
Association between the M98K variant of the OPTN gene and the risk for primary open angle glaucoma: an updated meta-analysis
Introduction
Primary open angle glaucoma (POAG), characterized by progressive neurodegeneration of retinal ganglion cells (RGCs) and their axons, is the leading cause of irreversible blindness worldwide (1). It is a heritable disease with the evidence from twin studies and familial clustering studies (2,3). There is a significant progress in the genetic basis of POAG. About 5% of POAG is known as a Mendedian disease and caused by a single gene (4). Others are attributed to multiple genetic and environmental risk factors.
Optineurin (OPTN) is one of disease-causing genes of POAG, which is firstly investigated in a large British family with normal tension glaucoma (NTG) (5,6). Four mutations, Glu50→Lys (E50K), Arg545→Gln (R545Q), c.691_692insAG (Premature stop) and Met98→Lys (M98K), were identified in the original kindred (6). The rare, high-penetrance mutation, E50K, is the clearest disease-causing OPTN mutation for POAG (7). Glaucoma patients who had the E50K mutation were reported to have a younger age of onset, more advanced optic disc cupping, smaller neuroretinal rim area and higher rate of filtration surgery required (7). Among the above OPTN mutations, only M98K variant was present in normal controls (9/422), although the frequency was statistically lower than that in POAG patients (6). Multiple subsequent studies of different ethnicities were performed to evaluate the association between the risk allele (M98K) in the causative POAG gene (OPTN) and the risk of POAG, which produce conflicting results. Although three meta-analysis studies (8-10) have tried to clarify this association, the most recent meta-analysis included only 5 studies and new association studies were published after the other two meta-analyses published in 2006 (8) and 2010 (9). Importantly, the results among these meta-analyses differs, which makes an updated in depth analysis on this topic imperative to reach a definitive conclusion. We present the following article in accordance with the PRISMA reporting checklist (available at http://dx.doi.org/10.21037/aes-20-124).
Methods
Search strategy
In this study, we searched the PubMed, Embase, Web of Science, and China National Knowledge Infrastructure (CNKI) databases to find all articles describing the relationships between the M98K variant and POAG, which were published from the inception to 31 December, 2019. The following search terms were used as a text word: “Optineurin or OPTN or M98K or Met98Lys” and “primary open angle glaucoma or open angle glaucoma or POAG or normal pressure glaucoma or low tension glaucoma or normal tension glaucoma or high tension glaucoma”. The detailed search strategy was provided as Appendix 1. The language was restricted to English and Chinese. Two independent reviewers (C.G. and X.Y.) screened the titles and abstracts of all relevant articles, manually examined the reference lists and relevant reviews for additional publications, and evaluated the full texts to identify eligible studies.
Eligibility criteria
Included studies were case-control studies in assessing the associations between OPTN (M98K) mutation and the risk of POAG and its subgroups, the age and intraocular pressure (IOP) at diagnosis in POAG patients. Studies without available data, case-only studies, reviews, conference abstracts or family-based studies were excluded. If more than one population with available data were included in a single study, each population was regarded as separate data in the analyses. We included the most comprehensive study when duplicate sequencing data existed in multiple studies.
Quality assessment
The Newcastle-Ottawa Scale (NOS) was used to assess the quality of each case-control study with its ‘star system’ by two reviewers (C.G. and X.Y.), which is judged on three broad perspectives: the selection of the study groups; the comparability of the groups; and the ascertainment of either the exposure or outcome of interest for case-control studies respectively (11). The disagreement between the two reviewers was solved by a senior reviewer (Z.F.).
Data extraction
Two reviewers (C.G. and X.Y.) extracted data into a customized table independently. Any discrepancy was resolved by consensus. The extracted information included first author, title, published year, country, ethnicity, age at diagnosis, IOP at diagnosis, genotypes distribution of M98K in POAG patients [NTG and high tension glaucoma (HTG) patients; Asian and non-Asian POAG patients; juvenile open angle glaucoma (JOAG) and adult-onset POAG] and controls, the total number of patients and controls.
