
Cytomegalovirus retinitis in the highly active anti-retroviral therapy era
Introduction
Cytomegalovirus (CMV) is a member of the herpesvirus family and is a well-known opportunistic infection in patients who are immunosuppressed (1). Specifically, for patients with late-stage AIDS, ocular involvement of the virus was the leading cause of blindness in developed countries (2). CMV retinitis previously affected one-third of patients with AIDS, with most vision loss due to macula-involving retinitis or retinal detachment (1,3). Risk factors for CMV infection include CD4 T-lymphocyte counts less than 50 cells/mL and among patients with this CD4 count, the rate of infection of the retina was 20 cases/100 person-years (PY) (3). Since the advent of highly active antiretroviral therapy (HAART), Western countries have seen an 80% reduction in the incidence of CMV retinitis, with an estimated rate of CMV retinitis of 5.6 cases/100 PY (4). However, CMV retinitis has not completely disappeared. Specifically, patients can still develop CMV retinitis if they are either HAART unresponsive or HAART naïve, and may be faced with serious complications of the disease and treatment (4-6). HARRT failure is defined as having high HIV viral load and or low CD4 count despite treatment. CMV retinitis can also present in patients that have been immunosuppressed for other reasons including transplantation and systemic immunosuppressive therapy.
Clinical diagnosis and characteristics
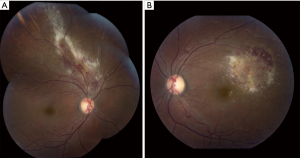
Diagnosis of CMV retinitis is made by indirect ophthalmoscopy with a dilated pupillary exam (1). Patients can present symptomatically with decreased visual acuity, floaters, blurred vision, or a visual field defect/scotoma. The fulminant type of CMV retinitis is characterized by yellow-white, fluffy, or granular retinal lesions, usually located near retinal vessels and associated with hemorrhage (Figure 1), while the indolent form of the disease presents as peripheral granular opacities with occasional hemorrhage (7-10). Rarely, CMV presents as a frosted branch angiitis described as retinal sheathing predominantly around the veins but also involves the arteries (11). Histologically, full-thickness necrosis is observed that progresses towards the posterior pole (12). The areas of the eye effected by the retinitis is categorized into zones of involvement. Zone 1 involves the area within 500 micrometers of the optic nerve or within 3,000 micrometers of the center of the macula. Zone 2 extends from zone one to the clinical equator of the eye, and zone 3 extends from zone 2 to the ora serrata (9,13,14). All of these exam findings in the setting of immunocompromise are suggestive of CMV retinitis (9,12).
It has been suggested that the clinical characteristics of CMV retinitis may have changed since the development and use of HAART in patients with HIV/AIDS (15). In those who have failed HAART, studies have shown that patients frequently present with less lesion opacification, which correlates with less viral activity (3). Because these patients have lower levels of viral replication, their lesions are of lesser severity (3). Additionally, individuals categorized as HAART failure patients are usually asymptomatic, have bilateral retinitis, better visual acuity, and less zone 1 disease (3,9,10). In a more recent report, it was noted in HAART failure patients when compared to HAART responsive patients with CMV retinitis, that the failure group had a higher chance of developing a relapse and more commonly had a three-line loss of vision (16). These differing studies stress the importance of follow-up and close monitoring, especially in at-risk populations.
CMVR in Non-HIV patients
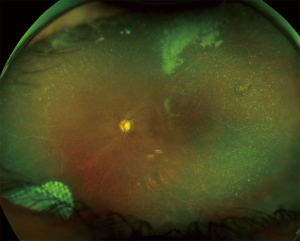
CMV retinitis can develop in immunocompromised patients who are HIV negative, including patients with solid organ or hematopoietic stem cell transplants, intraocular steroids (Figure 2), and in patients requiring immunosuppressive treatment for other diseases (17). Although rare, it has also been reported in immunocompetent patients; this includes patients with some immune dysfunction, such as diabetics and elderly patients, and those on cytotoxic medications (18). CMV retinitis in non-HIV patients has a non-classical presentation, now termed chronic retinal necrosis. Patients with chronic retinal necrosis, which differs phenotypically from CMV retinitis seen in AIDS patients, may present with a more indolent, granular retinitis, pan-retinal occlusive vasculitis, and is associated with more severe intraocular inflammation in comparison to the limited extent of the retinitis (18-20). In a study by Davis et al., 71% of elderly immunocompetent patients with CMV retinitis also had concomitant retinal arteriolar occlusions, and were more likely to have vascular occlusions compared to HIV-infected patients (21). This atypical presentation is similar to that seen in HSV and VZV infections with features of acute retinal necrosis (ARN) (18). Other studies have also shown that the visual prognosis and complications from CMV retinitis in HIV-positive and HIV-negative patients (e.g., incidence of retinal detachment, zonal involvement and bilateral versus unilateral disease) were comparable (22).
