
Rapid retinal functional testing
Introduction
Age-related macular degeneration (AMD), glaucoma, and diabetic retinopathy (DR) are the most prominent causes of vision loss in those older than 50 years in the developed world. Screening for each of these diseases is crucial, and eye care currently fails a large segment of our population, with catastrophic consequences in visual disability and cost. Here, we evaluate and review 4 visual function tests that are administered on a tablet for screening in primary care. These tests would also be ideal for combined use with any other screening modalities such as non-mydriatic fundus photography with automated cameras. They can also be easily incorporated into eye clinics for research on these same diseases. In addition to reviewing published results, we also describe in more detail the results of a study with one test, the cone photostress recovery time (PRT) for detection of DME.
PRT is a classic marker for macular disease, such as AMD and DME associated with DR (1,2). After bleaching the photoreceptor photopigment, PRT is the time to recover visual sensitivity (regeneration of photopigment) (3). While of academic interest, this measure of visual function has not become a part of routine evaluation of the macula that is dominated by optical coherence tomography (OCT). However, in the recent paradigm shift to wider screening in primary care settings, and because PRT is affected early in disease (4), a novel, inexpensive photostress recovery test, the Annulus Adaptometer, can screen subjects for early referral.
Other measures of visual function such as brightness-perception, which is a subjective test, and the visual evoked potential (VEP), which is an objective test, provide key information for evaluating the health of the macula and visual pathways. Brightness sense or VEP imbalance between the eyes is a sensitive but nonspecific marker for visual dysfunction. While VEP is unsuitable for primary care, many psychophysical tests of brightness imbalance have been described (relative color saturation (5), Pulfrich effect (6), non-rivalrous brightness sense (7), and rivalrous brightness sense (8), that have been useful for studying a variety of diseases, including macular disease (6,9,10), optic nerve diseases (7,8,11,12), (including glaucoma) (13,14), and amblyopia (6,9,15-17). However, only the brightness sense test is suitable for primary care and telemedicine. Glaucoma in particular is well-poised to be assessed by brightness-perception as brightness asymmetry is very common in glaucoma and is significantly correlated with visual field loss (14).
Color Vision defects in glaucoma are documented in literature (18-22). The common age-related eye diseases of interest, including glaucoma, AMD and DR produce blue-yellow color vision deficiencies, at least in the pre-clinical or early stages (22-24).
Uncorrected refractive error (URE) causes visual disability worldwide (25), and in the underserved in metropolitan areas (Carol Horwitz, Director, Institute for Health Equity Research, personal communication). Deep learning (DL) can measure spherical error (only) from retinal photos (26), but not full refraction. Further, high technology, such as an autorefractor is not practical for screening. Therefore, a practical solution would be a simple app to identify URE for optometric referral, and uncorrectable decreased acuity for ophthalmic referral, a broad safety net for otherwise undetected eye disease.
Such testing modalities could become highly sensitive screenings for early detection of potentially blinding diseases.
Review of literature: method
Due to the fact that retinal functional testing is a broad topic, we did not perform a formal PubMed search, but rather consulted experts in the field of Ophthalmology for references on functional testing modalities for various disorders. These include all known handheld and portable devices suitable for telemedicine. We then reviewed all of those references for possible connections and applications in a primary care clinic. From all such tests, we selected those which fell into the 4 large categories described in the Introduction that have been the most widely researched and have the broadest potential applicability. These are PRT, brightness balance, color vision, and visual acuity. Visual acuity has the evident capacity to detect eye disease in the screening programs to which all these measures are directed, it suffices to demonstrate the simplicity and ease of testing on a portable device, we evaluated the benefits and limitations, while assessing the reproducibility, specificity, and sensitivity for any test. Herein, we review, demonstrate, and evaluate 4 new tests of visual function that are easily administered on a tablet for screening in primary care. We compare these selected high impact functional tests with other devices in the literature. In order to avoid bias towards the testing portfolio on the iPad, we clarify that the goal of the review is to identify the tests’ advantages and application in primary care, and thus only briefly discuss a large number of prior techniques that fail this criterion.
