has lower sensitivity than serology testing (9). Moreover, there are few reports about PCR in intraocular fluids (28,29).
Multimodal imaging can be useful in demonstrating ocular findings associated with CSD. FAF may show hypoautofluorescent lesions corresponding to macular star exudates. Teleangiectasia are better shown by fluorescein angiography (FA) than fundoscopic examination. FA may also demonstrate late optic disc hyperfluorescence, foci of retinochoroiditis (Figure 4) and artery or vein occlusion. Indocyanine green angiography may also display optic disc hypercyanescence. As discussed above, OCT is helpful in diagnosis and follow-up of patients with CSD neuroretinitis.
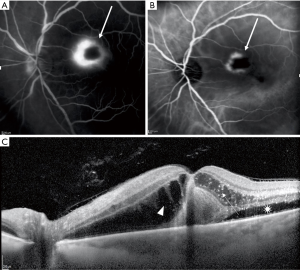
OCT angiography (OCTA), a recent non-invasive imaging technique, has been used to identify neovascularization within foci of chorioretinitis (30). OCTA may also detect optic disc teleangiectatic vessels (11).
Differential diagnosis
The differential diagnosis is based on history, systemic signs and symptoms and the pattern of ocular manifestations.
CSD is the most common cause of neuroretinitis. However, other diseases should be ruled out, including tuberculosis, toxoplasmosis, toxocariasis, Lyme disease, Rocky Mountain Spotted Fever, leptospirosis, rickettsiosis, syphilis, dengue fever, chikungunya and other conditions.
Noninfectious diseases presenting with optic disc swelling and macular star, such as severe systemic hypertension, diabetes, increased intracranial pressure and other ocular conditions, including sarcoidosis, Beçhet disease, anterior ischemic optic neuropathy and retinal vein occlusion should be regarded in the differential diagnosis of CSD (11). Idiopathic and recurrent neuroretinitis has been reported (16).
Treatment
CSD is usually self-limited and most cases do not require therapy. There is no consensus on treatment and management of CSD and its ocular complications. A wide range of antibiotics have been prescribed, including doxycycline, azithromycin and other macrolides (clarithromycin and erythromycin), rifampin, ciprofloxacin, ceftriaxone and trimethoprim-sulfamethoxazole (27).
The most frequently antibiotic used in adults is doxycycline per os at the dosage of 100 mg twice a day for 2–4 weeks in immunocompetent individuals, prolonged up to 4 months in immunocompromised patients. Azithromycin could represent a viable option for treatment of CSD (31). Additionally, in children may be better to use macrolide antibiotics because of longer term side effects of doxycycline in younger patients.
Intravenous therapy or combination of antibiotics, for example doxycycline and rifampicin, can be used in severe cases (11).
The use of corticosteroids remains controversial but should be considered in association with antibiotic therapy in case of severe inflammation. In a recent comparative study, the patients treated with oral corticosteroids in addition to antibiotic had better visual outcome than patients having antibiotic alone (32).
Anti-vascular endothelial growth factor (VEGF) agents have been used for macular complications of CSD, including macular edema and choroidal neovascularization (27).
Prognosis
CSD-associated ocular complications are usually self-limited in immunocompetent patients with full visual acuity recovery within weeks or months.
The macular star disappears most frequently within few weeks but may persist for a year. Optic disc inflammation usually disappears in few months and only in rare cases a mild optic disc pallor may persist.
Visual outcomes are worse when retinochoroiditis, retinal vascular occlusions or macular complications occur.
Conclusions
In this article, we reviewed the current systemic and ophthalmic manifestations of CSD and its diagnosis, therapy and prognosis. CSD is a worldwide infectious disease due to an intracellular bacillus, B. henselae and spp., transmitted to humans by infected cats. CSD produces local lymphatic disease, most frequently with suppurative adenopathies, but ocular involvement is also possible.