Clinical course
In patients with unilateral CMV retinitis, precautions are taken to reduce the incidence of second eye involvement including early detection and timely treatment. The Longitudinal Studies of the Ocular Complications of AIDS showed that patients treated with HAART were still at risk for developing contralateral eye disease if their CD4+ cell count was <50 mL (23). Additionally, previous studies have shown that HAART has been associated with decreased rates of retinitis progression (6,23). and visual acuity loss (6), decreased risk of retinal detachment (15), and overall improved survival in patients with CMV retinitis, without reaching the threshold CD4+ cell count that is associated with treatment (above 100 cells/mL) (24).
Typically, the greatest risk of developing CMV retinitis occurs when the CD4+ cell count drops below 50 cell/mL (14,24). The goal of treatment for CMV retinitis is to reach a threshold of greater than 100 cells/mL. This is because the risk of progression is 30-fold less compared to the progression of retinitis prior to the HAART era (6). HAART raises the CD4+ cell count, leading to less of the population being affected by CMV retinitis (24). It allows for HIV suppression in the blood, leading to immune recovery and specific anti-CMV immunity (6).
In a study by Jabs et al., rates for progression of CMV retinitis were higher in those with newly-diagnosed CMV retinitis compared to those with previously diagnosed CMV retinitis (25). Knowing this, it is important to provide thorough screening of CMV retinitis in at-risk populations, given that patients may have mild visual symptoms and despite ophthalmic disease manifestations (1,13,18,19,26,27). It is suggested that patients with CD4+ cell counts <50 cells/µL should be screened every two to three months, 51 to 250 cells/µL should be screened every three to four months, and 250 to 500 cells/µL should be screened every five to six months (18,20,22). A dilated fundus examination performed by an ophthalmologist is highly sensitive and specific for identifying CMV retinitis (1,7,14,28,29).
Treatment
CMV retinitis should be treated promptly due its rapid progression if not managed. It will typically progress towards the posterior pole at an average rate of 24 micrometers per day (30), and the zone of retinitis grows at a rate of 750 microns every three weeks (1). The progressive necrotizing retinitis can cause destruction of the entire retina in three to six months if untreated (1). HAART has been pivotal in the role of immune reconstitution in patients with HIV/AIDS suffering from CMV retinitis. Its administration results in improved CD4+ cell counts, reduced HIV replication, and reduced mortality in those with HIV/AIDS (5,28). For this reason, immunocompromised hosts with CMV retinitis should first begin HAART before initiating other treatment (8,9,30). The different modes of therapy include systemic, intravitreal, and combined systemic and intravitreal treatment. The goals of therapy are to stop acute inflammation and delay the time to relapse (10).
Systemic therapy
Systemic antivirals remain the gold standard of treatment for CMV retinitis, given its efficacy and contribution to a reduction in overall morbidity and preventing disease in the contralateral eye if unilateral disease is present (31). First-line treatment for both HIV-positive and HIV-negative patients with CMV retinitis is typically induction therapy with either intravenous (IV) ganciclovir or oral valganciclovir (31-33), followed by maintenance therapy (12,33,34). Oral ganciclovir has poor bioavailability (only 6–9%), and therefore is not the preferred treatment for CMV retinitis induction therapy (35). It is typically reserved for maintenance therapy and primary prophylaxis (3,10).
Ganciclovir is a nucleoside analog that acts against human CMV (HCMV) by preferentially phosphorylating HCMV-infected cells by the virus-encoded UL97 kinase, which essentially inhibits DNA synthesis (36). Induction therapy is given at a dose of 5 mg/kg every 12 hours for 14–21 days, and can also be used as maintenance therapy at a dose of 5 mg/kg/day (10,30). Limiting factors of long-term treatment with IV ganciclovir include side effects such as myelosuppression (neutropenia, anemia, and thrombocytopenia), catheter-related sepsis, as well as the development of resistance (3,10,37,38).