New functional testing methods
General
Four new functional tests, each deployed on a handheld device, are described here in comparison to tests in the literature in each of the four main categories just mentioned. The new tests comprise the iOS DiagnosticGame®, and are deployed on an iPad (iPad mini-4, Apple Inc, Cupertino, CA) with easily understood and intuitive “Touch the Screen” instructions for rapid (~30 sec per eye), streamlined, and automatic administration. The panel of applications includes one in each category: a Cone Photostress test (the Annulus Adaptometer, sensitive to central macular dysfunction), brightness balance perception, color vision testing, and visual acuity check.
Cone photostress recovery time (CPRT) for macular disease
For the photostress recovery test, called the Annulus Adaptometer, the iPad screen luminance is adjusted to 330 cd/m2. This was established as a dependable calibration endpoint by measuring the central luminance of the screens of 5 iPads with an International Light photometer: the range was 331–351 cd/m2, with test/retest variation 1%. The non-dilated eye is then tested with near correction at 16” and fellow eye occluded. The Annulus Adaptometer test takes about one minute per eye. The central 2 degrees of the macula are bleached for 30 seconds by central target fixation at the fixed brightness of the device screen (Figure 1).This is compared in brightness, after bleaching, to an annulus of surrounding unbleached retina of the same eye to measure the cone photostress recovery time (CPRT). The recovery time is the time for the dim appearing central bleached circle to appear of equal brightness to the surrounding annulus of unbleached retina, a relative endpoint that is easily discernible. Hence, the eye serves as its own control. Further, bleaching for 30 secs generates a sufficiently dark after image for comparison, much less than the time for peripheral rod spot bleaching (4).
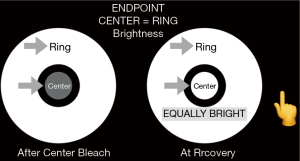
CPRT was measured in a group of 37 diabetics who also underwent spectral domain OCT (SD-OCT) measurement of central foveal thickness (CFT) at the NYU Medical Center diabetes outpatient clinic. DME was defined as CFT >300 µm. The CPRT was then correlated with presence or absence of DME (15-17).
Brightness balance perception—optic nerve function and glaucoma
Brightness perception by the two eyes is precisely balanced in humans, and the Amblyometer® of the iOS DiagnosticGame®, detects when brightness is imbalanced (Figure 2). The test is based on the principle of the Wheatstone bridge, an electrical circuit used to measure an unknown electrical resistance with high precision by balancing two legs of a bridge circuit, and is the first such vision test (https://patents.google.com/patent/US9560960B2/en). Reduced brightness in one eye causes conduction delay of nerve impulses from that eye to the brain, which is recorded as a prolonged latency in the VEP (27). In glaucoma, visual fields assess focal damage, which is summated by the mean deviation (MD) index for comparison to global nerve loss measured by VEP or brightness sense imbalance.
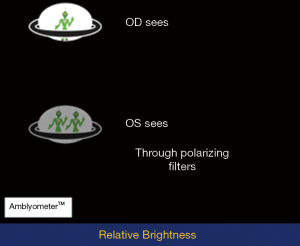
The subject wears polarizing glasses of opposite polarity on the right and left eyes, and observes test objects that are also polarized to dissociate the perceptions of the two eyes under binocular viewing. The subject then touches the brighter object, while the program titrates individual brightness with staircase algorithms, aka ascending and descending methods of limits (13). The mean and variance of 4 repetitions are calculated, and high variance requires repeat testing. The mean testing time (4 tests) is 2 mins. The difference between the two eyes, if any, is measured in steps of 0.3 log units of stimulus brightness.
This game was tested on 286 school children for accuracy in detecting amblyopia.
Color vision screening—optic nerve and macular disease
The automated NeuroColor® game of the DiagnosticGame® app was designed to detect hereditary and acquired color vision (CV) defects. It presents copies of the AO Hardy-Rand-Rittler (HRR) pseudoisochromatic color plates on the iPad so that only touching the color is required.
In this game, the color plates are: (I) a demonstration plate visible to all except those malingering or totally blind, (II) a control plate with no color symbols, (III) 2 plates for detecting red-green defects and (IV) 2 plates for detecting blue-yellow defects (Figure 3). If a plate is missed, it is presented once more. This game was tested on 211 children for accuracy in measuring color vision in comparison to the gold standard HRR color plates.