Statistical analyses
Meta-analysis was conducted using Comprehensive Meta-Analysis software version 2.2.064 (Biostat Inc., NJ, USA). The statistical significance of the pooled odds ratio (OR) and 95% confidence interval (CI) was determined by Z-test. To reduce the type I error, the Bonferroni correction was used to determine the statistically significant genetic models. Because multiple comparisons were performed 35 times, the P value less than 0.05/35 (0.0014) was considered as statistical significance after Bonferroni correction. The heterogeneity across studies was assessed and qualified by the I2 index, with I2 of greater than 50% considered as large or extreme heterogeneity (12). Additionally, a Q-statistic test was performed. All meta-analyses and subgroup analyses were performed by the random-effects model. The contour-enhanced funnel plot with trim-and-fill method and Egger’s tests were used to assess the potential publication bias. The Power and Sample Size Calculation software (13) was used to assess the expected statistical power of our meta-analysis in detecting the association between M98K and risk of POAG at a level of significance of 0.0014.
Results
Inclusion of studies
A total of 1,218 records were identified, yielding 767 studies after removal of duplicates. Following screening, 719 publications were excluded based on the title and abstract, and 48 full text articles were assessed for eligibility. After excluding 9 studies without data on M98K and 5 studies with duplicate data, a total of 34 publications including 39 populations with available data were included in the final analyses. Ayala-Hugo et al. (14) reported 4 populations, including Asian, African, Hispanic, and Caucasian subjects; Alward et al. (15) reported 2 populations, including Caucasian (Iowa and Australia) and Japanese samples; Melki et al. (16) reported 2 populations, including French and Moroccan. The flow chart of literature selection was shown in Figure 1. A total of 7,310 POAG patients and 5,173 controls were involved. The characteristics of subjects were summarized in Table 1. According to the NOS, our included studies achieved an average of 6.21 stars for quality assessment (Table 2, Figure 2).
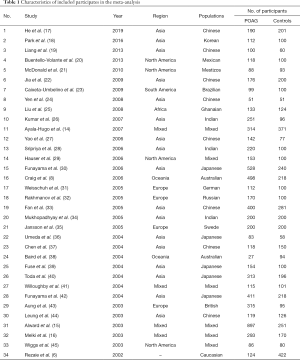
Full table
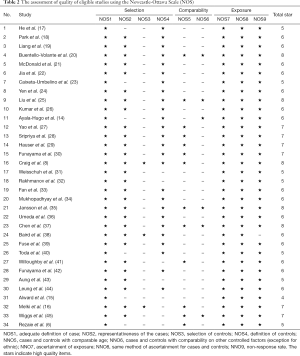
Full table
Meta-analyses and subgroup analyses
In the analyses of association between M98K and risk of each POAG subgroup, including NTG, HTG, Asian POAG, non-Asian POAG, JOAG, and adult-onset POAG, under the multiple genetic models, we performed the Bonferroni correction to reduce the type I error (Table 3).
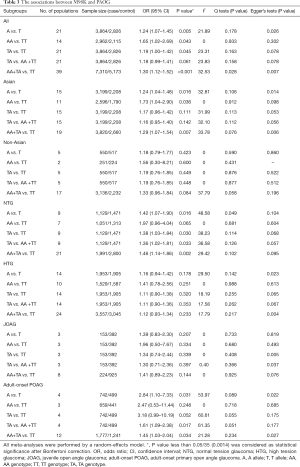
Full table
We found significantly more OPTN M98K carriers in the overall POAG patients than those in the controls under the dominant model (OR =1.30, 95% CI, 1.12–1.52, P<0.001; Power =0.958) (Figure 3).