Valganciclovir is an oral prodrug that is rapidly converted to ganciclovir in the body (30,39). It has been shown to be as effective as IV ganciclovir, and because of its high bioavailability, it can be used as both an induction and maintenance therapy. Martin et al. compared the effects of induction therapy using oral valganciclovir with IV ganciclovir, and found that using twice daily 900 mg of oral valganciclovir was similar to IV ganciclovir (39). Additionally, oral valganciclovir can avoid catheter-related sepsis related to parenteral antiviral administration (3,30,39,40). Boivin and colleagues found that the rate of emergence of ganciclovir-resistant viruses is no greater with the use of valganciclovir versus IV ganciclovir, although valganciclovir is commonly associated with a low incidence of viral resistance (3,41).
Foscarnet is typically used as a second-line therapy for CMV retinitis, particularly for CMV retinitis that is resistant to ganciclovir or valganciclovir, or for patients who cannot be treated with ganciclovir due to dose-limiting neutropenia or leukopenia (3,37). Jab et al. found that of patients of the 44 foscarnet-treated patients in their prospective observational study, about one-third developed resistance to foscarnet, and possibly at similar rates to those of ganciclovir resistance (42). Nonetheless, the choice of therapy continues to rely on clinical decision-making and monitoring of treatment response. The Studies of Ocular Complications of AIDS research trial revealed that foscarnet is equivalent to IV ganciclovir for induction and maintenance therapy for HIV-associated CMV retinitis (10,43,44). Induction therapy consists of 180 mg/kg total daily and 90 mg/kg/day for maintenance, ranging from weeks to months, given intravenously (3). It has been shown that in patients with HIV/AIDS and CMV retinitis, foscarnet offers a survival benefit over treatment with IV ganciclovir (45). However, a potential risk of foscarnet is related to nephrotoxicity and electrolyte abnormalities (46).
Cidofovir is another antiviral medication administered intravenously that has activity against CMV. A randomized control trial compared cidofovir to oral and intravitreal ganciclovir, and found that both groups had similar efficacy in treating CMV retinitis (30,47). The administration of cidofovir is also limited by its adverse effects, such as nephrotoxicity, neutropenia, ocular inflammation, and ocular hypotony (47,48). For this reason, probenecid is given before and after infusion of cidofovir, as well as IV fluids, in order to decrease renal toxicity (10,31,33).
The use of systemic therapy with ganciclovir, foscarnet, and cidofovir, have been related to resistance involving the viral genome and viral DNA polymerase mutations, UL97 and UL54 respectively (49,50). Ganciclovir resistance mutations have been identified in both the UL97 and UL54 genes, whereas foscarnet and cidofovir have only been associated with UL54 mutations (51,52). Resistance remains a challenge for immunocompromised patients who require long-term anti-CMV therapy. However, due to the immunological benefits of HAART in HIV/AIDS patients, the incidence of HCMV drug resistance has decreased significantly (53,54). Studies have shown that CMV viral load can help identify patients who are unlikely to develop resistance to ganciclovir or those who are at increased risk of developing resistance (high negative predictive value) (53,55). Consequently, management of CMV retinitis has evolved to include treatment that targets viral genome sites different from UL97 and UL54. Letermovir, a drug that targets UL56 (CMV viral terminase complex), is currently only FDA approved as a prophylactic drug for CMV retinitis, but has been studied as a potential antiviral treatment in patients with resistance to other therapies (56).
Intravitreal therapy
Ganciclovir and foscarnet can be administered via intravitreal injection and have been used for the treatment of CMV retinitis. The intraocular ganciclovir implant demonstrated efficacy for the treatment of CMV retinitis but is no longer manufactured for clinical use. The rationale for intravitreal therapy is that administration of these medications to the location of the infectious process can inhibit viral concentrations in the vitreous, while avoiding complications that may accompany systemic therapy. Studies have shown that weekly injections of intravitreal ganciclovir are effective in treating the acute infectious stage of the disease (57,58). However, use of any of these medications as a sole therapy is uncommon; it is often used in conjunction with systemic treatment or as short-term management (35). In one study, 17 (70.8%) eyes of non-HIV patients had a negative DNA viral load one month after treatment with high-dose intravitreal ganciclovir, and the mean CMV DNA load in aqueous humor had decreased from about 2.6×105 copies/mL at baseline to about 1×104 copies/mL (59). Additionally, Jabs and colleagues found that generally, patients who are treated with intravitreal injections alone fared more poorly than patients treated with regimens where systemic antiviral was employed (60). However, monotherapy with intravitreal injections is associated with a 22–35% incidence of new CMV retinitis occurring in the contralateral eye (1,12).