Visual acuity (VA) testing
The visual acuity test, the digital tumbling E visual acuity (TEVA) game of the DiagnosticGame® app, is based on matching vertically aligned Es (or Cs), and is independent of literacy (Figure 4). The best vision protocol includes testing with a +2.5 D lens over the distance glasses, and, for visual acuity less than 20/40, the addition of a pinhole disc simulating universal focus. The accuracy of this testing is reliant on the appropriate distance between the iPad and the patient’s eyes, and therefore proper distance is needed for accurate results. The possible abnormal outcomes and proposed dispositions are: uncorrected refractive error (URE) for optometric referral, and uncorrectable decreased acuity for ophthalmic referral. This Digital TEVA at 16 inches was tested on 65 children.

Validation of new methods
Cone photostress recovery time and retinal disease
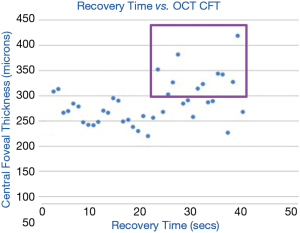
The CPRT in 37 diabetics was associated with the severity of DME (28): recovery time greater than 21 seconds correlated significantly with central foveal thickness (CFT) >300 µm as measured by OCT (P=0.02, Chi square). True positives are points included in the purple box (Figure 5). As a test for DME, sensitivity was 83% and specificity was 55%.
Brightness balance perception—validation and amblyopia
The Amblyometer® achieved a sensitivity and specificity of 100% for identifying amblyopic children in an office practice (16). It was further validated through screening 208 school children, with highly correlated test and retest scores (r2=0.985, P<0.00001) (16), and the accuracy for amblyopia detection was as follows: Of the 208 children that were recruited as subjects and tested at school, 121 were girls and 87 were boys with ages from 3 to 14 years and a mean of 7.8 years. In screening these children, 2 amblyopes were detected (2 true positives) and 206 true negatives (17).
Color vision screening
The NeuroColor® game was validated in school children, (American Academy of Pediatrics, 2019) (17) in comparison with the AO HRR plates. 211 children were tested, 205 children tested were normal, and 6 children displayed a color vision deficiency with both the AO HRR color plates and the NeuroColor® game, identical results. Further, of the 6 abnormal results, 5 were bilaterally identical, classified as hereditary CV deficiency by both tests. The 1 acquired defect was monocular and occurred in the amblyopic eye of a 10-year-old. The mean testing time was 53 seconds.
Visual acuity (VA) testing
Digital tumbling E visual acuity (TEVA) at 16 inches was validated (17) by comparison to Snellen acuity with letter sizes of the same visual angles on 65 children. The results were significant correlated and no significant differences were found. The mean testing time for both eyes was 89 seconds. The mean difference between iPad and E-Chart visual acuities with pinhole was 0.02 logMAR, with 95% limits of agreement from −0.08 to +0.11 logMAR.
Comparisons of testing methods and discussion
A strength of functional tests for screening is that they are generally not specific for one disease, such as Color fundus photography (CFP) specificity for AMD or DR, but rather are sensitive to many vision disorders that merit specialist referral. This low specificity and high sensitivity is key to effectively screening, rather than diagnosing, eye diseases. However, a desirable specificity still applies, in that a positive test has a high probability of identifying a true positive that is meaningful in population screening, i.e., a significant vision disorder. Further, they provide strong protection against false negatives from other tests, such as from AI, that could “pass” on serious disease.
Cone photostress recovery time and macular disease
A major test advantage and innovation of the CPRT on the iPad is that it is relative: a bleached center is compared to the unbleached surround. Further, the test is quick, about a minute per eye, and is sensitive, e.g., to DME (28) (Figure 5). The abnormally increased recovery times in the diabetic subjects with normal CFT may be due to other DR pathology to be determined. Thus, the lower specificity is not necessarily a disadvantage here, and in fact may be useful in research and eventually clinical care. For example, prolonged recovery times may be due to macular ischemia in the absence of edema.