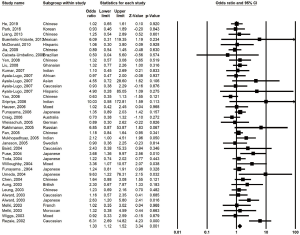
In the stratification analysis, M98K was not associated with NTG and HTG under any model. No association was observed between M98K and POAG risk in the non-Asian and the Asian population. Also, M98K was not associated with JOAG and adult-onset POAG under any model.
Association between M98K and IOP/age at diagnosis in POAG patients
No significant association was observed between M98K and IOP at diagnosis in POAG patients (SMD =−0.19, 95% CI, −0.46 to 0.07; P=0.156). Also, M98K was not associated with age at diagnosis in POAG patients (SMD =0.17, 95% CI, −0.15 to 0.49; P=0.294).
Publication bias and sensitivity analysis
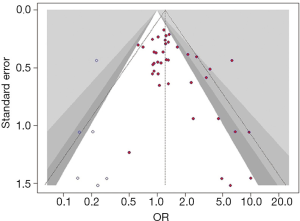
For the significant association in our study (Table 3), the Egger tests suggested statistically significant publication biases existed in the associations between M98K and overall POAG risk under the dominant model (P=0.007).
The counter-enhanced funnel plot with trim-and-fill method was used to assess the influence of publication bias in the pooled effects (Figure 4). The association between M98K and overall POAG risk under the dominant model (OR: 1.22, 95% CI, 1.02–1.46; P=0.029) was still statistically significant after adding 6 studies.
Conclusions
This is an updated meta-analysis to investigate the association between M98K polymorphism and risk for POAG. Our results indicate that M98K increases risks of overall POAG under the dominant model.
The three reported meta-analyses on the M98K in the risk of POAG had controversial results, with positive associations by Chen et al. (10) and Craig et al. (8) while negative association by Cheng et al. (9). The difference in included populations may explain this inconsistency. The latest meta-analysis only included 5 studies (10), and the other two published in 2010 (9) and 2006 (8) included 23 and 19 studies respectively. In this comprehensive study, a total of 34 publications including 39 populations were included, which made us possible to make a more definitive conclusion.
Being in accordance with the dominant inherited mode of OPTN in the POAG pedigree (5,6), M98K was found to be positively associated with overall POAG under the dominant model. Hubens et al. (46) recently reported that OPTN was intensively expressed in the retina, including RGCs, in the healthy mouse eyes. There is some evidence that OPTN might play a neuro-protective role by reducing RGCs susceptibility to apoptosis through negatively regulating TNF-α-induced NF-κB activation (47,48). The M98K, evolutionarily conservative in various species (Figure 5), was reported to induce cell death when expressed in RGC-5 (49) and 661W (50) cells, which may indicate the M98K could directly induce the death of RGCs. In this study, we found that the M98K is associate with NTG at a nominal level of significance, but not with HTG patients, in which the myocilin (MYOC) mutations may be the common causes. Besides, the age at diagnosis showed no significant association with M98K mutation in POAG patients, the mean age at diagnosis was 61.07 (95% CI, 57.00–65.14) years in POAG patients with M98K pooled from 4 studies (8,32,36,42).

Some concerns remain in this study, though. Firstly, the effects estimated here were modest although several significant associations were observed, suggesting the possible mild role of this risk allele in POAG patients. Secondly, since publication bias existed, we performed trim-and-fill method to evaluate the influence of these biases. As a result, the association remained statistical significant, although the association should be further studied with a large sample size. Thirdly, only articles published in English and Chinese were included, which may cause language bias.
In summary, our updated meta-analysis provided the most comprehensive role of M98K in the susceptibility of POAG patients. This common variant, M98K, could contribute to POAG susceptibility.
Acknowledgments
Funding: This work was supported by Major Project of National Natural Science Foundation of China (NSFC)-Guangdong Province Joint Fund (grant number 3030902113080); the Science and Technology Planning Project of Guangdong Province (grant number 303090100502050-18); Guangzhou Science and Technology Plan Project (grant number 2018-1202-SF-0019); Research Funds of the State Key Laboratory of Ophthalmology (grant number 30306020240020153, 30306020240020192, 3030902113058, 3030902113118, PT1001022); and Fundamental Research Funds of Sun Yat-sen University (grant number 16ykjc31).