Combination therapy
In cases of resistant CMV retinitis, a combination of IV ganciclovir and foscarnet, or oral valganciclovir and IV foscarnet, has been found to be more effective than monotherapy (31,57). In one study, patients with unilateral cytomegalovirus retinitis were randomly assigned to either a ganciclovir implant plus oral ganciclovir, a ganciclovir implant plus oral placebo, or IV ganciclovir alone. Results showed that after one year, patients on oral ganciclovir or IV ganciclovir took a longer time to develop new CMV disease than those receiving the placebo (61). Patients who have had previous treatment for CMV retinitis, have relapsed and can tolerate the medications, can try a combination of foscarnet and ganciclovir, or any combination of systemic and intraocular therapy for controlling CMV retinitis.
Prophylaxis
Valganciclovir, oral ganciclovir, and letermovir are medications that have been used for CMV prophylaxis. However, since the introduction of HAART, anti-CMV prophylactic treatment for opportunistic infections in patients with HIV has decreased. Before HAART, patients with CMV retinitis were advised to remain on anti-CMV treatment as a lifelong therapy (62). Now, maintenance therapy (or secondary prophylaxis) is recommended to be continued until adequate immune recovery due to the effectiveness of HAART (63). A prospective study by Berenguer et al. supports this. They found that in the 35 patients who were on HAART for 75 weeks (median) and had a median CD4+ count of 287 cells/mL, there was no progression of CMV retinitis after cessation of secondary prophylaxis (62). In most patients, immune recovery from HAART can allow for the discontinuation of anti-CMV medications without rebound activation, and in some patients, HAART alone is sufficient in suppressing CMV retinitis (3).
Immune reconstitution usually occurs 3 to 6 months after initiation of HAART (33,64). Patients who are considered for discontinuation of anti-CMV medications are those whose non-sight threatening retinitis has been quiescent for six months and who have immune reconstitution with a CD4+ cell count above 100 cells/mL (3). A study by Jabs et al. showed that following immune recovery, patients may remain off anti-CMV therapy. However, longer follow-up is needed to determine at what CD4+ count treatment should be reinitiated to prevent disease relapse (65). Therefore, it is important to be vigilant with routine follow-up examinations in order to catch recurring disease in those who have discontinued treatment. In general, treatment and prophylaxis should be individualized based on the location and size of the retinitis, adverse effects of the medications, and the patient’s experience with HAART (3).
Complications
Treatment for CMV retinitis is also dependent on the timing of the commencement of HAART. Studies have suggested that patients with preexisting CMV retinitis who are treated with HAART can experience immune recovery uveitis (12). The incidence is estimated to be 5 to 30 percent of patients with preexisting CMV retinitis starting HAART (5). Specifically, immune recovery uveitis is likely the result of previously exposed antigens from CMV retinitis that are now recognized by the improved immune response (5,30,64). This manifests as an anterior or intermediate uveitis and vitritis, and is associated with structural ocular complications such as cystoid macular edema, cataracts, and epiretinal membrane formation (6,9,30,64). Because this phenomenon can cause significant vision loss, treatment with oral or periocular steroids may be needed for the further improvement of visual acuity via reduction of cystoid macular edema (5,30).
Rhegmatogenous retinal detachments occur in about 20 to 30 percent of patients with CMV retinitis (31). Retinal detachments occur due to necrotic breaks in the retina, but can also result from peripheral anterior lesions on the retina (this location is particularly thin and is prone to breaks and tears) (9,66). The anterior retina also underlies the vitreous base, making a lesion in this area more likely to cause vitreoretinal traction (2). Studies have shown that the major risk factors associated with retinal detachments in CMV retinitis are an increased area of retinal involvement, the size of the lesion (at diagnosis and at the visit before detachment), a low CD4+ cell count, and bilateral disease (1,2,23,66). Additionally, studies have shown that the use of HAART has the greatest effect on the risk of retinal detachment, by decreasing the risk by 60%, an effect that is enhanced in patients that exhibit immune recovery (15).