Research has shown that PRTs are delayed with aging, AMD, diabetic retinopathy, and central serous retinopathy. Repeatability was excellent in some, e.g., Newsome et al. (29), but not others (30,31). The existing tests are high quality, but still require specialized equipment, expert personnel, and therefore remain impractical, especially in a primary care setting. For example, the method of Newsome is considered excellent, and yet is not suitable for the reasons given. Thus, although good repeatability can be achieved in modern research settings (4,32), there is currently only one reliable photostress test proposed for detecting disease economically and easily, the cone photostress test (CPRT) on the iPad. It is quite analogous to the Newsome test, also with good repeatability, and thus just as effective in screening. However, in contrast to the Newsome test, the CPRT on the iPad, a widely available, inexpensive commercial device, provides eminent practicality. In the CPRT, the central bleaching and measured recovery in brightness to surrounding unbleached retina is impressively all done on the iPad, providing complete ease for patients.
Many devices used varying methods of bleaching and endpoints. Technical expertise and cost also vary (28,30,31,33). With the AdaptDx dark adaptometer (4), delayed rod recovery time, beyond 6.5 minutes, was a 91% sensitive and specific early marker for AMD (4). However, the adaptation time reported of cone recovery in a central photostress test was also significantly prolonged in most (69%) AMD eyes (34). Required testing was much shorter than with the AdaptDx. Normal recovery was complete in 20 to 50 seconds, and longer in most AMD subjects. Further research comparing rod dark adaptation (DA) and cone PRT found both had excellent diagnostic capacity for early AMD (32), suggesting cone testing is rapid and effective in screening. In addition, the Annulus Adaptometer has a more comfortable luminance (~330 cd/m2) than other photostress tests (34).
Brightness balance perception—optic nerve function and glaucoma
In glaucoma, visual fields assess focal damage and global nerve loss is measured by VEP, which is summated by the MD (mean deviation) index for comparison to or brightness-sense imbalance. In glaucoma patients, the MD index significantly correlated with prolonged VEP latency (35) and similarly for the pattern reversal VEP.(11) More important for screening, brightness-sense imbalance in a validated office test analogous to the Amblyometer® of the iOS DiagnosticGame®, was found in 100% of 20 primary open angle glaucoma (POAG) patients and in only 3 of 61 age-matched controls (13); in another study of POAG patients, 86% of 28 (14) patients had brightness imbalance significantly correlated with visual field loss. The Amblyometer® of the iOS DiagnosticGame®, which detects when brightness is imbalanced, is analogous to measuring an unknown electrical resistance with high precision by balancing two legs of a Wheatstone Bridge circuit, the first vision test of its kind. The high sensitivity of this tool can capture glaucoma patients and others that need referral.
Color vision screening
Color vision screening is of particular importance because color vision defects in glaucoma are documented in literature (18-22). B/Y defects in particular increase with age, and with the presence of cataract. Our common age-related eye diseases of interest (glaucoma, AMD and DR) all produce blue-yellow color vision deficiencies, at least in the pre-clinical or early stages (22-24), and therefore they are non-specific. However, only 20% of those younger than 75 years of age, patients with cataracts included, made errors on the Adams desaturated D-15 (36) but B/Y defects were 30-50% in ages greater than 75, which was mostly due to aging and cataract. This suggests that subjects younger than 75 with B/Y defects, but not older, should be referred for evaluation. The NeuroColor® game has been validated, and will serve these functions easily, as has just been shown. This compares favorably to complex tests of hue discrimination and allows quick, effective testing on a handheld device equivalent to standard isochromatic plate testing.
Visual acuity testing
The digital tumbling E visual acuity game for testing in the office or for screening is easily accessible to assess patients’ visual status. Individuals are asked to identify shapes without the need to read letters, and therefore literacy is not required for (Figure 4). The possible abnormal outcomes and proposed dispositions are widely applicable to general populations: uncorrected refractive error (URE) for optometric referral, and uncorrectable decreased acuity for ophthalmic referral
Conclusions
With telehealth programs currently expanding rapidly in the face of COVID-19, and clearly here to stay, these accessible, user-friendly, cheap, and simple screening methods will be an ideal part of this future. These screening tests allow patients to be identified in the early stages of disease for referral to specialists, proper assessment and treatment.
Acknowledgments
These results have been presented in part at Association for Research in Vision and Ophthalmology meetings and meetings of the American Academy of Pediatrics.
Funding: This work was supported in part by a Challenge Grant from Research to Prevent Blindness, NY. The functional tests (iOS DiagnosticGame®) were invented by Albert Hofeldt, MD. CEO of AMA Optics (Miami, FL). The software is patent pending.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Eye Science for the series “Retinal Imaging and Diagnostics”. The article has undergone external peer review.
