
Periocular necrotising fasciitis after traumatic laceration and concurrent COVID-19 infection: a case report
Introduction
Necrotising fasciitis (NF) is a rare but severe necrotising infection of the subcutaneous tissues. NF is often preceded by trauma or recent surgery (1). Four types of NF have been described, classified as Type I to IV, with Types I and II being the most common (2). Group A haemolytic Streptococcus and/or Staphylococcus aureus, are often the identified pathogens, however other aerobic or anaerobic pathogens may be implicated (3). We present a case of a 33-year-old female, with a concurrent COVID-19 infection, who developed right-sided periocular NF following a traumatic injury. We present the following case in accordance with the CARE reporting checklist (available at https://aes.amegroups.com/article/view/10.21037/aes-22-52/rc).
Case presentation
A 33-year-old previously healthy female patient presented to our ophthalmology clinic with right-sided progressive periocular swelling and pain.
Two days prior, she had been involved in a physical altercation and sustained a laceration to the right superolateral brow from a clenched fist. She presented to the emergency department (ED) two days later with an enlarging area of erythema and swelling, along with a fever. Initial computed tomography (CT) conducted prior to admission, demonstrated gas locules in the lower lid pre-septal space and temporal fascia extending to the anterior temporal fossa, for which she was referred for ophthalmic management.
This patient had a concurrent COVID-19 infection, with the virus first detected on nasopharyngeal polymerase chain reaction (PCR) swab thirteen days prior to presentation. A repeat COVID-19 nasopharyngeal PCR swab detected the virus, with high viral load, again at presentation. The patient received their first dose of the Pfizer Comirnaty COVID-19 vaccination two days before the virus was first detected. She had mild respiratory symptoms, but did not have an oxygen requirement. She had no other diagnosed medical conditions.
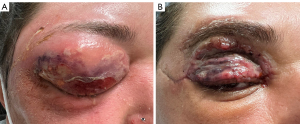
On examination, right visual acuity (VA) was at least 6/120 and left best corrected VA was 6/7.5 with pin hole. There was a −1 restriction of upgaze and abduction in right eye movements, whilst left eye extraocular movements were normal. There was no relative afferent pupillary defect. External examination revealed a large bulbous collection of the right upper lid with fluctuance and overlying erythema, while a communicating sinus drained frank pus from the superolateral brow (Figure 1). Slit lamp examination, although limited by profound swelling, was otherwise unremarkable. Initial laboratory investigations revealed a white cell count (WCC) of 21.84×10³/µL with neutrophilia and a C-reactive protein (CRP) of 400.4 mg/L. Blood cultures showed no growth.
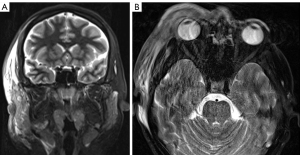
Due to the significant suspicion for NF, the patient was commenced on intravenous (IV) meropenem, clindamycin and vancomycin, and planned for urgent surgical exploration and debridement with the assistance of the Plastics and Reconstructive Surgery unit. Pre-operative T2-weighted magnetic resonance imaging (MRI) demonstrated fascial hyperintensity involving the pre-septal tissues and extending to the anterior temporal fossa (Figure 2A,2B).
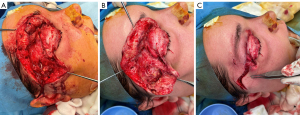
Initial debridement demonstrated right upper lid necrosis involving the dermal and pre-septal layers, including the orbicularis, but sparing the tarsus (Figure 3A-3C). There was a large amount of grossly necrotic tissue primarily confined to the pre-septal space of the right upper and lower lids with only minimal orbital extension inferior to the lacrimal gland. There was superficial and deep temporal fascia necrosis extending to the anterior temporal fossa. Streptococcus Pyogenes was isolated as the causative organism from culture of the pus and necrotic tissue obtained from debridement. Post-operatively, the patient experienced hypotension without tachycardia or fever, likely due to post-operative septic shock. 48 hours after her initial debridement, she underwent a relook debridement demonstrating minimal further necrotic tissue in the previously noted surgical planes, with no necrotic extension. With consideration of the causative organism, the antibiotics regime was rationalised to IV benzylpenicillin only.
Following the two surgical debridements and an ongoing course of IV antibiotics, the patient remained stable and continued to improve for the remainder of her admission, with down-trending WCC and CRP levels. The periocular defects were repaired with a right sided brow adipo-fascial flap based on the supratrochlear artery, browpexy and dual full thickness skin grafts on the right upper lid and flap (Figure 1B). There were no complications of reconstruction, and the patient was satisfied with the cosmetic outcome at discharge. The patient had no visual compromise and was discharged twelve days post admission to a community hospital for completion of her IV antibiotics.
At one month follow-up, the patient remained on oral antibiotics, with all wound sites healing well and no evidence of infection. The patient demonstrated fixed ptosis, 2 mm blink lagophthalmos and had no levator or frontalis function, with further consideration of ptosis repair at her next three-monthly review.
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Periocular NF has been well documented within the literature (3-6). It is a rare, but life-threatening soft-tissue infection. It is characterised by fulminant spread of inflammation and necrosis of skin, subcutaneous fat and fascia. While more common in diabetic and immunocompromised patients after minor trauma, burns and insect bites, NF may also develop in non-traumatised skin (1,5). NF may present with a wide range of clinical signs and symptoms, making early diagnosis challenging. Clinical features include pain out of proportion to clinical findings, skin necrosis, oedema extending beyond the visible erythema, poorly demarcated erythema and crepitus (7). Systemic symptoms such as fever and shock are usually present, however their absence does not exclude NF (8). Early intervention is vital with a significant reduction in morbidity and mortality, with mortality rates of untreated NF ranging between 30–70% (5). In patients with periocular NF, mortality rate has been reported to be 10%, and it has been suggested that periocular NF has a better prognosis compared to NF affecting other locations (1,4,5). Management involves early and aggressive surgical debridement of necrotic tissue with appropriate antibiotics. However, conservative approaches, involving treatment with antibiotics only, have also been reported within the literature (3,4). Reported complications of periocular NF include central retinal artery occlusion, with loss of vision and exenteration for deep orbital extension (6).