Conclusions
CMV retinitis is a vision-threatening opportunistic infection that may still affect patients living with HIV and AIDS in the post-HAART era. Following HIV and anti-CMV therapy, immune recovery may result in adverse complications such as immune recovery uveitis, and treatment with anti-CMV medications may lead to side effects that require laboratory and health monitoring. CMV retinitis in the post-HAART era carries a much better prognosis, and incidence has significantly declined. However, because these patients still have increased mortality rates than the rest of the population, immunosuppressed patients should be properly screened, with guidance dependent on the CD4+ counts. Moreover, CMV retinitis may complicate the clinical course of other patients with systemic and local immunosuppressed status.
Acknowledgments
Funding: This project was supported by the National Eye Institute/National Institutes of Health core grant P30-EY06360 (Department of Ophthalmology, Emory University School of Medicine), National Eye Institute, National Institutes of Health R01 EY029594 (Yeh) and K23 EY030158 (Shantha). Funding support was also provided via an Unrestricted Grant from Research to Prevent Blindness (Emory Eye Center, Emory University School of Medicine). Research support has also been provided by the Association for Research in Vision and Ophthalmology Mallinckrodt Award and the Stanley M. Truhlsen Family Foundation, Inc. The funding organization had no role in the design or conduct of this research. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Annals of Eye Science for the series “Innovations in the Diagnosis and Management of Uveitis”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aes.amegroups.com/article/view/10.21037/aes-21-18/coif). The series “Innovations in the Diagnosis and Management of Uveitis” was commissioned by the editorial office without any funding or sponsorship. SY served as the unpaid Guest Editor of the series. SY received consulting fees from Bausch and Lomb, Adverum, and Regenxbio. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Heiden D, Ford N, Wilson D, et al. Cytomegalovirus retinitis: the neglected disease of the AIDS pandemic. PLoS Med 2007;4:e334. [Crossref] [PubMed]
- Yen M, Chen J, Ausayakhun S, et al. Retinal detachment associated with AIDS-related cytomegalovirus retinitis: risk factors in a resource-limited setting. Am J Ophthalmol 2015;159:185-92. [Crossref] [PubMed]
- Stewart MW. Optimal management of cytomegalovirus retinitis in patients with AIDS. Clin Ophthalmol 2010;4:285-99. [Crossref] [PubMed]
- Jabs DA, Martin BK, Forman MS, et al. Mortality associated with resistant cytomegalovirus among patients with cytomegalovirus retinitis and AIDS. Ophthalmology 2010;117:128-132.e2. [Crossref] [PubMed]
- Sobrin L, Foster CS. Cytomegalovirus retinitis after one decade of HAART. Int Ophthalmol Clin 2007;47:155-64. [Crossref] [PubMed]
- Thorne JE, Jabs DA, Kempen JH, et al. Incidence of and risk factors for visual acuity loss among patients with AIDS and cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Ophthalmology 2006;113:1432-40. [Crossref] [PubMed]
- Bagheri N, Wajda B, Calvo C, et al. The Wills Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease. Lippincott Williams & Wilkins, 2016.
- Smith B, Regillo C. How to Treat Cytomegalovirus Retinitis [Internet]. American Academy of Ophthalmology. 2005 [cited 2019 Oct 28]. Available online: https://www.aao.org/eyenet/article/how-to-treat-cytomegalovirus-retinitis
- Kim L, Palestine A, Ballard B, et al. CMV Retinitis - EyeWiki [Internet]. [cited 2019 Nov 2]. Available online: https://eyewiki.aao.org/CMV_Retinitis
- Jouan M, Katlama C. Management of CMV retinitis in the era of highly active antiretroviral therapy. Int J Antimicrob Agents 1999;13:1-7. [Crossref] [PubMed]