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at http://dx.doi.org/10.21037/aes-20-124
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aes-20-124). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Prum BE Jr, Rosenberg LF, Gedde SJ, et al. Primary Open-Angle Glaucoma Preferred Practice Pattern(®) Guidelines. Ophthalmology 2016;123:41-P111. [Crossref]
- Wang K, Gaitsch H, Poon H, et al. Classification of common human diseases derived from shared genetic and environmental determinants. Nat Genet 2017;49:1319-25. [Crossref] [PubMed]
- Polubriaginof FCG, Vanguri R, Quinnies K, et al. Disease Heritability Inferred from Familial Relationships Reported in Medical Records. Cell 2018;173:1692-704.e11. [Crossref] [PubMed]
- Fingert JH. Primary open-angle glaucoma genes. Eye (Lond) 2011;25:587-95. [Crossref] [PubMed]
- Sarfarazi M, Child A, Stoilova D, et al. Localization of the fourth locus (GLC1E) for adult-onset primary open-angle glaucoma to the 10p15-p14 region. Am J Hum Genet 1998;62:641-52. [Crossref] [PubMed]
- Rezaie T, Child A, Hitchings R, et al. Adult-onset primary open-angle glaucoma caused by mutations in optineurin. Science 2002;295:1077-9. [Crossref] [PubMed]
- Aung T, Rezaie T, Okada K, et al. Clinical features and course of patients with glaucoma with the E50K mutation in the optineurin gene. Invest Ophthalmol Vis Sci 2005;46:2816-22. [Crossref] [PubMed]
- Craig JE, Hewitt AW, Dimasi DP, et al. The role of the Met98Lys optineurin variant in inherited optic nerve diseases. Br J Ophthalmol 2006;90:1420-4. [Crossref] [PubMed]
- Cheng JW, Li P, Wei RL. Meta-analysis of association between optineurin gene and primary open-angle glaucoma. Med Sci Monit 2010;16:CR369-77. [PubMed]
- Chen M, Yu X, Xu J, et al. Association of Gene Polymorphisms With Primary Open Angle Glaucoma: A Systematic Review and Meta-Analysis. Invest Ophthalmol Vis Sci 2019;60:1105-21. [Crossref] [PubMed]
- Wells GA, Shea B, O'Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed July 31 2019.
- Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ 2003;327:557-60. [Crossref] [PubMed]
- Dupont WD, Plummer WD Jr. Power and sample size calculations. A review and computer program. Control Clin Trials 1990;11:116-28. [Crossref] [PubMed]
- Ayala-Lugo RM, Pawar H, Reed DM, et al. Variation in optineurin (OPTN) allele frequencies between and within populations. Mol Vis 2007;13:151-63. [PubMed]
- Alward WL, Kwon YH, Kawase K, et al. Evaluation of optineurin sequence variations in 1,048 patients with open-angle glaucoma. Am J Ophthalmol 2003;136:904-10. [Crossref] [PubMed]
- Melki R, Belmouden A, Akhayat O, et al. The M98K variant of the OPTINEURIN (OPTN) gene modifies initial intraocular pressure in patients with primary open angle glaucoma. J Med Genet 2003;40:842-4. [Crossref] [PubMed]
- He JN, Lu S, Chen LJ, et al. Coding region mutation screening in optineurin in Chinese normal-tension glaucoma patients. Dis Markers 2019;2019:5820537 [Crossref] [PubMed]
- Park J, Kim M, Park CK, et al. Molecular analysis of myocilin and optineurin genes in Korean primary glaucoma patients. Mol Med Rep 2016;14:2439-48. [Crossref] [PubMed]
- Liang S, Huang L, Ying F, et al. Study on optineurin gene polymorphism in Chinese patients with primary open angle glaucoma. Chinese Journal of Experimental Ophthalmology 2013;31:863-6.