Conflicts of Interest: The authors have completed the ICMJE uniform disclosure form (available at https://dx.doi.org/10.21037/aes-21-22). The series “Retinal Imaging and Diagnostics” was commissioned by the editorial office without any funding or sponsorship. Dr. Smith served as the unpaid Guest Editor of the series, and serves as an unpaid editorial board member of Annals of Eye Science from May 2019 to Apr 2021. Dr. Smith reports a Challenge Grant from Research to Prevent Blindness, NY. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Sohrab MA, Smith RT, Salehi-Had H, et al. Image registration and multimodal imaging of reticular pseudodrusen. Invest Ophthalmol Vis Sci 2011;52:5743-8. [Crossref] [PubMed]
- Greenstein VC, Shapiro A, Hood DC, et al. Chromatic and luminance sensitivity in diabetes and glaucoma. J Opt Soc Am A Opt Image Sci Vis 1993;10:1785-91. [Crossref] [PubMed]
- Severin SL, Harper JY Jr, Culver JF, et al. Photostress test for the evaluation of macular function. Arch Ophthalmol 1963;70:593-7. [Crossref] [PubMed]
- Jackson GR, Scott IU, Kim IK, et al. Diagnostic sensitivity and specificity of dark adaptometry for detection of age-related macular degeneration. Invest Ophthalmol Vis Sci 2014;55:1427-31. [Crossref] [PubMed]
- Danesh-Meyer HV, Papchenko TL, Savino PJ, et al. Brightness sensitivity and color perception as predictors of relative afferent pupillary defect. Invest Ophthalmol Vis Sci 2007;48:3616-21. [Crossref] [PubMed]
- Hofeldt AJ, Hofeldt GT, Weiss MJ, et al. Pulfrich psychophysical stereo-illusion and visual-evoked potentials in epiretinal membrane of the macula. J Neuroophthalmol 1996;16:36-40. [Crossref] [PubMed]
- Sadun AA, Lessell S. Brightness-sense and optic nerve disease. Arch Ophthalmol 1985;103:39-43. [Crossref] [PubMed]
- MacMillan ES, Cummins D, Heron G, et al. The simultaneous interocular brightness sense test. A test of optic nerve function. Arch Ophthalmol 1994;112:1190-7. [Crossref] [PubMed]
- Sokol S, Bloom B. Visually evoked cortical responses of amblyopes to a spatially alternating stimulus. Invest Ophthalmol 1973;12:936-9. [PubMed]
- Bass SJ, Sherman J, Bodis-Wollner I, et al. Visual evoked potentials in macular disease. Invest Ophthalmol Vis Sci 1985;26:1071-4. [PubMed]
- Ruchi K, Ramji S, Smita S, et al. The Potential Use of Pattern Reversal Visual Evoked Potential for Detecting and Monitoring Open Angle Glaucoma. Curr Neurobiol 2012;3:39-45.
- Kothari R, Bokariya P, Singh S, et al. Significance of Visual Evoked Potentials in the Assessment of Visual Field Defects in Primary Open-Angle Glaucoma: A Review. Neurosci J 2013;2013:418320 [Crossref] [PubMed]
- Cummins D, MacMillan ES, Heron G, et al. Simultaneous interocular brightness sense testing in ocular hypertension and glaucoma. Arch Ophthalmol 1994;112:1198-203. [Crossref] [PubMed]
- Teoh SL, Allan D, Dutton GN, et al. Brightness discrimination and contrast sensitivity in chronic glaucoma--a clinical study. Br J Ophthalmol 1990;74:215-9. [Crossref] [PubMed]
- Hofeldt TS, Hofeldt AJ. Measuring colour rivalry suppression in amblyopia. Br J Ophthalmol 1999;83:1283-6. [Crossref] [PubMed]
- Gise R, Kane S. Detection of Amblyopia in Young Children via Retinal Rivalry Using a Video Game Style Interface on a Tablet Device. Invest Ophthalmol Vis Sci 2016;57.
- Kane S. Automated Brightness Sense Screening for Amblyopia. Pediatrics 2019;144 (2 MeetingAbstract):240.