- Abu El-Asrar AM, Herbort CP, Tabbara KF. Differential diagnosis of retinal vasculitis. Middle East Afr J Ophthalmol 2009;16:202-18. [PubMed]
- Wu XN, Lightman S, Tomkins-Netzer O. Viral retinitis: diagnosis and management in the era of biologic immunosuppression: A review. Clin Exp Ophthalmol 2019;47:381-95. [Crossref] [PubMed]
- Cunningham ET Jr, Hubbard LD, Danis RP, et al. Proportionate topographic areas of retinal zones 1, 2, and 3 for use in describing infectious retinitis. Arch Ophthalmol 2011;129:1507-8. [Crossref] [PubMed]
- Ausayakhun S, Keenan JD, Ausayakhun S, et al. Clinical features of newly diagnosed cytomegalovirus retinitis in northern Thailand. Am J Ophthalmol 2012;153:923-931.e1. [Crossref] [PubMed]
- Kempen JH, Jabs DA, Dunn JP, et al. Retinal detachment risk in cytomegalovirus retinitis related to the acquired immunodeficiency syndrome. Arch Ophthalmol 2001;119:33-40. [PubMed]
- Arantes TE, Garcia CR, Saraceno JJ, et al. Clinical features and outcomes of AIDS-related cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Arq Bras Oftalmol 2010;73:16-21. [Crossref] [PubMed]
- Ho M, Invernizzi A, Zagora S, et al. Presenting Features, Treatment and Clinical Outcomes of Cytomegalovirus Retinitis: Non-HIV Patients Vs HIV Patients. Ocul Immunol Inflamm 2020;28:651-8. [Crossref] [PubMed]
- Schneider EW, Elner SG, van Kuijk FJ, et al. Chronic retinal necrosis: cytomegalovirus necrotizing retinitis associated with panretinal vasculopathy in non-HIV patients. Retina 2013;33:1791-9. [Crossref] [PubMed]
- Pearce WA, Yeh S, Fine HF. Management of Cytomegalovirus Retinitis in HIV and Non-HIV Patients. Ophthalmic Surg Lasers Imaging Retina 2016;47:103-7. [Crossref] [PubMed]
- Shapira Y, Mimouni M, Vishnevskia-Dai V. Cytomegalovirus retinitis in HIV-negative patients - associated conditions, clinical presentation, diagnostic methods and treatment strategy. Acta Ophthalmol 2018;96:e761-7. [Crossref] [PubMed]
- Davis JL, Haft P, Hartley K. Retinal arteriolar occlusions due to cytomegalovirus retinitis in elderly patients without HIV. J Ophthalmic Inflamm Infect 2013;3:17. [Crossref] [PubMed]
- Kim DY, Jo J, Joe SG, et al. Comparison of visual prognosis and clinical features of cytomegalovirus retinitis in HIV and non-HIV patients. Retina 2017;37:376-81. [Crossref] [PubMed]
- Jabs DA, Van Natta ML, Thorne JE, et al. Course of cytomegalovirus retinitis in the era of highly active antiretroviral therapy: 2. Second eye involvement and retinal detachment. Ophthalmology 2004;111:2232-9. [Crossref] [PubMed]
- Sugar EA, Jabs DA, Ahuja A, et al. Incidence of cytomegalovirus retinitis in the era of highly active antiretroviral therapy. Am J Ophthalmol 2012;153:1016-24.e5. [Crossref] [PubMed]
- Jabs DA, Ahuja A, Van Natta M, et al. Course of cytomegalovirus retinitis in the era of highly active antiretroviral therapy: five-year outcomes. Ophthalmology 2010;117:2152-61.e1. [Crossref] [PubMed]
- Jacobson MA, Zegans M, Pavan PR, et al. Cytomegalovirus retinitis after initiation of highly active antiretroviral therapy. Lancet 1997;349:1443-5. [Crossref] [PubMed]
- Hoover DR, Saah AJ, Bacellar H, et al. Clinical manifestations of AIDS in the era of pneumocystis prophylaxis. Multicenter AIDS Cohort Study. N Engl J Med 1993;329:1922-6. [Crossref] [PubMed]
- Holland GN, Vaudaux JD, Shiramizu KM, et al. Characteristics of untreated AIDS-related cytomegalovirus retinitis. II. Findings in the era of highly active antiretroviral therapy (1997 to 2000). Am J Ophthalmol 2008;145:12-22. [Crossref] [PubMed]
- Tun N, London N, Kyaw MK, et al. CMV retinitis screening and treatment in a resource-poor setting: three-year experience from a primary care HIV/AIDS programme in Myanmar. J Int AIDS Soc 2011;14:41. [Crossref] [PubMed]
- Port AD, Alabi RO, Koenig L, et al. Cytomegalovirus retinitis in the post-cART era. Curr Ophthalmol Rep 2018;6:133-44. [Crossref] [PubMed]