- Buentello-Volante B, Elizondo-Olascoaga C, Miranda-Duarte A, et al. Association study of multiple gene polymorphisms with the risk of adult-onset primary open-angle glaucoma in a Mexican population. Exp Eye Res 2013;107:59-64. [Crossref] [PubMed]
- McDonald KK, Abramson K, Beltran MA, et al. Myocilin and optineurin coding variants in Hispanics of Mexican descent with POAG. J Hum Genet 2010;55:697-700. [Crossref] [PubMed]
- Jia LY, Tam PO, Chiang SW, et al. Multiple gene polymorphisms analysis revealed a different profile of genetic polymorphisms of primary open-angle glaucoma in northern Chinese. Mol Vis 2009;15:89-98. [PubMed]
- Caixeta-Umbelino C, de Vasconcellos JP, Costa VP, et al. Lack of association between optineurin gene variants T34T, E50K, M98K, 691_692insAG and R545Q and primary open angle glaucoma in Brazilian patients. Ophthalmic Genet 2009;30:13-8. [Crossref] [PubMed]
- Yen YC, Yang JJ, Chou MC, et al. Absence of optineurin (OPTN) gene mutations in Taiwanese patients with juvenile-onset open-angle glaucoma. Mol Vis 2008;14:487-94. [PubMed]
- Liu Y, Akafo S, Santiago-Turla C, et al. Optineurin coding variants in Ghanaian patients with primary open-angle glaucoma. Mol Vis 2008;14:2367-72. [PubMed]
- Kumar A, Basavaraj MG, Gupta SK, et al. Role of CYP1B1, MYOC, OPTN and OPTC genes in adult-onset primary open-angle glaucoma: Predominance of CYP1B1 mutations in Indian patients. Mol Vis 2007;13:667-76. [PubMed]
- Yao HY, Cheng CY, Fan BJ, et al. Polymorphisms of myocilin and optineurin in primary open angle glaucoma patients. Zhonghua Yi Xue Za Zhi 2006;86:554-9. [PubMed]
- Sripriya S, Nirmaladevi J, George R, et al. OPTN gene: profile of patients with glaucoma from India. Mol Vis 2006;12:816-20. [PubMed]
- Hauser MA, Sena DF, Flor J, et al. Distribution of optineurin sequence variations in an ethnically diverse population of low-tension glaucoma patients from the United States. J Glaucoma 2006;15:358-63. [Crossref] [PubMed]
- Funayama T, Mashima Y, Ohtake Y, et al. SNPs and interaction analyses of noelin 2, myocilin, and optineurin genes in Japanese patients with open-angle glaucoma. Invest Ophthalmol Vis Sci 2006;47:5368-75. [Crossref] [PubMed]
- Weisschuh N, Neumann D, Wolf C, et al. Prevalence of myocilin and optineurin sequence variants in German normal tension glaucoma patients. Mol Vis 2005;11:284-7. [PubMed]
- Rakhmanov VV, Nikitina NY, Zakharova FM, et al. Mutations and polymorphisms in the genes for myocilin and optineurin as the risk factors of primary open-angle glaucoma. Russian Journal of Genetics 2005;41:1295-301. [Crossref]
- Fan BJ, Wang DY, Fan DS, et al. SNPs and interaction analyses of myocilin, optineurin, and apolipoprotein E in primary open angle glaucoma patients. Mol Vis 2005;11:625-31. [PubMed]
- Mukhopadhyay A, Komatireddy S, Acharya M, et al. Evaluation of Optineurin as a candidate gene in Indian patients with primary open angle glaucoma. Mol Vis 2005;11:792-7. [PubMed]
- Jansson M, Wadelius C, Rezaie T, et al. Analysis of rare variants and common haplotypes in the optineurin gene in Swedish glaucoma cases. Ophthalmic Genet 2005;26:85-9. [Crossref] [PubMed]