COVID-19 infection can lead to an impaired immune system during the period of disease, as it damages lymphocytes, especially B-cells, T-cells and NK-cells (9,10). Bacterial, viral and fungal co-infections have been described in the literature and are reported to significantly influence COVID-19 progression and prognosis, especially in patients with severe disease (10). Infection of intestinal cells with COVID-19 can reportedly lead to an altered intestinal and airway microbiota predisposing to co-infection, however, such changes have not been reported on skin flora (10,11). Nevertheless, cases of NF with concurrent COVID-19 infection have been reported in recent literature. These case reports demonstrate involvement of preperitoneal and retroperitoneal, breast, neck and upper extremity NF (12-15).
To our knowledge, periocular NF associated with a concurrent COVID-19 infection has not yet been reported. The relationship between periocular NF and concurrent COVID-19 infection remains to be investigated. However, it is known that COVID-19 may increase susceptibility to developing overwhelming infection, and delays in patient presentation may also lead to serious health-related emergencies. This is significant as timely assessment of NF, and initiation of appropriate treatment, can influence prognosis, patient morbidity and mortality (13).
Case reports have suggested that concomitant COVID-19 infection may complicate early management and prognosis of NF, due to delayed presentation and COVID-19 associated immunocompromise (12-15). It has been hypothesised within these case reports, that lymphopenia associated with COVID-19 infection, although not observed in this patient, may predispose patients to developing NF and complicate secondary bacterial superinfections (12,13). Furthermore, within Sankaranarayanan et al.’s case report of upper limb NF, it has been hypothesised that hypercoagulability associated with COVID-19, may have contributed to the onset and/or progression of acute limb ischemia, and the subsequent gangrenous infection (14).
In conclusion, we report a unique case of periocular necrotisiing fasciitis with concurrent COVID-19 infection. Several hypotheses regarding the related associations and pathogenesis of NF with COVID-19 have been proposed and explored. However, the significance of COVID-19 and necrotising superinfection remains to be explored, with literature lacking in this area.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://aes.amegroups.com/article/view/10.21037/aes-22-52/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aes.amegroups.com/article/view/10.21037/aes-22-52/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Misiakos EP, Bagias G, Patapis P, et al. Current concepts in the management of necrotizing fasciitis. Front Surg 2014;1:36. [Crossref] [PubMed]
- Davoudian P, Flint NJ. Necrotizing fasciitis. CEACCP 2012;12:245-50. [Crossref]
- Poitelea C, Wearne MJ. Periocular necrotising fasciitis--a case report. Orbit 2005;24:215-7. [Crossref] [PubMed]
- Rajak SN, Figueira EC, Haridas AS, et al. Periocular necrotising fasciitis: a multicentre case series. Br J Ophthalmol 2016;100:1517-20. [Crossref] [PubMed]
- Tambe K, Tripathi A, Burns J, et al. Multidisciplinary management of periocular necrotising fasciitis: a series of 11 patients. Eye (Lond) 2012;26:463-7. [Crossref] [PubMed]
- Elner VM, Demirci H, Nerad JA, et al. Periocular necrotizing fasciitis with visual loss pathogenesis and treatment. Ophthalmology 2006;113:2338-45. [Crossref] [PubMed]
- Goh T, Goh LG, Ang CH, et al. Early diagnosis of necrotizing fasciitis. Br J Surg 2014;101:e119-25. [Crossref] [PubMed]
- Damisa J, Ahmed S, Harrison S. Necrotising fasciitis: a narrative review of the literature. Br J Hosp Med (Lond) 2021;82:1-9. [Crossref] [PubMed]
- Wang M, Luo L, Bu H, et al. One case of coronavirus disease 2019 (COVID-19) in a patient co-infected by HIV with a low CD4+ T-cell count. Int J Infect Dis 2020;96:148-50. [Crossref] [PubMed]
- Chen X, Liao B, Cheng L, et al. The microbial coinfection in COVID-19. Appl Microbiol Biotechnol 2020;104:7777-85. [Crossref] [PubMed]
- Wang H, Wang H, Sun Y, et al. Potential Associations Between Microbiome and COVID-19. Front Med (Lausanne) 2021;8:785496. [Crossref] [PubMed]
- Gheisari M, Baghani M, Ganji R, et al. Huge carbuncle leading to necrotizing fasciitis in the COVID-19 pandemic era. Clin Case Rep 2021;9:1583-6. [Crossref] [PubMed]
- Elashry MM, Alagha M, Salama M. Pre and retroperitoneal necrotizing fasciitis after COVID-19 infection: A case report. Radiol Case Rep 2021;16:2949-52. [Crossref] [PubMed]
- Sankaranarayanan S, Spielman AF, Lessard AS, et al. Upper extremity necrotizing fasciitis in a Covid-19 patient. Case Reports Plast Surg Hand Surg 2022;9:46-51. [Crossref] [PubMed]
- Kamagate N, DeVito R. Necrotizing Soft Tissue Infection of the Breast during COVID-19 Pandemic. Case Rep Surg 2020;2020:8876475. [Crossref] [PubMed]
Cite this article as: Ang T, Rogerson T, Nagolla T, Caplash Y, Selva D. Periocular necrotising fasciitis after traumatic laceration and concurrent COVID-19 infection: a case report. Ann Eye Sci 2023;8:2.