- Yeung I, Downes K, Cunningham E, et al. CMV retinitis: Reduced incidence, still a threat [Internet]. Reviewofophthalmology.com. 2016 [cited 2019 Nov 4]. Available online: https://www.reviewofophthalmology.com/article/cmv-retinitis-reduced-incidence-still-a-threat
- Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV [Internet]. Accessed 20 Nov 2021. Available online: https://clinicalinfo.hiv.gov/sites/default/files/guidelines/documents/AdultOITablesOnly.pdf
- Tan BH. Cytomegalovirus Treatment. Curr Treat Options Infect Dis 2014;6:256-70. [Crossref] [PubMed]
- Katlama C. Management of CMV retinitis in HIV infected patients. Genitourin Med 1997;73:169-73. [Crossref] [PubMed]
- Munro M, Yadavalli T, Fonteh C, et al. Cytomegalovirus Retinitis in HIV and Non-HIV Individuals. Microorganisms 2019;8:55. [Crossref] [PubMed]
- Chen H, Beardsley GP, Coen DM. Mechanism of ganciclovir-induced chain termination revealed by resistant viral polymerase mutants with reduced exonuclease activity. Proc Natl Acad Sci U S A 2014;111:17462-7. [Crossref] [PubMed]
- Biron KK. Antiviral drugs for cytomegalovirus diseases. Antiviral Res 2006;71:154-63. [Crossref] [PubMed]
- Drew WL, Ives D, Lalezari JP, et al. Oral ganciclovir as maintenance treatment for cytomegalovirus retinitis in patients with AIDS. Syntex Cooperative Oral Ganciclovir Study Group. N Engl J Med 1995;333:615-20. [Crossref] [PubMed]
- Martin DF, Sierra-Madero J, Walmsley S, et al. A controlled trial of valganciclovir as induction therapy for cytomegalovirus retinitis. N Engl J Med 2002;346:1119-26. [Crossref] [PubMed]
- Paya C, Humar A, Dominguez E, et al. Efficacy and safety of valganciclovir vs. oral ganciclovir for prevention of cytomegalovirus disease in solid organ transplant recipients. Am J Transplant 2004;4:611-20. [Crossref] [PubMed]
- Boivin G, Gilbert C, Gaudreau A, et al. Rate of emergence of cytomegalovirus (CMV) mutations in leukocytes of patients with acquired immunodeficiency syndrome who are receiving valganciclovir as induction and maintenance therapy for CMV retinitis. J Infect Dis 2001;184:1598-602. [Crossref] [PubMed]
- Jabs DA, Enger C, Forman M, et al. Incidence of foscarnet resistance and cidofovir resistance in patients treated for cytomegalovirus retinitis. The Cytomegalovirus Retinitis and Viral Resistance Study Group. Antimicrob Agents Chemother 1998;42:2240-4. [Crossref] [PubMed]
- Cytomegalovirus (CMV) culture results, drug resistance, and clinical outcome in patients with AIDS and CMV retinitis treated with foscarnet or ganciclovir. Studies of Ocular Complications of AIDS (SOCA) in collaboration with the AIDS Clinical Trial Group. J Infect Dis 1997;176:50-8. [Crossref] [PubMed]
- Shafran SD, Conly JM. Treatment of cytomegalovirus retinitis: A growing number of options. Can J Infect Dis 1996;7:353-5. [Crossref] [PubMed]
- Studies of Ocular Complications of AIDS Research Group. Mortality in patients with the acquired immunodeficiency syndrome treated with either foscarnet or ganciclovir for cytomegalovirus retinitis. N Engl J Med 1992;326:213-20. [Crossref] [PubMed]
- Avery RK, Arav-Boger R, Marr KA, et al. Outcomes in Transplant Recipients Treated With Foscarnet for Ganciclovir-Resistant or Refractory Cytomegalovirus Infection. Transplantation 2016;100:e74-80. [Crossref] [PubMed]
- Studies of Ocular Complications of AIDS Research Group. The ganciclovir implant plus oral ganciclovir versus parenteral cidofovir for the treatment of cytomegalovirus retinitis in patients with acquired immunodeficiency syndrome: The Ganciclovir Cidofovir Cytomegalovirus Retinitis Trial. Am J Ophthalmol 2001;131:457-67. [Crossref] [PubMed]
- Vadlapudi AD, Vadlapatla RK, Mitra AK. Current and emerging antivirals for the treatment of cytomegalovirus (CMV) retinitis: an update on recent patents. Recent Pat Antiinfect Drug Discov 2012;7:8-18. [Crossref] [PubMed]