- Umeda T, Matsuo T, Nagayama M, et al. Clinical relevance of optineurin sequence alterations in Japanese glaucoma patients. Ophthalmic Genet 2004;25:91-9. [Crossref] [PubMed]
- Chen JH, Xu L, Li Y. Study on the optic neuropathy induced response protein gene mutation in Chinese patients with primary open-angle glaucoma. Zhonghua Yi Xue Za Zhi 2004;84:1098-102. [PubMed]
- Baird PN, Richardson AJ, Craig JE, et al. Analysis of optineurin (OPTN) gene mutations in subjects with and without glaucoma: the Blue Mountains Eye Study. Clin Exp Ophthalmol 2004;32:518-22. [Crossref] [PubMed]
- Fuse N, Takahashi K, Akiyama H, et al. Molecular genetic analysis of optineurin gene for primary open-angle and normal tension glaucoma in the Japanese population. J Glaucoma 2004;13:299-303. [Crossref] [PubMed]
- Toda Y, Tang S, Kashiwagi K, et al. Mutations in the optineurin gene in Japanese patients with primary open-angle glaucoma and normal tension glaucoma. Am J Med Genet A 2004;125A:1-4. [Crossref] [PubMed]
- Willoughby CE, Chan LL, Herd S, et al. Defining the pathogenicity of optineurin in juvenile open-angle glaucoma. Invest Ophthalmol Vis Sci 2004;45:3122-30. [Crossref] [PubMed]
- Funayama T, Ishikawa K, Ohtake Y, et al. Variants in optineurin gene and their association with tumor necrosis factor-α polymorphisms in Japanese patients with glaucoma. Invest Ophthalmol Vis Sci 2004;45:4359-67. [Crossref] [PubMed]
- Aung T, Ebenezer ND, Brice G, et al. Prevalence of optineurin sequence variants in adult primary open angle glaucoma: implications for diagnostic testing. J Med Genet 2003;40:e101 [Crossref] [PubMed]
- Leung YF, Fan BJ, Lam DS, et al. Different optineurin mutation pattern in primary open-angle glaucoma. Invest Ophthalmol Vis Sci 2003;44:3880-4. [Crossref] [PubMed]
- Wiggs JL, Auguste J, Allingham RR, et al. Lack of association of mutations in optineurin with disease in patients with adult-onset primary open-angle glaucoma. Arch Ophthalmol 2003;121:1181-3. [Crossref] [PubMed]
- Hubens WHG, Breddels EM, Walid Y, et al. Mapping mRNA Expression of Glaucoma Genes in the Healthy Mouse Eye. Curr Eye Res 2019;44:1006-17. [Crossref] [PubMed]
- Kachaner D, Genin P, Laplantine E, et al. Toward an integrative view of Optineurin functions. Cell Cycle 2012;11:2808-18. [Crossref] [PubMed]
- Sarfarazi M, Rezaie T. Optineurin in primary open angle glaucoma. Ophthalmol Clin North Am 2003;16:529-41. [Crossref] [PubMed]
- Sirohi K, Chalasani ML, Sudhakar C, et al. M98K-OPTN induces transferrin receptor degradation and RAB12-mediated autophagic death in retinal ganglion cells. Autophagy 2013;9:510-27. [Crossref] [PubMed]
- Sayyad Z, Sirohi K, Radha V, et al. 661W is a retinal ganglion precursor-like cell line in which glaucoma-associated optineurin mutants induce cell death selectively. Sci Rep 2017;7:16855. [Crossref] [PubMed]
Cite this article as: Guo C, Yu X, Zhang D, Zhao Z, Zhang J, Zhang M, Sun N, Fan Z. Association between the M98K variant of the OPTN gene and the risk for primary open angle glaucoma: an updated meta-analysis. Ann Eye Sci 2021;6:15.