- Pacheco-Cutillas M, Edgar DF, Sahraie A, et al. Acquired colour vision defects in glaucoma-their detection and clinical significance. Br J Ophthalmol 1999;83:1396-402. [Crossref] [PubMed]
- Pacheco-Cutillas M, Barbur JL, Edgar DF. Chromatic and achromatic loss of sensitivity in glaucoma. Ophthalmic Physiol Opt 2002;22:574. [Crossref]
- Papaconstantinou D, Georgalas I, Kalantzis G, et al. Acquired color vision and visual field defects in patients with ocular hypertension and early glaucoma. Clin Ophthalmol 2009;3:251-7. [PubMed]
- Ouchi J, Kunikata H, Omodaka K, et al. Color visual acuity in preperimetric glaucoma and open-angle glaucoma. PLoS One 2019;14:e0215290 [Crossref] [PubMed]
- Adams AJ, Rodic R, Husted R, et al. Spectral sensitivity and color discrimination changes in glaucoma and glaucoma-suspect patients. Invest Ophthalmol Vis Sci 1982;23:516-24. [PubMed]
- Lovie-Kitchin J, Feigl B. Assessment of age-related maculopathy using subjective vision tests. Clin Exp Optom 2005;88:292-303. [Crossref] [PubMed]
- Feitosa-Santana C, Paramei GV, Nishi M, et al. Color vision impairment in type 2 diabetes assessed by the D-15d test and the Cambridge Colour Test. Ophthalmic Physiol Opt 2010;30:717-23. [Crossref] [PubMed]
- Resnikoff S, Pascolini D, Mariotti SP, et al. Global magnitude of visual impairment caused by uncorrected refractive errors in 2004. Bull World Health Organ 2008;86:63-70. [Crossref] [PubMed]
- Varadarajan AV, Poplin R, Blumer K, et al. Deep Learning for Predicting Refractive Error From Retinal Fundus Images. Invest Ophthalmol Vis Sci 2018;59:2861-8. [Crossref] [PubMed]
- Froehlich J, Kaufman DI. Effect of decreased retinal illumination on simultaneously recorded pattern electroretinograms and visual-evoked potentials. Invest Ophthalmol Vis Sci 1991;32:310-8. [PubMed]
- Boddu S, Nabati C, Saade C, et al. Use of a retinal acuity meter (RAM) and brightness acuity meter (BAM) to identify early diabetic macular edema. Invest Ophthalmol Vis Sci 2014;55:2292.
- Newsome DA, Negreiro M. Reproducible measurement of macular light flash recovery time using a novel device can indicate the presence and worsening of macular diseases. Curr Eye Res 2009;34:162-70. [Crossref] [PubMed]
- Wolffsohn JS, Anderson SJ, Mitchell J, et al. Effect of age related macular degeneration on the Eger macular stressometer photostress recovery time. Br J Ophthalmol 2006;90:432-4. [Crossref] [PubMed]
- Loughman J, Ratzlaff M, Foerg B, et al. Suitability and repeatability of a photostress recovery test device, the macular degeneration detector (MDD-2), for diabetes and diabetic retinopathy assessment. Retina 2014;34:1006-13. [Crossref] [PubMed]
- Dimitrov PN, Robman LD, Varsamidis M, et al. Visual function tests as potential biomarkers in age-related macular degeneration. Invest Ophthalmol Vis Sci 2011;52:9457-69. [Crossref] [PubMed]
- Glaser JS, Savino PJ, Sumers KD, et al. The photostress recovery test in the clinical assessment of visual function. Am J Ophthalmol 1977;83:255-60. [Crossref] [PubMed]
- Phipps JA, Guymer RH, Vingrys AJ, et al. Loss of cone function in age-related maculopathy. Invest Ophthalmol Vis Sci 2003;44:2277-83. [Crossref] [PubMed]
- Nykänen H, Raitta C. The correlation of visual evoked potentials (VEP) and visual field indices (Octopus G1) in glaucoma and ocular hypertension. Acta Ophthalmol (Copenh) 1989;67:393-5. [Crossref] [PubMed]
- Schneck ME, Haegerstrom-Portnoy G, Lott LA, et al. Comparison of panel D-15 tests in a large older population. Optom Vis Sci 2014;91:284-90. [Crossref] [PubMed]
Cite this article as: Weitzman SA, Greenstein V, Bhuiyan A, Hofeldt A, Kane S, Smith RT. Rapid retinal functional testing. Ann Eye Sci 2021;6:26.