CSD is found to be the main cause of neuroretinitis. Typical findings are optic disc swelling and a lipid macular exudation arranged in a partial or complete macular star, often associated with mild vitreitis. Since the macular exudates typically follow optic disc inflammation, it is mandatory not miss the diagnosis before macular star appears. Differential diagnosis includes infectious diseases, such as tuberculosis, toxoplasmosis, syphilis, and noninfectious diseases, including sarcoidosis, malignant systemic hypertension and diabetes. Other ocular manifestations of CSD are POGS, and less frequently, retinochoroiditis, retinal vein occlusion and retinal branch artery occlusion.
Diagnosis of CSD is currently made by history of contact with a cat, clinical features and positive serology. IgM positivity or IgG titers higher than 1:256 allow to define the diagnosis of CSD. PCR-based detection of B. henselae is a novel technique that could be useful to diagnose CSD.
Treatment guidelines are not well defined. The mainstay treatment of CSD consists of doxycycline 100 mg per os twice a day for 2–4 weeks in immunocompetent patients, up to 4 months in immunocompromised patients. Oral corticosteroids should be considered in association with antibiotic therapy when severe inflammation is present.
Most of patients have a self-limited course and a complete visual recovery; however, potential sight-threatening complications can also occur in CSD.
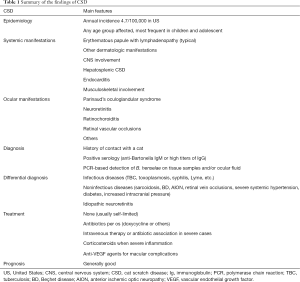
Table 1 summarizes the core points and findings of CSD.
Full table
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes-20-109). LC serves as an unpaid editorial board member of Annals of Eye Science from May 2020 to Apr. 2022. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Müller A, Walker R, Bittencourt P, et al. Prevalence, hematological findings and genetic diversity of Bartonella spp. in domestic cats from Valdivia, Southern Chile. Parasitology 2017;144:773-82. [Crossref] [PubMed]
- Iannino F, Salucci S, Di Provvido A, et al. Bartonella infections in humans, dogs and cats. Vet Ital 2018;54:63-72. [PubMed]
- Mazur-Melewska K, Mania A, Kemnitz P, et al. Cat-scratch disease: a wide spectrum of clinical pictures. Postepy Dermatol Alergol 2015;32:216-20. [Crossref] [PubMed]
- Nelson CA, Saha S, Mead PS. Cat-Scratch Disease in the United States, 2005-2013. Emerg Infect Dis 2016;22:1741-6. [Crossref] [PubMed]
- Sun J, Fu G, Lin J, et al. Seroprevalence of Bartonella in Eastern China and analysis of risk factors. BMC Infect Dis 2010;10:121. [Crossref] [PubMed]
- da Costa PS, Brigatte ME, Greco DB. Antibodies to Rickettsia rickettsii, Rickettsia typhi, Coxiella burnetii, Bartonella henselae, Bartonella quintana, and Ehrlichia chaffeensis among healthy population in Minas Gerais, Brazil. Mem Inst Oswaldo Cruz 2005;100:853-9. [Crossref] [PubMed]
- Angelakis E, Raoult D. Pathogenicity and treatment of Bartonella infections. Int J Antimicrob Agents 2014;44:16-25. [Crossref] [PubMed]
- Ben-Ami R, Ephros M, Avidor B, et al. Cat-scratch disease in elderly patients. Clin Infect Dis 2005;41:969-74. [Crossref] [PubMed]
- Biancardi AL, Curi ALL. Cat scratch disease. Ocul Immunol Inflamm 2014;22:148-54. [Crossref] [PubMed]
- Samarkos M, Antoniadou V, Vaiopoulos AG, et al. Encephalopathy in an adult with cat-scratch disease. BMJ Case Rep 2018;2018:bcr2017223647 [Crossref] [PubMed]