- Erice A. Resistance of human cytomegalovirus to antiviral drugs. Clin Microbiol Rev 1999;12:286-97. [Crossref] [PubMed]
- Smith IL, Cherrington JM, Jiles RE, et al. High-level resistance of cytomegalovirus to ganciclovir is associated with alterations in both the UL97 and DNA polymerase genes. J Infect Dis 1997;176:69-77. [Crossref] [PubMed]
- Lurain NS, Chou S. Antiviral drug resistance of human cytomegalovirus. Clin Microbiol Rev 2010;23:689-712. [Crossref] [PubMed]
- Chou S. Foscarnet resistance mutations mapping to atypical domains of the cytomegalovirus DNA polymerase gene. Antiviral Res 2017;138:57-60. [Crossref] [PubMed]
- Martin BK, Ricks MO, Forman MS, et al. Change over time in incidence of ganciclovir resistance in patients with cytomegalovirus retinitis. Clin Infect Dis 2007;44:1001-8. [Crossref] [PubMed]
- Deayton J, Mocroft A, Wilson P, et al. Loss of cytomegalovirus (CMV) viraemia following highly active antiretroviral therapy in the absence of specific anti-CMV therapy. AIDS 1999;13:1203-6. [Crossref] [PubMed]
- Jabs DA, Martin BK, Forman MS, et al. Cytomegalovirus (CMV) blood DNA load, CMV retinitis progression, and occurrence of resistant CMV in patients with CMV retinitis. J Infect Dis 2005;192:640-9. [Crossref] [PubMed]
- Tsui E, Gonzales JA, Shantha JG, et al. Letermovir for the Management of Cytomegalovirus-associated Uveitis. Ocul Immunol Inflamm 2021;29:169-74. [Crossref] [PubMed]
- Fan JJ, Tao Y, Hwang DK. Comparison of intravitreal ganciclovir monotherapy and combination with foscarnet as initial therapy for cytomegalovirus retinitis. Int J Ophthalmol 2018;11:1638-42. [PubMed]
- Choopong P, Vivittaworn K, Konlakij D, et al. Treatment outcomes of reduced-dose intravitreal ganciclovir for cytomegalovirus retinitis. BMC Infect Dis 2016;16:164. [Crossref] [PubMed]
- Qian Z, Li H, Tao Y, et al. Initial intravitreal injection of high-dose ganciclovir for cytomegalovirus retinitis in HIV-negative patients. BMC Ophthalmol 2018;18:314. [Crossref] [PubMed]
- Jabs DA, Ahuja A, Van Natta M, et al. Comparison of treatment regimens for cytomegalovirus retinitis in patients with AIDS in the era of highly active antiretroviral therapy. Ophthalmology 2013;120:1262-70. [Crossref] [PubMed]
- Martin DF, Kuppermann BD, Wolitz RA, et al. Oral ganciclovir for patients with cytomegalovirus retinitis treated with a ganciclovir implant. Roche Ganciclovir Study Group. N Engl J Med 1999;340:1063-70. [Crossref] [PubMed]
- Berenguer J, González J, Pulido F, et al. Discontinuation of secondary prophylaxis in patients with cytomegalovirus retinitis who have responded to highly active antiretroviral therapy. Clin Infect Dis 2002;34:394-7. [Crossref] [PubMed]
- Tabatabaei SA, Cheraqpour K, Pour EK, et al. Long-term prophylaxis in an immunocompetent patient with Cytomegalovirus retinitis: a case report and review of literature. J Ophthalmic Inflamm Infect 2020;10:16. [Crossref] [PubMed]
- Jabs DA, Van Natta ML, Holland GN, et al. Cytomegalovirus Retinitis in Patients With Acquired Immunodeficiency Syndrome After Initiating Antiretroviral Therapy. Am J Ophthalmol 2017;174:23-32. [Crossref] [PubMed]
- Jabs DA, Bolton SG, Dunn JP, et al. Discontinuing anticytomegalovirus therapy in patients with immune reconstitution after combination antiretroviral therapy. Am J Ophthalmol 1998;126:817-22. [Crossref] [PubMed]
- Rhegmatogenous retinal detachment in patients with cytomegalovirus retinitis: the Foscarnet-Ganciclovir Cytomegalovirus Retinitis Trial. The Studies of Ocular Complications of AIDS (SOCA) Research Group in Collaboration with the AIDS Clinical Trials Group (ACTG). Am J Ophthalmol 1997;124:61-70. [Crossref] [PubMed]
Cite this article as: Ude IN, Yeh S, Shantha JG. Cytomegalovirus retinitis in the highly active anti-retroviral therapy era. Ann Eye Sci 2022;7:5.