- Ksiaa I, Abroug N, Mahmoud A, et al. Update on Bartonella neuroretinitis. J Curr Ophthalmol 2019;31:254-61. [Crossref] [PubMed]
- Cassady JV, Culbertson CS. Cat-scratch disease and Parinaud’s oculoglandular syndrome. AMA Arch Ophthalmol 1953;50:68-74. [Crossref] [PubMed]
- Jawad AS, Amen AA. Cat-scratch disease presenting as the oculoglandular syndrome of Parinaud: a report of two cases. Postgrad Med J 1990;66:467-8. [Crossref] [PubMed]
- Grando D, Sullivan LJ, Flexman JP, et al. Bartonella henselae associated with Parinaud’s oculoglandular syndrome. Clin Infect Dis 1999;28:1156-8. [Crossref] [PubMed]
- Sweeney VP, Drance SM. Optic neuritis and compressive neuropathy associated with cat scratch disease. Can Med Assoc J 1970;103:1380-1. [PubMed]
- Purvin V, Sundaram S, Kawasaki A. Neuroretinitis: Review of the Literature and New Observations. J Neuroophthalmol 2011;31:58-68. [Crossref] [PubMed]
- Johnson A. Ocular complications of cat scratch disease. Br J Ophthalmol 2020;104:1640-6. [Crossref] [PubMed]
- Habot-Wilner Z, Zur D, Goldstein M, et al. Macular findings on optical coherence tomography in cat- scratch disease neuroretinitis. Eye 2011;25:1064-8. [Crossref] [PubMed]
- Zatreanu L, Sibony PA, Kupersmith MJ. Optical Coherence Tomography in Neuroretinitis: Epipapillary Infiltrates and Retinal Folds. J Neuroophthalmol 2017;37:176-8. [Crossref] [PubMed]
- Chi SL, Stinnett S, Eggenberger E, et al. Clinical characteristics in 53 patients with cat scratch optic neuropathy. Ophthalmology 2012;119:183-7. [Crossref] [PubMed]
- Curi AL, Machado D, Heringer G, et al. Cat-scratch disease: ocular manifestations and visual outcome. Int Ophthalmol 2010;30:553-8. [Crossref] [PubMed]
- Williams A, Sheldon CD, Riordan T. Cat scratch disease. BMJ 2002;324:1199-200. [Crossref] [PubMed]
- Cunningham ET, Koehler JE. Ocular bartonellosis. Am J Ophthalmol 2000;130:340-9. [Crossref] [PubMed]
- Solley WA, Martin DF, Newman NJ, et al. Cat scratch disease: posterior segment manifestations. Ophthalmology 1999;106:1546-53. [Crossref] [PubMed]
- Ghazi NG, Sams WA. A case of cat-scratch disease with unusual ophthalmic manifestations. Middle East Afr J Ophthalmol 2012;19:243-6. [Crossref] [PubMed]
- Albini TA, Lakhanpal RR, Foroozan R, et al. Macular hole in cat scratch disease. Am J Ophthalmol 2005;140:149-51. [Crossref] [PubMed]
- Mabra D, Yeh S, Shantha JG. Ocular manifestations of bartonellosis. Curr Opin Ophthalmol 2018;29:582-7. [Crossref] [PubMed]
- Drancourt M, Berger P, Terrada C, et al. High prevalence of fastidious bacteria in 1520 cases of uveitis of unknown etiology. Medicine (Baltimore) 2008;87:167-76. [Crossref] [PubMed]
- Brydak-Godowska J, Kopacz D, Borkowski PK, et al. Seroprevalence of Bartonella Species in Patients with Ocular Inflammation. Adv Exp Med Biol 2017;1020:33-42. [Crossref] [PubMed]
- Pichi F, Srivastava SK, Levinson A, et al. A Focal Chorioretinal Bartonella Lesion Analyzed by Optical Coherence Tomography Angiography. Ophthalmic Surg Lasers Imaging Retina 2016;47:585-8. [Crossref] [PubMed]
- Tan CL, Fhun LC, Tai EL, et al. Clinical Profile and Visual Outcome of Ocular Bartonellosis in Malaysia. J Trop Med 2017;2017:7946123 [Crossref] [PubMed]
- Habot-Wilner Z, Trivizki O, Goldstein M, et al. Cat-scratch disease: ocular manifestations and treatment outcome. Acta Ophthalmol 2018;96:e524-e532. [Crossref] [PubMed]
Cite this article as: Cirone D, Mandarà E, De Simone L, Pellegrini F, Cimino L. Ophthalmic manifestations of cat scratch disease. Ann Eye Sci 2021;6:17.


